Abstract
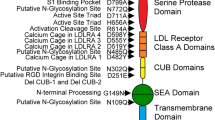
Hepatocyte growth factor activator inhibitor type I (HAI-1) is a membrane-bound, serine protease inhibitor with two protease-inhibitory domains (Kunitz domain I and II). HAI-1 is known as a physiological inhibitor of a membrane-bound serine protease, matriptase. Paradoxically, however, HAI-1 has been found to be required for the extracellular appearance of the protease in an expression system using a monkey kidney COS-1 cell line. In the present study, we show using COS-1 cells that co-expression of recombinant variants of HAI-1 with the inhibition activity toward matriptase, including a variant consisting only of Kunitz domain I (the domain responsible for inhibition of matriptase), allowed for the appearance of this protease in the conditioned medium, whereas that of the variants without the activity did not. These findings suggest that the inhibition activity toward matriptase is critical for the extracellular appearance of protease in COS-1 cells.





Similar content being viewed by others
Abbreviations
- HAI-1:
-
Hepatocyte growth factor activator inhibitor type I
- HGF:
-
Hepatocyte growth factor
- HRP:
-
Horseradish peroxidase
- LDLRA domain:
-
Low-density lipoprotein receptor A domain
- MMPs:
-
Matrix metalloproteinases
References
Bugge TH, List K, Szabo R (2007) Matriptase-dependent cell surface proteolysis in epithelial development and pathogenesis. Front Biosci 12:5060–5070
Carney TJ, von der hardt S, Sonntag C et al (2007) Inactivation of serine protease Matriptase1a by its inhibitor Hai1 is required for epithelial integrity of the zebrafish epidermis. Development 134:3461–3471
Darragh MR, Bhatt AS, Craik CS (2008) MT-SP1 proteolysis and regulation of cell-microenvironment interactions. Front Biosci 13:528–539
Denda K, Shimomura T, Kawaguchi T et al (2002) Functional characterization of Kunitz domains in hepatocyte growth factor activator inhibitor type 1. J Biol Chem 277:14053–14059
Désilets A, Béliveau F, Vandal G et al (2008) Mutation of G827R in matriptase causing autosomal recessive ichthyosis with hypotrichosis yields an inactive protease. J Biol Chem 283:10535–10542
Kataoka H, Suganuma T, Shimomura T et al (1999) Distribution of hepatocyte growth factor activator inhibitor type-1 (HAI-1) in human tissues: cellular surface localization of HAI-1 in simple columnar epithelium and its modulated expression in injured and regenerative tissues. J Histochem Cytochem 47:673–682
Kilpatrick LM, Harris RL, Owen KA et al (2006) Initiation of plasminogen activation on the surface of monocytes expressing the type II transmembrane serine protease matriptase. Blood 108:2616–2623
Kim MG, Chen C, Lyu MS et al (1999) Cloning and chromosomal mapping of a gene isolated from thymic stromal cells encoding a new mouse type II membrane serine protease, epithin, containing four LDL receptor modules and two CUB domains. Immunogenetics 49:420–428
Kim C, Cho Y, Kang CH et al (2005) Filamin is essential for shedding of the transmembrane serine protease, epithin. EMBO Rep 6:1045–1051
Kojima K, Tsuzuki S, Fushiki T et al (2008) Roles of functional and structural domains of hepatocyte growth factor activator inhibitor type 1 in the inhibition of matriptase. J Biol Chem 283:2478–2487
Kojima K, Tsuzuki S, Fushiki T et al (2009a) The activity of a type II transmembrane serine protease, matriptase, is dependent solely on the catalytic domain. Biosci Biotechnol Biochem 73:454–456
Kojima K, Tsuzuki S, Fushiki T et al. (2009b) Role of the stem domain of matriptase in the interaction with its physiological inhibitor, hepatocyte growth factor activator inhibitor type I. J Biochem 145:783–790
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Lee S, Dickson RB, Lin CY (2000) Activation of hepatocyte growth factor and urokinase/plasminogen activator by matriptase, an epithelial membrane serine protease. J Biol Chem 275:36720–36725
Lee MS, Tseng IS, Wang Y et al (2007) Autoactivation of matriptase in vitro: requirement for biomembrane and LDL receptor domain. Am J Physiol Cell Physiol 293:C95–C105
Lin CY, Tseng IC, Chou FP et al (2008) Zymogen activation, inhibition, and ectodomain shedding of matriptase. Front Biosci 13:621–635
Mathias JR, Dodd ME, Walters KB et al (2007) Live imaging of chronic inflammation caused by mutation of zebrafish Hai-1. J Cell Sci 120:3372–3383
Miyake Y, Yasumoto M, Tsuzuki S et al. (2009) Activation of a membrane-bound serine protease matriptase on the cell surface. J Biochem doi:10.1093/jb/mvp066
Miyazawa K, Shimomura T, Kitamura A et al (1993) Molecular cloning and sequence analysis of the cDNA for a human serine protease reponsible for activation of hepatocyte growth factor. Structural similarity of the protease precursor to blood coagulation factor XII. J Biol Chem 268:10024–10028
Oberst MD, Singh B, Ozdemirli M et al (2003a) Characterization of matriptase expression in normal human tissues. J Histochem Cytochem 51:1017–1025
Oberst MD, Williams CA, Dickson RB et al (2003b) The activation of matriptase requires its noncatalytic domains, serine protease domain, and its cognate inhibitor. J Biol Chem 278:26773–26779
Oberst MD, Chen LY, Kiyomiya K et al (2005) HAI-1 regulates activation and expression of matriptase, a membrane-bound serine protease. Am J Physiol Cell Physiol 289:C462–C470
Paroutis P, Touret N, Grinstein S (2004) The pH of the secretory pathway: measurement, determinants, and regulation. Physiology 19:207–215
Satomi S, Yamasaki Y, Tsuzuki S et al (2001) A role for membrane-type serine protease (MT-SP1) in intestinal epithelial turnover. Biochem Biophys Res Commun 287:995–1002
Shimomura T, Denda K, Kitamura A et al (1997) Hepatocyte growth factor activator inhibitor, a novel Kunitz-type serine protease inhibitor. J Biol Chem 272:6370–6376
Shimomura T, Denda K, Kawaguchi T et al (1999) Multiple sites of proteolytic cleavage to release soluble forms of hepatocyte growth factor activator inhibitor type 1 from a transmembrane form. J Biochem 126:821–828
Szabo R, Hobson JP, List K et al (2008) Potent inhibition and global co-localization implicate the transmembrane Kunitz-type serine protease inhibitor hepatocyte growth factor activator inhibitor-2 in the regulation of epithelial matriptase activity. J Biol Chem 283:29495–29504
Takeuchi T, Shuman MA, Craik CS (1999) Reverse biochemistry: Use of macromolecular protease inhibitors to dissect complex biological processes and identify a membrane-type serine protease in epithelial cancer and normal tissue. Proc Natl Acad Sci USA 96:11054–11061
Tsuzuki S, Murai N, Miyake Y et al (2005) Evidence for the occurrence of membrane-type serine protease 1/matriptase on the basolateral sides of enterocytes. Biochem J 388:679–687
Yamasaki Y, Satomi S, Murai N et al (2003) Inhibition of membrane-type serine protease 1/matriptase by natural and synthetic protease inhibitors. J Nutr Sci Vitaminol (Tokyo) 49:27–32
Zhang T, Cai X, Schlegelberger B et al (1998) Assignment1 of human putative tumor suppressor genes ST13 (alias SNC6) and ST14 (alias SNC19) to human chromosome bands 22q13 and 11q24–>q25 by in situ hybridization. Cytogenet Cell Genet 83:56–57
Acknowledgments
This study was supported in part by Grants-in-Aid for Scientific Research (Nos. 14658203 and 17380065 to K. I.) from the Japan Society of the Promotion of Sciences. We thank K. Kojima and Seiya Mochida for their technical assistance and advice.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Miyake, Y., Tsuzuki, S., Yasumoto, M. et al. Requirement of the activity of hepatocyte growth factor activator inhibitor type 1 for the extracellular appearance of a transmembrane serine protease matriptase in monkey kidney COS-1 cells. Cytotechnology 60, 95–103 (2009). https://doi.org/10.1007/s10616-009-9219-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10616-009-9219-7