Abstract
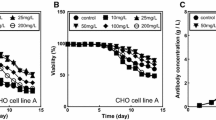
We developed a substitute for serum to produce fed-batch cultures of hybridoma cells in serum-free medium and confirmed that the cells could be successfully cultivated this way. Our substitute consisted of 12 components. The specific production rates of lactate and ammonia, which are harmful byproducts from the cells, were significantly reduced compared with a conventional serum-containing batch culture. This reduction led to a higher cell concentration and a longer production lifetime. As a result, the final concentration of monoclonal antibody was 400 mg/L, or five times greater than that in the conventional serum-containing batch culture. The developed substitute is expected to enable fed-batch cultivation in a serum-free condition.




Similar content being viewed by others
Abbreviations
- ΔVF :
-
Volume of feeding medium fed to the reactor since the nth sample was taken, L
- β:
-
Total stoichiometric coefficient of glucose, amino acids, and vitamins, nmole cell−1
- Nt :
-
Total cell number in the reactor at culture time t, number of cells
- Nt,n :
-
Total cells in the reactor when the nth sample was taken, number of cells
- Ct :
-
Total concentration of glucose, amino acids, and vitamins in the supplemental medium, mM
References
Abordo EA, Minhas HS, Thornalley PJ (1999) Accumulation of alpha-oxoaldehydes during oxidative stress: a role in cytotoxicity. Biochem Pharmacol 58(4):641–648
Bibila TA, Robinson DK (1995) In pursuit of the optimal fed-batch process for monoclonal antibody production. Biotechnological Prog 11:1–13
Birch JR, Racher AJ (2006) Antibody production. Adv Drug Deliv Rev 58(5–6):671–685
De Meirleir L (2002) Defects of pyruvate metabolism and the Krebs cycle. J Child Neurol 17(Suppl 3):3S26–3S33 (discussion 3S33–3S34)
Distefano DJ, Mark GE, Robinson DK (1996) Feeding of nutrients delays apoptotic death in fed-batch cultures of recombinant NS0 myeloma cells. Biotechnol Lett 18:1067–1072
Eagle H (1955) Nutrition needs of mammalian cells in tissue culture. Science 122:501–504
Etherton TD, Chung CS, Wiggins JP (1984) Receptor-dependent and independent degradation of insulin by isolated swine adipocytes at 37°C. J Anim Sci 59:366–375
Fahrner RL, Whitney DH, Vanderlaan M, Blank GS (1999) Performance comparison of protein A affinity-chromatography sorbents for purifying recombinant monoclonal antibodies. Biotechnol Appl Biochem 30(2):121–128
Glacken MW (1987) Development of mathematical descriptions of mammalian cell culture kinetics for the optimization of fed-batch bioreactors. Ph.D. thesis, MIT, Cambridge, MA, USA
Guérin PJ, Gauthier ER (2003) Induction of cellular necrosis by the glutathione peroxidase mimetic ebselen. J Cell Biochem 89(1):203–211
Holley RW, Kiernan JA (1974) Control of the initiation of DNA synthesis in 3T3 cells: low-molecular weight nutrients. Proc Natl Acad Sci USA 71(8):2942–2945
Ishaque A, Al-Rubeai M (2002) Role of vitamins in determining apoptosis and extent of suppression by bcl–2 during hybridoma cell culture. Apoptosis 7(3):231–239
Karran P, Lindahl T (1980) Hypoxanthine in deoxyribonucleic acid: generation by heat-induced hydrolysis of adenine residues and release in free form by a deoxyribonucleic acid glycosylase from calf thymus. Biochemistry 19:6005–6011
Khoudi H, Laberge S, Ferullo JM, Bazin R, Darveau A, Castonguay Y, Allard G, Lemieux R, Vézina LP (1999) Production of a diagnostic monoclonal antibody in perennial Alfalfa plants. Biotechnol Bioeng 64:135–143
Liddell E, Weeks I (1995) Anitibody technology. Bios Scientific Publishers, UK, pp 65–130
Nakamura MM, Liew SY, Cummings CA, Brinig MM, Dieterich C, Relman DA (2006) Growth phase- and nutrient limitation-associated transcript abundance regulation in Bordetella pertussis. Infect Immun. 74(10):5537–5548
Reuveny S, Velez D, Riske F, MacMillan JD, Miller L (1985) Production of monoclonal antibodies in culture. Dev Biol Stand 60:185–197
Sismour AM, Benner SA (2005) The use of thymidine analogs to improve the replication of an extra DNA base pair: a synthetic biological system. Nucleic Acids Res 33(17):5640–5646 (Print 2005)
Sjöholm A (1993) Role of polyamines in the regulation of proliferation and hormone production by insulin-secreting cells. Am J Physiol 264(3 Pt 1):C501–C518
Spier RE (1994) Animal cell biotechnology in the 1990 s, from models to morals. In: Spier RE, Griffiths JB (eds) Animal cell biotechnology. Cambridge, UK, pp 1–43
Tirosh O, Sen CK, Roy S, Kobayashi MS, Packer L (1999) Neuroprotective effects of alpha-lipoic acid and its positively charged amide analogue. Free Radic Biol Med 26(11–12):1418–1426
Tsao YS, Cardoso AG, Condon RG, Voloch M, Lio P, Lagos JC, Kearns BG, Liu Z (2005) Monitoring Chinese hamster ovary cell culture by the analysis of glucose and lactate metabolism. J Biotechnol 118(3):316–327
Umpleby AM, Russell-Jones DL (1996) The hormonal control of protein metabolism. Baillieres Clin Endocrinol Metab 10(4):551–570
Whiteside-Carlson V, Carlson WW (1949) Studies on the effect of para-aminobenzoic acid, folic acid, and sulfanilamide on dextran synthesis by leuconostoc. J Bacteriol 58(2):143–149
Xie L, Wang DI (1994) Applications of improved stoichiometric model in medium design and fed-batch cultivation of animal cells in bioreactor. Cytotechnology 15(1–3):17–29
Yabe N, Kato M, Matsuya Y, Yamane I, Iizuka M, Takayoshi H, Suzuki K (1987) Role of iron chelators in growth-promoting effect on mouse hybridoma cells in a chemically defined medium. In vitro Cell Dev Biol 23(12):815–820
Yoshimura T, Jhee KH, Soda K (1996) Stereospecificity for the hydrogen transfer and molecular evolution of pyridoxal enzymes. Biosci Biotechnol Biochem 60(2):181–187
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shibuya, K., Haga, R. & Namba, M. A serum substitute for fed-batch culturing of hybridoma cells. Cytotechnology 57, 187–197 (2008). https://doi.org/10.1007/s10616-008-9155-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10616-008-9155-y