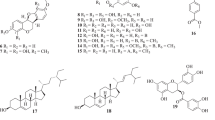
Eleven compounds, including a new compound, methyl 3-phenylpropionate-1-O-glucuronic acid methyl ester (1), four known biflavones 2–5, one known flavone (6), and five other known compounds (7–11) have been isolated from the pericarp of Garcinia yunnanensis. Their structures were elucidated by spectral analysis and by comparison of their spectral data with those in the published literatures. All the compounds were isolated from G. yunnanensis for the first time and screened for their scavenging activity against DPPH radicals. Of all the samples tested, compounds 1–6 showed significant activities with IC50 values of 1.83, 0.87, 0.17, 3.51, 0.75, and 2.21 mM, respectively.


Similar content being viewed by others
References
You-Min Ying, Kai-Ming Yu, Tian-Song Lin, Lie-Feng Ma, Ling Fang, Jian-Biao Yao, Bing-Yu Chen, Ru-Wei Wang, Wei-Guang Shan, Zhen Wang, and Zha-Jun Zhan, Chem. Nat. Compd., 53, 555 (2017).
Jih-Jung Chen, Ming-Chi Hung, Hsiang-Ruei Liao, Yueh-Hsiung Kuo, Chih-Wen Shu, Ping-Jyun Sung, Yun-Ping Lim, Wen-Lung Kuo, Lin-Yang Cheng, Sin-Ling Wang, and Jui-Jen Wu, Chem. Nat. Compd., 53, 649 (2017).
Z. Y. Wu and Y. H. Li, in: Flora Reipublicae Popularis Sinicae, Vol. 50, Science Press, Beijing, 1990.
G. Xu, C. Feng, Y. Zhou, Y. Zhou, Q. B. Han, C. F. Qiao, S. X. Huang, D. C. Chang, Q. S. Zhao, K. Q. Luo, and H. X. Xu, J. Agric. Food Chem., 56, 11144 (2008).
K. Masao, I. Yasumasa, and M. Shotaro, Tetrahedron Lett., 48, 4203 (1970).
C. G. Karanjgaokar, P. V. Fkdhalcrishnan, and K. Venkataraman, Tetrahedron Lett., 33, 3195 (1967).
Z. J. Zou, J. S. Yang, and J. H. Ju, Chin. Trad. Herb. Drugs, 37, 1303 (2006).
D. Simcikova, M. Kotik, L. Weignerova, P. Halada, H. Pelantova, K. Adamcova, and V Kren, Adv. Stnth. Catal., 357, 107 (2015).
S. C. Ha and S. W. Won, J. Nat. Prod., 64, 1579 (2001).
X. Y. Hao, N. H. Tan, and J. Zhou, Acta Bot. Yunnanica, 22, 81 (2000).
L. Q. Ana, A. D. Maria, and A. R. S. Luciana, Quim. Nova, 6, 1427 (2008).
H. S. Erge and F. Karadeniz, Int. J. Food Prop., 14, 968 (2011).
M. Irshadl, I. Ahmad1, S. J. Mehdi, H. C. Goel, and M. M. A. Rizvi, Int. J. Food Properties, 17, 179 (2014).
N. Cotelle, J. L. Bernier, J. P. Catteau, J. Pommery, and J. C. Wallet, Free Rad. Biol. Med., 20, 35 (1996).
Y. L. Lee, J. H. Yang, and J. L. Maua, Food Chem., 106, 1128 (2008).
Acknowledgment
This research was supported by the National Natural Science Foundation of China (Nos. 81273403, 81303188, and 81173485), Shanghai Municipal Commission of Health and Family Planning (ZY3-CCCX-3-5001), and Science and Technology Commission of Shanghai Municipality (15DZ2292000).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Published in Khimiya Prirodnykh Soedinenii, No. 2, March–April, 2018, pp. 194–195.
Rights and permissions
About this article
Cite this article
Fang, X., Fu, Z., Zhang, H. et al. Chemical Constituents of Garcinia yunnanensis and Their Scavenging Activity Against DPPH Radicals. Chem Nat Compd 54, 232–234 (2018). https://doi.org/10.1007/s10600-018-2310-6
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10600-018-2310-6