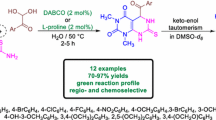
The racemic epi-cytoxazone was synthesized, starting from p-anisaldehyde, in five steps and 48% overall yield. An efficient synthesis of azido alcohols has been achieved by regioselective ring opening of glycidates using NaN3 in MeOH–H2O. The key step of the process is reductive cyclization of azide carbonates by Zn–TMSCl.

Similar content being viewed by others
References
a) H. Kakeya, M. Morishita, K. Kobinata, M. Osono, M. Ishizuka, and H. Osada, J. Antibiot., 51, 1126 (1998); b) H. Kakeya, M. Morishita, H. Koshino, T. Morita, K. Kobayashi, and H. Osada, J. Org. Chem., 64, 1052 (1999).
a) M. Seki and K. Mori, Eur. J. Org. Chem., 11, 2965 (1999); b) Y. Sakamoto, A. Shiraishi, J. Seonhee, and T. Nakata, Tetrahedron Lett., 40, 4203 (1999).
A. Grajewska and M. D. Rozwadowska, Tetrahedron: Asymmetry, 18, 803 (2007).
a) A. Madhan, A. R. Kumar, and B. V. Rao, Tetrahedron: Asymmetry, 12, 2009 (2001); b) S. G. Kim and T. H. Park, Tetrahedron: Asymmetry, 19, 1626 (2008); c) A. R. Kumar, G. Bhaskar, A. Madhan, and B. V. Rao, Synth. Commun., 33 (16), 2907 (2003).
O. Miyata, H. Asai, and T. Naito, Synlett, 12, 1915 (1999).
a) J. N. Park, Y. S. Ko, and H. Y. Koh, Tetrahedron Lett., 41, 5553 (2000); b) S. Milicevic, R. Matovic, and R. N. Saicic, Tetrahedron Lett., 45, 955 (2004); c) A. S. Paraskar and A. Sudalai, Tetrahedron, 62, 5756 (2006); d) J. Boruwa, J. C. Borah, B. Kalita, and N. C. Barua, Tetrahedron Lett., 45, 7355 (2004).
a) P. H. Carter, J. R. Laporte, P. A. Scherle, and C. P. Decicco, Bioorg. Med. Lett., 13, 1237 (2003); b) S. Matsunaga, T. Yoshida, H. Morimoto, N. Kumagai, and M. Shibasaki, J. Am. Chem. Soc., 126, 8777 (2004); c) J. D. Kim, I. S. Kim, C. H. Jin, O. P. Zee, and Y. H. Jung, Org. Lett., 7, 4025 (2005).
S. Sugiyama, S. Arai, and K. Ishii, Tetrahedron: Asymmetry, 15, 3149 (2004).
Z. Hamersak, E. Ljubovic, M. Mercep, M. Mesic, and V. Sunjic, Synthesis, 13, 1989 (2001).
a) K. C. Babu, R. N. Reddy, S. Y. Rao, P. Venkateshwarlu, and G. Madhusudhan, Synth. Commun., 42, 2624 (2012); b) N. R. Swamy, P. Karishnaiah, N. S. Reddy, and Y. Venkateswarlu, J. Carbohydr. Chem., 23 (4), 217 (2004); c) S. G. Davies, D. G. Hughes, R. L. Nicholson, A. D. Smith, and A. J. Wright, Org. Biomol. Chem., 2, 1549 (2004); d) R. K. Mishra, C. M. Coates, K. D. Revell, and E. Turos, Org. Lett., 9 (4), 575 (2007); e) J. D. Kim, I. S. Kim, C. H. Jin, O. P. Zee, and Y. H. Jung, Org. Lett., 7 (18), 4025 (2005); f) M. Carda, F. Gonzalez, R. Sanchez, and J. A. Marco, Tetrahedron: Asymmetry, 13, 1005 (2002); g) Z. T. Vujosevic, G. Petrovic, B. Rakic, R. Matovic, and R. N. Saicic, Synth. Commun., 35, 435 (2005).
M. Er and N. Coskun, J. Chem. Soc. Pak., 32 (2), 198 (2010).
M. Er and N. Coskun, Arkivoc, xii, 153 (2009).
M. Asano, C. Nagasawa, M. Suzuki, S. Nishiyama, and T. Sugai, Biosci. Biotechnol. Biochem., 1, 145 (2005).
B. M. Adger, J. V. Barkley, S. Bergeron, M. W. Cappi, B. E. Flowerdew, M. P. Jackson, R. McCague, T. C. Nugent, and S. M. Roberts, J. Chem. Soc., Perkin Trans. 1, 3501 (1997).
Acknowledgment
The Uludap University Research Fund is gratefully acknowledged for its financial support (Project No 2001-2).
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Khimiya Prirodnykh Soedinenii, No. 6, November–December, 2014, pp. 932–935.
Rights and permissions
About this article
Cite this article
Er, M., Coskun, N. A Short and Efficient Asymmetric Synthesis of (±)-epi-Cytoxazone from cis-(1R,2S,5R)-Menthyl 4-Methoxyphenylglycidates. Chem Nat Compd 50, 1071–1074 (2014). https://doi.org/10.1007/s10600-014-1161-z
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10600-014-1161-z