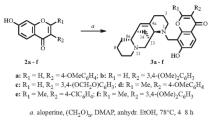
A series of 7-O-β-D-glucosaminides of isoflavone derivatives were synthesized by glycosylation of phenolic hydroxyls using solid K2CO3:CH3CN with crown-ether catalysis and K2CO3 solution (0.18 M):CHCl3 with tetrabutylammonium-bromide catalysis.
Similar content being viewed by others
References
E. Middleton, C. Kandaswami, and C. T. Theoharis, Pharmacol. Rev., 52, 673 (2000).
Z. Li, G. Ngojeh, P. DeWitt, Z. Zheng, M. Chen, B. Lainhart, V. Li, and P. Felpo, Tetrahedron Lett., 49, 7243 (2008).
W. Yang, J. Sun, W. Lu, Y. Li, L. Shan, W. Han, W.-D. Zhang, and B. Yu, J. Org. Chem., 75, 6879 (2010).
Y. Du, G. Wei, and R. J. Linhardt, Tetrahedron Lett., 44, 6887 (2003).
M. Li, X. Han, and B. Yu, Tetrahedron Lett., 43, 9467 (2002).
A. E. Zemlyakov, V. O. Kur’yanov, T. A. Chupakhina, V. Ya. Chirva, V. V. Ishchenko, M. M. Garazd, and V. P. Khilya, Chem. Nat. Compd., 38, 149 (2002).
A. Aitmambetov, L. G. Grishko, and V. P. Khilya, Chem. Nat. Compd., 29, 720 (1993).
A. Aitmambetov, L. G. Grishko, and V. P. Khilya, Chem. Nat. Compd., 29, 726 (1993).
D. Horton, Methods in Carbohydrate Chemistry, Vol. 6, Academic, New York, 1972 [Russ. transl., p. 221].
Acknowledgment
We thank Dr. Chem. Sci. A. V. Turov (Taras Shevchenko Kiev Nat. Univ.) for assistance in recording NMR spectra.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Khimiya Prirodnykh Soedinenii, No. 6, November–December, 2013, pp. 868–870.
Rights and permissions
About this article
Cite this article
Zemlyakov, A.E., Tsikalova, V.N., Zemlyakov, S.A. et al. Synthesis of N-acetylglucosaminides of Isoflavone Derivatives. Chem Nat Compd 49, 1011–1014 (2014). https://doi.org/10.1007/s10600-014-0811-5
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10600-014-0811-5