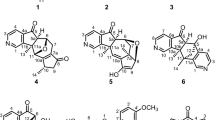
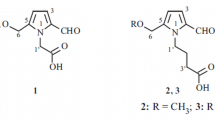
The novel pyrrole alkaloid alhagifoline A (1) together with the two known analogs pyrrolezanthine (2) and pyrrolezanthine-6-methyl ether (3) were isolated from the aerial part of Alhagi sparsifolia. Their structures were established based on spectral (HR-ESI-MS, 1 H and 13C NMR, 1 H–1 H COSY, HSQC, HMBC) data. Compounds 2 and 3 were isolated from the genus Alhagi for the first time.

Similar content being viewed by others
References
Editorial Committee of Chinese Flora, Flora Republicae Popularis Sinicae, Science Press, Beijing, 1998, p. 163.
A. Malik, Z. A. Kuliev, U. A. Akhmedov, A. D. Vdovin, and N. D. Abdullaev, Chem. Nat. Comp., 33, 174 (1997).
X. C. Su, L. Chen, and H. A. Aisa, Chem. Nat. Comp., 44, 365 (2008).
Y. P. Yang, M. J. Cheng, C. M. Teng, Y. L. Chang, I. L. Tsai, and I. S. Chen, Phytochemistry, 61, 567 (2002).
G. H. Xu, Y. H. Kim, S. J. Choo, I. J. Ryoo, J. K. Yoo, J. S. Ahn, and I. D. Yoo, Arch. Pharm. Res., 32, 1215 (2009).
Acknowledgment
The work was supported financially by the Western Chinese Academy of Sciences (XBBS200917) and the Chinese National Science Foundation for Talented Young Scientists (No. 30925045). We also thank Prof. G. M. Shen for identifying the plant material.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Khimiya Prirodnykh Soedinenii, No. 4, July–August, 2012, pp. 571–572.
Rights and permissions
About this article
Cite this article
Zou, G.A., Mansur, S., Hu, S.C. et al. Pyrrole alkaloids from Alhagi sparsifolia . Chem Nat Compd 48, 635–637 (2012). https://doi.org/10.1007/s10600-012-0330-1
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10600-012-0330-1