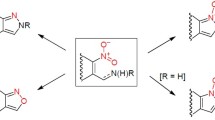
A facile one-pot synthesis of N-fused 1,2,4-triazoles from heterocyclic hydrazines and aldehydes is reported. The reaction is efficiently promoted by manganese dioxide to afford the desired products mostly in high yields and in relatively short times. The mild nature of the synthesis, cheap oxidizing agent, and short reaction time are notable advantages of the developed protocol. This protocol is effective toward various substrates having different functionalities.


Similar content being viewed by others
References
Bhadoriya, U.; Jain, D. K.; Int. J. Pharm. Sci. Res. 2016, 7, 1874.
Bhatt, A.; Singh, R. K.; Kant, R. Chem. Biol. Lett. 2017, 4, 73.
Endo, K.; Deguchi, K.; Matsunaga, H.; Tomaya, K.; Yamada, K. Bioorg. Med. Chem. 2014, 22, 3072.
Nagamatsu, T.; Fujita, T.; Endo, K. J. Chem. Soc., Perkin Trans. 1 2000, 33.
Nagamatsu, T.; Yamasaki, H.; Fujita, T.; Endo, K.; Machida, H. J. Chem. Soc., Perkin Trans. 1 1999, 3117.
Kobe, B.; Kobe, J.; Smee, D. F.; Jerman-Blazic-Dzonova, B.; Solmajer, T. Eur. J. Med. Chem. 1992, 27, 259.
Mukaiyama, H.; Nishimura, T.; Kobayashi, S.; Ozawa, T.; Kamada, N.; Komatsu, Y.; Kikuchi, S.; Oonota, H.; Kusama, H. Bioorg. Med. Chem. 2007, 15, 868.
Ozawa, T.; Tsuji, E.; Ozawa, M.; Handa, C.; Mukaiyama, H.; Nishimura, T.; Kobayashi, S.; Okazaki, K. Bioorg. Med. Chem. 2008, 16, 10311.
Scheltens, P.; Prins, N.; Lammertsma, A.; Yaqub, M.; Gouw, A; Wink, A. M.; Chu, H. M.; van Berckel, B. N. M.; Alam, J. Ann. Clin. Transl. Neurol. 2018, 5, 464.
Collin, X.; Sauleau, A.; Coulon, J. Bioorg. Med. Chem. Lett. 2003, 13, 2601.
Papakonstantinou-Garoufalias, S.; Pouli, N.; Marakos, P.; Chytyroglou-Ladas, A. Farmaco 2002, 57, 973.
De Clercq, E. J. Clin. Virol. 2004, 30, 115.
Navidpour, L.; Shadnia, H.; Shafaroodi, H.; Amini, M.; Dehpour, A. R.; Shafiee, A. Bioorg. Med. Chem. 2007, 15, 1976.
Naito, Y.; Akahoshi, F.; Takeda, S.; Okada, T.; Kajii, M.; Nishimura, H.; Sugiura, M.; Fukaya, C.; Kagitani, Y. J. Med. Chem. 1996, 39, 3019.
Saha, A. K.; Liu, L.; Simoneaux, R.; Decorte, B.; Meyer, C.; Skrzat, S.; Breslin, H. J.; Kukla, M. J.; End, D. W. Bioorg. Med. Chem. Lett. 2005, 15, 5407.
Hester, J. B., Jr.; Rudzik, A. D.; Kamdar, B. V. J. Med. Chem. 1971, 14, 1078.
Hitotsuyanagi, Y.; Motegi, S.; Fukaya, H.; Takeya, K. J. Org. Chem. 2002, 67, 3266.
Dash, C.; Shaikh, M. M.; Ghosh, P. J. Chem. Sci. 2011, 123, 97.
Holm, S. C.; Straub, B. F. Org. Prep. Proced. Int. 2011, 43, 319.
Meanwell, N. A.; Romine, J. L.; Rosenfeld, M. J.; Martin, S. W.; Trehan, A. K.; Wright, J. J. K.; Malley, M. F.; Gougoutas, J. Z.; Brassard, C. L.; Buchanan, J. O.; Federic, M. E.; Fleming, J. S.; Gamberdella, M.; Hartl, K. S.; Zavoico, G. B.; Seiler, S. M. J. Med. Chem. 1993, 36, 3884.
Aggarwal, R.; Sumran, G.; Kumar, R.; Aggarwal, A.; Parkash, V. Indian J. Heterocycl. Chem. 2006, 16, 1.
Hutchinson, J. H.; Seiders, T. J.; Stearns, B. A.; Wang, B.; Scott, J. M.; Truong, Y. P. US Patent 20070173508.
Jørgensen, M.; Bruun, A. T.; Rasmussen, L. K. WO Patent 2013034758.
Jørgensen, M.; Bruun, A. T.; Rasmussen, L. K.; Larsen, M. WO Patent 2013034755.
Kumar, D.; Chandra, S. K.; Dhillon, H.; Rao, V. S.; Varma, R. S. Green Chem. 2004, 6, 156.
Dubey, P. K.; Naidu, A.; Shiva Kumar, K. Org. Chem.: Indian J. 2008, 4, 475.
Aggarwal, R.; Sumran, G. Synth. Commun. 2006, 36, 1873.
Nitlikar, L. H.; Darandale, S. N.; Shinde, D. B. Lett. Org. Chem. 2013, 10, 348.
Graceffa, R.; Kaller, M.; La, D.; Lopez, P.; Patel, V. F.; Zhong, W. US Patent 20100120774.
Kim, D.; Wang, L.; Beconi, M.; Eiermann, G. J.; Fisher, M. H.; He, H.; Hickey, G. J.; Kowalchick, J. E.; Leiting, B.; Lyons, K.; Marsilio, F.; McCann, M. E.; Patel, R. A.; Petrov, A.; Scapin, G.; Patel, S. B.; Roy, R. S.; Wu, J. K.; Wyvratt, M. J.; Zhang, B. B.; Zhu, L.; Thornberry, N. A.; Weber, A. E. J. Med. Chem. 2005, 48, 141.
Guan, L. P.; Zhang, R. P.; Chang, Y.; Gan, X. X. Asian J. Chem. 2013, 25, 3660.
Comas, H.; Bernardinelli, G.; Swinnen, D. J. Org. Chem. 2009, 74, 5553.
Bourgeois, P.; Cantegril, R.; Chêne, A.; Gelin, J.; Mortier, J.; Moyroud, J. Synth. Commun. 1993, 23, 3195.
(a) Inturi, S. B.; Kalita, B.; Ahamed, A. J. Org. Biomol. Chem. 2016, 14, 11061. (b) Nakka, M.; Tadikonda, R.; Rayavarapu, S.; Sarakula, P.; Vidavalur, S. Synthesis, 2015, 517. (c) Li, E.; Hu, Z.; Song, L.; Yu, W.; Chang, J. Chem.–Eur. J. 2016, 22, 11022. (d) Thiel, O. R.; Achmatowicz, M. M.; Reichelt, A.; Larsen, R. D. Angew. Chem., Int. Ed. 2010, 49, 8395. (e) Srinivasan, R.; Sembian Ruso, J.; Nagarajan, N. S.; Senthil Kumaran, R.; Manickam, G. J. Heterocycl. Chem. 2016, 53, 606. (f) Ciesielski, M.; Pufky, D.; Döring, M. Tetrahedron 2005, 61, 5942. (g) Naqui, S.; Srinivasan, V. R. Indian J. Chem. 1965, 3, 162.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary information file containing 1H and 13C NMR spectra of all synthesized compounds is available at the journal website at http://link.springer.com/journal/10593.
Published in Khimiya Geterotsiklicheskikh Soedinenii, 2018, 54(12), 1111–1116
Electronic supplementary material
ESM 1
(PDF 9196 kb)
Rights and permissions
About this article
Cite this article
Bhatt, A., Singh, R.K. & Kant, R. Facile one-pot synthesis of N-fused 1,2,4-triazoles via oxidative cyclization using manganese dioxide. Chem Heterocycl Comp 54, 1111–1116 (2018). https://doi.org/10.1007/s10593-019-02400-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10593-019-02400-0