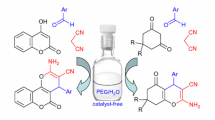
One-pot three-component reaction of arylglyoxals, 3-aryl-3-oxopropanenitriles, and 5-amino-1-aryl-3-methylpyrazoles using various solvent systems and different catalysts under reflux conditions afforded the corresponding 4-aroyl-1,6-diaryl-3-methyl-1H-pyrazolo[3,4-b]- pyridine-5-carbonitrile derivatives. The best yields (70–91%) were obtained using Al2O3 as a nanocatalyst in H2O–EtOH, 1:1, under reflux conditions. The simplicity of workup procedure, the novelty of pyrazolopyridines, green solvent system, easy preparation of a nanocatalyst, and good to excellent yields of the products are the advantages of this synthetic strategy. The structures of all products were confirmed by FT-IR, 1H and13C NMR, and mass spectral data.





Similar content being viewed by others
References
Hao, Y.; Xu, X.-P.; Chen, T.; Zhao, L.-L.; Ji, S.-J. J. Org. Biomol. Chem. 2012, 10, 724.
Tu, X.-J.; Hao, W.-J.; Ye, Q.; Wang, S.-S.; Jiang, B.; Li, G.; Tu, S.-J. J. Org. Chem. 2014, 79, 11110.
Lavecchia, G.; Berteina-Raboin, S. B.; Guillaumet, G. Tetrahedron Lett. 2004, 45, 2389.
Petrova, O. N.; Zamigajlo, L. L.; Gella, I. M.; Musatov, V. I.; Shishkina, S. V.; Shishkin, O. V.; Vashchenko, E. V.; Borisov, A. V.; Lipson, V. V. Chem. Heterocycl. Compd. 2014, 50, 514. [Khim. Geterotsikl. Soedin. 2014, 562.]
Potapov, A. Yu.; Vandyshev, D. Yu.; Kosheleva, Y. A.; Polikarchuk, V. A.; Potapov, M. A.; Shikhaliev, Kh. S. Chem. Heterocycl. Compd. 2017, 53, 207. [Khim. Geterotsikl. Soedin. 2017, 53, 207.]
Govindaraju, S.; Tabassum, S.; Khan, R.-u.-R.; Afzal Pasha, M. Chin. Chem. Lett. 2017, 28, 437.
Abdel-Monem, Y. K.; El-Enein, S. A. A.; El-Sheikh-Amer, M. M. J. Mol. Struct. 2017, 1127, 386.
Leal, B.; Afonso, I. F.; Rodrigues, C. R.; Abreu, P. A.; Garrett, R.; Carlos, L.; Pinheiro, S.; Azevedo, A. R.; Borges, J. C.; Vegi, P. F.; Santos, C. C.; da Silveira, F. C. A.; Cabral, L. M.; Frugulhetti, I. C. P. P.; Bernardino, A. M. R.; Santos, D. O.; Castro, H. C. Bioorg. Med. Chem. 2008, 16, 8196.
Melha, S. A. Arch. Pharm. 2013, 346, 912.
Chandak, N.; Kumar, S.; Kumar, P.; Sharma, C.; Aneja, K. R.; Sharma, P. K. Med. Chem. Res. 2013, 22, 5490.
de Mello, H. D.; Echevarria, A.; Bernardino, A. M.; Canto-Cavalheiro, M.; Leon, L. L. J. Med. Chem. 2004, 47, 5427.
Anand, D.; Yadav, P. K.; Patel, O. P. S.; Parmar, N.; Maurya, R. K.; Vishwakarma, P.; Raju, K. S. R.; Taneja, S.; Wahajuddin, M.; Kar, S.; Yadav, P. P. J. Med. Chem. 2017, 60, 1041.
Salem, M. S.; Ali, M. A. M. Biol. Pharm. Bull. 2016, 39, 473.
Nagender, P.; Reddy, G. M.; Kumar, R. N.; Poornachandra, Y.; Kumar, C. G.; Narsaiah, B. Bioorg. Med. Chem. Lett. 2014, 24, 2905.
Gouda, M. A. Arch. Pharm. 2012, 345, 155.
Saini, D.; Jain, S.; Kumar, A.; Jain, N. EXCLI J. 2016, 15, 730.
Mohamed, M. S.; Awad, Y. E. E. D.; El-Hallouty, S. M.; El-Araby, M. Open J. Med. Chem. 2012, 2, 78.
Lavrard, H.; Popowycz, F. Eur. J. Org. Chem. 2017, 2017, 600.
Cioc, R.; Ruijter, E.; Orru, R. Green Chem. 2014, 16, 2958.
Sarkar, S.; Das, D. K.; Khan, A. T. RSC Adv. 2014, 4, 53752.
Petrova, O. N.; Zamigajlo, L. L.; Ostras, K. S.; Shishkina, S. Sh.; Shishkin, O. V.; Borisov, A. V.; Musatov, V. I.; Shirobokova, M. G.; Lipson, V. V. Chem. Heterocycl. Compd. 2015, 51, 310. [Khim. Geterotsikl. Soedin. 2015, 51, 310.]
Satish, G.; Sharma, A.; Gadidasu, K. K.; Vedula, R. R.; Penta, S. Chem. Heterocycl. Compd. 2016, 52, 409. [Khim. Geterotsikl. Soedin. 2016, 52, 409.]
Govindaraju, S.; Tabassum, S.; Khan, R.-u.-R.; Afzal Pasha, M. Chem. Heterocycl. Compd. 2016, 52, 964. [Khim. Geterotsikl. Soedin. 2016, 52, 964.]
Poursattar Marjani, A.; Khalafy, J.; Prager, R. H. Chem. Heterocycl. Compd. 2012, 48, 931. [Khim. Geterotsikl. Soedin. 2012, 1001.]
Poursattar Marjani, A.; Khalafy, J.; Salami, F.; Mohammadlou, M. Synthesis 2015, 1656.
Poursattar Marjani, A.; Khalafy, J.; Mahmoodi, S. ARKIVOC 2016, (iii), 262.
Poursattar Marjani, A.; Khalafy, J.; Rostampoor, A. J. Heterocycl. Chem. 2017, 54, 648.
Poursattar Marjani, A.; Khalafy, J.; Chitan, M.; Mahmoodi, S. Iran. J. Chem. Chem. Eng. 2017, 36, 1.
Ezzati, M.; Khalafy, J.; Poursattar Marjani, A.; Prager, R. H. Tetrahedron 2017, 73, 6587.
Poursattar Marjani, A.; Khalafy, J.; Haghi, A. J. Heterocycl. Chem. 2017, 54, 3294.
Ganesan, A.; Heathcock, C. H. J. Org. Chem. 1993, 58, 6155.
Kordik, C. P.; Luo, C.; Zanoni, B. C.; Dax, S. L.; McNally, J. J.; Lovenberg, T. W.; Wilson, S. J.; Reitz, A. B. Bioorg Med. Chem. Lett. 2001, 11, 2283.
Lee, S.; Kim, T.; Lee, B. H.; Yoo, S.; Lee, K.; Yi, K. Y. Bioorg. Med. Chem. Lett. 2007, 17, 1291.
Riley, H. A.; Gray, A. R. In Organic Syntheses; Blatt, A. H., Ed.; Wiley & Sons: New York, 1943, Vol. 2, p 509.
Rogojan, R.; Andronescu, E.; Ghitulica, C.; Vasile, B. S. Sci. Bull. – “Politeh.” Univ. Bucharest, Ser. B 2011, 73, 1454.
The authors are grateful for support from the Urmia University.
We also thank Prof. R. H. Prager from the Flinders University of Australia for proof-reading and language editing of this article.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary information file containing 1H NMR spectra of compounds 4a–p and mass spectrum of compound is available at the journal website at http://link.springer.com/journal/10593.
Published in Khimiya Geterotsiklicheskikh Soedinenii, 2018, 54(1), 51–57
Electronic supplementary material
ESM 1
(PDF 4852 kb)
Rights and permissions
About this article
Cite this article
Arlan, F.M., Khalafy, J. & Maleki, R. One-pot three-component synthesis of a series of 4-aroyl-1,6-diaryl-3-methyl-1H-pyrazolo[3,4-b]pyridine-5-carbonitriles in the presence of aluminum oxide as a nanocatalyst. Chem Heterocycl Comp 54, 51–57 (2018). https://doi.org/10.1007/s10593-018-2229-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10593-018-2229-7