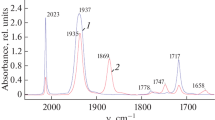
Newly synthesized indolyl(thienyl)maleimides containing naphthalimide linkers were found to exhibit weak fluorescence due to PET effect. Irradiation at 436 nm wavelength led to the formation of their cyclic isomers. The obtained compounds showed the properties of chromogenic and fluorogenic chemosensors for F– ions.





Similar content being viewed by others
References
(a) Molecular Switches; Feringa, B. L.; Browne, W. R., Eds.; Wiley: Weinheim, 2011, 2nd ed. (b) Minkin V. I. Russ. Chem. Bull., Int. Ed. 2008, 57, 687. [Izv. Akad. Nauk, Ser. Khim. 2008, 673.] (c) Organic Photochromic and Thermochromic Compounds; Crano, J. C.; Guglielmetti, R. J., Eds.; Plenum Press: New York, 1999.
(a) Andreasson, J.; Pischel, U. Chem. Soc. Rev. 2015, 44, 1053. (b) Velema, W. A.; Szymanski, W.; Feringa, B. L. J. Am. Chem. Soc. 2014, 136, 2178. (c) Zhang, J.; Zou, Q.; Tian, H. Adv. Mater. 2013, 25, 378. (d) Natali, M.; Giordani, S. Chem. Soc. Rev. 2012, 41, 4010. (e) Wang, G.; Zhang, J. J. Photochem. Photobiol., C 2012, 13, 299.
(a) Klajn, R. Chem. Soc. Rev. 2014, 43, 148. (b) Metelitsa, A. V.; Nikolaeva, O. G.; Cheprasov, A. S.; Karlutova, O. Yu.; Burtseva, A. A.; Dubonosov, A. D.; Bren, V. A.; Minkin, V. I. J. Photochem. Photobiol., A 2016, 321, 12. (c) Nikolaeva, O. G.; Gaeva, E. B.; Shepelenko, E. N.; Tsukanov, A. V.; Metelitsa, A. V.; Lukyanov, B. S.; Dubonosov, A. D.; Bren, V. A.; Minkin, V. I. Russ. J. Org. Chem. 2009, 45, 1091. [Zh. Org. Khim. 2009, 1102.] (d) Yokoyama, Y. Chem. Rev. 2000, 100, 1717. (e) Rueck-Braun, K.; Mayer, K.; Hebert, A.; Michalik, F. In CRC Handbook of Organic Photochemistry and Photobiology; Griesbeck, A.; Oelgemöller, M.; Ghetti, F., Eds.; CRC Press: Boca Raton, London, New York, 2012, p. 607. (f) Zmeeva, S. Yu.; Rybalkin, V. P.; Popova, L. L.; Tkachev, V. V.; Revinskii, Yu. V.; Tikhomirova, K. S.; Starikov, A. G.; Dubonosov, A. D.; Bren, V. A.; Aldoshin, S. M.; Minkin, V. I. Tetrahedron 2016, 72, 5776. (g) Irie, M.; Fukaminato, T.; Matsuda, K.; Kobatake, S. Chem. Rev. 2014, 114, 12174. (h) Lvov, A. G.; Shirinyan, V. Z. Chem. Heterocycl. Compd. 2016, 52, 658. [Khim. Geterotsikl. Soedin. 2016, 52, 658.] (i) Shepelenko, E. N.; Revinskii, Y. V.; Tikhomirova, K. S.; Karamov, O. G.; Dubonosov, A. D.; Bren, V. A.; Minkin, V. I. Mendeleev Commun. 2016, 26, 193. (j) Dubonosov, A. D.; Minkin, V. I.; Bren, V. A.; Popova, L. L.; Rybalkin, V. P.; Shepelenko, E. N.; Tkalina, N. N.; Tsukanov, A. V. ARKIVOC 2003, (xiii), 12. (k) Minkin, V. I.; Gribanova, T. N.; Dubonosov, A. D.; Bren, V. A.; Minyaev, R. M.; Shepelenko, E. N.; Tsukanov, A. V. Ros. Khim. Zh. 2004, 48(1), 30.
(a) Harvey, E. C.; Feringa, B. L.; Vos, J. G.; Browne, W. R.; Pryce, M. T. Coord. Chem. Rev. 2015, 282, 77. (b) Zou, Q.; Jin, J.; Xu, B.; Ding, L.; Tian, H. Tetrahedron 2011, 67, 915. (c) Nikolaeva, O. G.; Tsukanov, A. V.; Shepelenko, E. N.; Lukyanov, B. S.; Metelitsa, A. V.; Kostyrina, O. Y.; Dubonosov, A. D.; Bren, V. A.; Minkin, V. I. Int. J. Photoenergy 2009. DOI: https://doi.org/10.1155/2009/238615.
(a) Chemosensors: Principles, Strategies, and Applications; Wang, B.; Anslyn, E. V., Eds.; Wiley: Hoboken, 2011. (b) Zhou, Y.; Zhang, J. F.; Yoon, J. Chem. Rev. 2014, 114, 5511. (с) Kaur, N.; Kaur, G.; Fegade, U. A.; Singh, A.; Sahoo, S. K.; Kuwar, A. S.; Singh, N. TrAC, Trends Anal. Chem. 2017, 95, 86.
(a) Dubonosov, A. D.; Bren, V. A.; Minkin, V. I.; Shepelenko, E. N.; Tikhomirova, K. S.; Starikov, A. G.; Revinskii, Yu. V. Tetrahedron 2015, 71, 8817. (b) Shepelenko, E. N.; Makarova, N. I.; Podshibyakin, V. A.; Tikhomirova, K. S.; Dubonosov, A. D.; Metelitsa, A. V.; Bren, V. A.; Minkin, V. I. Russ. J. Org. Chem. 2017, 53, 366. [Zh. Org. Khim. 2017, 53, 368.] (c) Shepelenko, E. N.; Karamov, О. G.; Podshibyakin, V. A.; Revinskii, Yu. V.; Tikhomirova, K. S.; Dubonosov, A. D.; Bren, V. A.; Minkin, V. I. ARKIVOC 2017, (v), 196.
(a) Panchenko, P. A.; Fedorova, O. A.; Fedorov, Yu. V. Russ. Chem. Rev. 2014, 83, 155. [Usp. Khim. 2014, 83, 155.] (b) Xu, Y.; Mao, S.; Peng, H.; Wang, F.; Zhang, H.; Aderinto, S. O.; Wu, H. J. Lumin. 2017, 192, 56. (c) Zhang, H.; Liu, T.; Yin, C.; Wen, Y.; Chao, J.; Zhang, Y.; Huo, F. Spectrochim. Acta, Part A 2017, 174, 230.
(a) Makarova, N. I.; Levchenko, P. V.; Tkachev, V. V.; Shepelenko, E. N.; Metelitsa, A. V.; Rybalkin, V. P.; Popova, L. L.; Bren, V. A.; Aldoshin, S. M.; Minkin, V. I. Russ. Chem. Bull., Int. Ed. 2011, 60, 1090. [Izv. Akad. Nauk, Ser. Khim. 2011, 1065.] (b) Makarova, N. I.; Levchenko, P. V.; Shepelenko, E. N.; Metelitsa, A. V.; Kozyrev, V. S.; Rybalkin, V. P.; Bren, V. A.; Minkin, V. I. Russ. Chem. Bull., Int. Ed. 2011, 60, 1899. [Izv. Akad. Nauk, Ser. Khim. 2011, 1866.]
(a) de Silva, A. P.; Gunaratne, H. Q. N.; Gunnlaugsson, T. Tetrahedron Lett. 1998, 39, 5077. (b) Duke, R. M.; Veale, E. B.; Pfeffer, F. M.; Kruger, P. E.; Gunnlaugsson, T. Chem. Soc. Rev. 2010, 39, 3936.
(a) de Silva, A. P.; Gunaratne, H. Q. N.; Habib-Jiwan, J.-L.; McCoy, C. P.; Rice, T. E., Soumillion, J.-P. Angew. Chem., Int. Ed. 1995, 34, 1728. (b) de Silva, A. P.; Rice, T. E. Chem. Commun. 1999, 163.
Gel'man, N. E.; Terent'eva, E. A.; Shanina, T. M. Methods of Quantitative Organic Elemental Microanalysis [in Russian]; Gel'man, N. E., Ed.; Khimiya: Moscow, 1987.
This work received financial support from the Grants Council of the President of the Russian Federation (grant MK-6738.2016.3).
The work by E. N. Shepelenko and A. D. Dubonosov was supported by the State Contract 007-01114-16 PR 0256-2014-0009.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, 2018, 54(1), 32–37
Rights and permissions
About this article
Cite this article
Tikhomirova, K.S., Podshibyakin, V.A., Shepelenko, E.N. et al. Photo- and ionochromic indolyl(thienyl)maleimides containing naphthalimide linkers. Chem Heterocycl Comp 54, 32–37 (2018). https://doi.org/10.1007/s10593-018-2226-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10593-018-2226-x