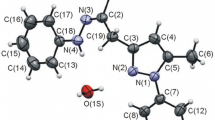
Reactions of 4-aryl-6-trifluoromethyl-2H-pyran-2-ones with sodium hydroxide followed by acidification provided the respective 6-hydroxy-5,6-dihydro derivatives, while the reactions of 4-aryl-6-trifluoromethyl-2H-pyran-2-ones with hydrazine and hydroxylamine were accompanied by rearrangement to N-substituted 6-hydroxy-5,6-dihydro-2-pyridones.


Similar content being viewed by others
References
(a) Hänsel, R.; Pelrer, A.; Schulz, J.; Hille, C. Chem. Ber. 1976, 109, 1617. (b) Hänsel, R.; Schulz, J. Arch. Pharm. 1982, 315, 147.
Nicotra, V. E.; Ramacciotti, N. S.; Gil, R. R.; Oberti, J. C.; Feresin, G. E.; Guerrero, C. A.; Baggio, R. F.; Garland, M. T.; Burton G. J. Nat. Prod. 2006, 69, 783.
(a) Nahrstedt, A.; Kant, J.-D.; Wray, V. Phytochem. 1982, 21, 101. (b) Hungeling, M.; Lechtenberg, M.; Fronczek, F. R.; Nahrstedt, A. Phytochem. 2009, 70, 270.
Ito, K.; Miyajima, S. Bull. Chem. Soc. Jpn. 1986, 59, 815.
(a) Caspi, E.; Schmid, W.; Khan, B. T. Tetrahedron 1962, 18, 767. (b) Kocór, M.; Kurek, A.; Dabrowski, J. Tetrahedron 1969, 25, 4257. (c) Frimer, A. A.; Ripstos, S.; Marks, V.; Aljadeff, G.; Hameiri-Buch J.; Gilinsky-Sharon, P. Tetrahedron 1991, 47, 8361. (d) Frimer, A. A.; GilinskySharon, P.; Aljadeff, G.; Gottlieb, H. E.; Hameiri-Buch, J.; Marks, V.; Philosof, R.; Rosental, Z. J. Org. Chem. 1989, 54, 4853. (e) Frimer, A. A.; Gilinsky-Sharon, P.; Aljadeff, G.; Marks, V.; Rosental, Z. J. Org. Chem. 1989, 54, 4866. (f) Frimer, A. A.; Gilinsky-Sharon, P.; Aljadeff, G. Tetrahedron Lett. 1982, 23, 1301. (g) Srinivasan, R.; Rinehart, K., Jr. J. Org. Chem. 1968, 33, 351. (h) Miyajima, S.; Ito, K. Bull. Chem. Soc. Jpn. 1985, 58, 2659. (i) Padwa, A.; Eisenbarth, P. Tetrahedron 1985, 41, 283. (j) Langer, J.; Fischer, R.; Görls, H.; Walther, D. J. Organomet. Chem. 2004, 689, 2952.
Yamada, K.; Naito, T.; Okuhara, K.; Nakata, K.; Hirata, Y. Bull. Chem. Soc. Jpn. 1960, 33, 1303.
(a) Zonov, Y. V.; Karpov, V. M.; Platonov. V. E. J. Fluorine Chem. 2007, 128, 1065. (b) Zonov, Y. V.; Karpov, V. M.; Platonov, V. E. Russ. J. Org. Chem. 2010, 46, 1517. [Zh. Org. Khim. 2010, 46, 1512.]
(a) Gerus, I. I.; Tolmachova, N. A.; Vdovenko, S. I.; Fröhlich, R.; Haufe, G. Synthesis 2005, 1269. (b) Tolmachova, N. A.; Dolovanyuk, V. G.; Gerus, I. I.; Kondratov, I. S.; Polovinko, V. V.; Bergander, K.; Haufe G. Synthesis 2011, 1149.
Modyanova, L.; Azerad, R. Tetrahedron Lett. 2000, 41, 3865.
Shone, R. L. Tetrahedron Lett. 1979, 24, 2185.
(a) Usachev, B. I.; Obydennov, D. L.; Röschenthaler, G.-V.; Sosnovskikh, V. Y. Org. Lett. 2008, 10, 2857. (b) Usachev, S. A.; Usachev, B. I.; Sosnovskikh, V. Y. Tetrahedron 2014, 70, 60. (с) Usachev, S. A.; Usachev, B. I.; Eltsov, O. S.; Sosnovskikh, V. Y. Tetrahedron 2014, 70, 8863. (d) Usachev, S. A.; Popova, N. V.; Moshkin, V. S.; Sosnovskikh, V. Ya. Chem. Heterocycl. Compd. 2015, 51, 913. [Khim. Geterotsikl. Soedin. 2015, 51, 913.]
Yeh, P.-P.; Daniels, D. S. B.; Cordes, D. B.; Slawin, A. M. Z.; Smith, A. D. Org. Lett. 2014, 16, 964.
Peet, N. P.; Sunder, S. Heterocycles 1986, 24, 393.
(a) Shusherina, N. P.; Dmitrieva, N. D.; Luk'yanets, E. A.; Levina, R. Ya. Russ. Chem. Rev. 1967, 36, 175. [Usp. Khim. 1967, 36, 437.] (b) Phillips, A. J.; Henderson, J. A.; Jackson, K. L. In Comprehensive Heterocyclic Chemistry III; Katritzky, A. R.; Ramsden, C. A.; Scriven, E. F. V.; Taylor, R. J. K., Eds.; Elsevier: Amsterdam, 2008, Vol. 7, p. 366. (с) Goel, A.; Ram, V. J. Tetrahedron 2009, 65, 7865.
Sheldrick, G. M. Acta Crystallogr., Sect. A: Found. Crystallogr. 2008, A64, 112.
This work was performed with financial support from the Russian Foundation for Basic Research (grant No. 1703-00340).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, 2017, 53(12), 1294–1301
Rights and permissions
About this article
Cite this article
Usachev, S.A., Usachev, B.I. & Sosnovskikh, V.Y. Synthesis of 6-hydroxy-5,6-dihydro-2-pyrones and -pyridones by reaction of 4-aryl-6-trifluoromethyl-2-pyrones with water, hydrazine, and hydroxylamine. Chem Heterocycl Comp 53, 1294–1301 (2017). https://doi.org/10.1007/s10593-018-2209-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10593-018-2209-y