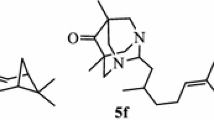
A series of novel heterocyclic derivatives of (–)-borneol has been prepared by the interaction of (1S,2R,4S)-1,7,7-trimethylbicyclo[2.2.1]-heptan-2-yl 2-chloroacetate and (1S,2R,4S)-1,7,7-trimethylbicyclo[2.2.1]heptan-2-yl 3-chloropropanoate with different N- and S-nucleophiles. The obtained products were screened for antiviral, antiulcer, and analgesic activity.

Similar content being viewed by others
References
Corrêa, P. R. C.; Miranda, R. R. S.; Duarte, L. P.; Silva, G. D. F.; Vieira Filho, S. A.; Okuma, A. A.; Carazza, F.; Morgado-Díaz, J. A.; Pinge-Filho, P.; Yamauchi, L. M.; Nakamura, C. V.; Yamada-Ogatta, S. F. Pathog. Global Health 2012, 106, 107.
Bing, S.; Di, L.; Shihui, W.; Lingyun, Z.; Lei, T.; Qipeng, Y.; Xing, W. RSC Adv. 2015, 5, 51947.
Vasconcelos, R. M.; Leite, F. C.; Leite, J. A.; Rodrigues-Mascarenhas, S.; Rodrigues, L. C.; Piuvezam, M. R. Immunopharmacol. Immunotoxicol. 2012, 34, 1028.
Šala, M.; De Palma, A. M.; Hȓebabecký, H.; Nencka, R.; Dračínsky', M.; Leyssen, P.; Neyts, J.; Holý, A. Bioorg. Med. Chem. 2010, 18, 4374.
Chai, G.; Pan, Y.; Li, F. Zhongguo Zhongyao Zazhi 2009, 34(6), 698.
Xu, X.-L.; Li, J.-J.; Han, S.-P.; Tao, C.-H.; Fang, L.; Sun, Y.; Zhu, J.-Z.; Liang, Z.-H.; Li, F.-Z. Eur. J. Pharm. Sci. 2016, 88, 178.
Chen, Y.; Wang, N. Zhongguo Zhongxiyi Jiehe Zazhi 2004, 24(7), 632.
Sokolova, A. S.; Yarovaya, O. I.; Shernyukov, A. V.; Gatilov, Yu. V.; Razumova, Yu. V.; Zarubaev, V. V.; Tretiak, T. S.; Pokrovsky, A. G.; Kiselev, O. I.; Salakhutdinov, N. F. Eur. J. Med. Chem. 2015, 105, 263.
Sokolova, A. S.; Yarovaya, O. I.; Korchagina, D. V.; Zarubaev, V. V.; Tretiak, T. S.; Anfimov, P. M.; Kiselev, O. I.; Salakhutdinov, N. F. Bioorg. Med. Chem. 2014, 22, 2141.
Sokolova, A. S.; Yarovaya, O. I.; Baev, D. S.; Shernyukov, A. V.; Shtro, A. A.; Zarubaev, V. V.; Salakhutdinov, N. F. Eur. J. Med. Chem. 2017, 127, 661.
Sokolova, A.; Pavlova, A.; Komarova, N.; Ardashov, O.; Shernyukov, A.; Gatilov, Yu.; Yarovaya, O.; Tolstikova, T.; Salakhutdinov, N. Med. Chem. Res. 2016, 25, 1608.
(a) Patil, A.; Ganguly, S.; Surana S. Rasayan J. Chem. 2008, 1, 447. (b) Naga Prashant, K.; Ravi Kumar, K. Int. J. PharmTech Res. 2015, 8(9), 60. (c) Azam, M. A.; Suresh, B. Sci. Pharm. 2012, 80, 789. (d) Lokwani, P.; Nagori, B. P.; Batra, N.; Goyal, A.; Gupta, S.; Singh, N. J. Chem. Pharm. Res. 2011, 3(3), 302.
D'Amico, J. J.; Suba, L.; Ruminski, P. G. J. Heterocycl. Chem. 1985, 22, 1479.
Gomtsyan, A. Chem. Heterocycl. Compd. 2012, 48, 7. [Khim. Geterotsikl. Soedin. 2012, 12.]
Pauls, F.; Wick, A. M.; MacKay, E. M. Gastroenterology 1947, 8, 774.
Dentel, H.; Chataigner, I.; Lohier, J.-F.; Gulea, M. Tetrahedron 2012, 68, 2326.
Mosmann, T. J. Immunol. Methods 1983, 65, 55.
Derelanko, M. J.; Long, J. F. Dig. Dis. Sci. 1980, 25(11), 830.
Koster, R.; Anderson, M.; De Beer, E. J. Fed. Proc. 1959, 18, 412.
Eddy, N. B.; Leimbach, D. J. Pharmacol. Exp. Ther. 1953, 107, 385.
This work was supported by the Russian Foundation for Basic Research (grant No. 15-03-00193).
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Khimiya Geterotsiklicheskikh Soedinenii, 2017, 53(3), 371–377
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 2578 kb)
Rights and permissions
About this article
Cite this article
Sokolova, A.S., Yarovaya, O.I., Shtro, A.A. et al. Synthesis and biological activity of heterocyclic borneol derivatives. Chem Heterocycl Comp 53, 371–377 (2017). https://doi.org/10.1007/s10593-017-2063-3
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10593-017-2063-3