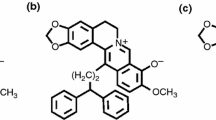
A method was developed for the synthesis of dihydroberberine nitroaryl derivatives on the basis of dihydroberberine reactions with aromatic electrophiles (picryl chloride, 4-chloro-7-nitrobenzofurazan, 4-chloro-5,7-dinitrobenzofurazan, and 7-chloro-4,6-dinitrobenzofuroxan). The obtained 13-substituted dihydroberberine derivatives represent structures with significant intramolecular charge transfer and, according to the results of molecular docking analysis, can effectively bind with G-quadruplexes of telomeric DNA fragments.



Similar content being viewed by others
References
Nechepurenko, I. V.; Salakhutdinov, N. F.; Tolstikov, G. A. Khimiya v Interesakh Ustoichivogo Razvitiya 2010, 18, 1.
Grycová, L.; Dostál, J.; Marek, R. Phytochemistry 2007, 68, 150.
Zhang, W.-J.; Ou, T.-M.; Lu, Y.-J.; Huang, Y.-Y.; Wu, W.-B.; Huang, Z.-S.; Zhou, J.-L.; Wong, K.-Y.; Gu, L.-Q. Bioorg. Med. Chem. 2007, 15, 5493.
Balasubramanian, S.; Hurley, L. H.; Neidle S. Nat. Rev. Drug Discovery 2011, 10, 261.
De Cian, A.; Lacroix, L.; Douarre, C.; Temime-Smaali, N.; Trentesaux, C.; Riou, J. F.; Mergny, J. L. Biochimie 2008, 90, 131.
Cavallari, M.; Garbesi, A.; Di Felice, R. J. Phys. Chem. B 2009, 113, 13152.
Li, Q; Xiang, J.-F.; Yang, Q.-F.; Sun, H.-X.; Guan, A.-J.; Tang, Y.-L. Nucleic Acids Res. 2013, 41, D1115.
Jiménez-Banzo, A.; Sagristà, M. L.; Mora, M.; Nonell, S. Free Radical Biol. Med. 2008, 44, 1926.
Xiong, Y.-X.; Huang, Z.-S.; Tan, J.-H. Eur. J. Med. Chem. 2015, 97, 538.
Bhadra, K.; Kumar, G. S. Biochim. Biophys. Acta 2011, 1810, 485.
Burov, O. N.; Kurbatov, S. V.; Morozov, P. G.; Kletskii, M. E.; Tatarov, A. V. Chem. Heterocycl. Compd. 2015, 51, 772. [Khim. Geterotsikl. Soedin. 2015, 51, 772.]
Lyamzaev, K. G.; Pustovidko, A. V.; Simonyan, R. A.; Rokitskaya, T. I.; Domnina, L. V.; Ivanova O. Y.; Severina, I. I.; Sumbatyan, N. V.; Korshunova, G. A.; Tashlitsky, V. N.; Roginsky, V. A.; Antonenko, Y. N.; Skulachev, M. V.; Chernyak, B. V.; Skulachev, V. P. Pharm. Res. 2011, 28, 2883.
Kurbatov, S.; Tatarov, A.; Minkin, V.; Goumont, R.; Terrier, F. Chem. Commun. 2006, 4279.
Semenyuk, Yu. P.; Kochubei, A. S.; Morozov, P. G.; Burov, O. N.; Kletskii, M. E.; Kurbatov, S. V. Chem. Heterocycl. Compd. 2014, 50, 1731. [Khim. Geterotsikl. Soedin. 2014, 1881.]
Larina, L. I.; Lopyrev, V. A. Nitroazoles: Synthesis, Structure and Applications; Springer: New York, 2009, 446 p.
Burov, O. N.; Kletskii, M. E.; Fedik, N. S; Kurbatov, S. V; Lisovin, A. V. Chem. Heterocycl. Compd. 2015, 51, 997. [Khim. Geterotsikl. Soedin. 2015, 51, 997.]
Tkachuk, A. V.; Samarin, K. A.; Kletskii, M. E.; Burov, O. N.; Golikov, A. Yu.; Kurbatov, S. V.; Minkin, V. I. Rus. J. Org. Chem. 2014, 50, 1223. [Zh. Org. Khim. 2014, 50, 1237.]
Tkachuk, A. V.; Kurbatov, S. V.; Burov, O. N.; Kletskii, M. E.; Morozov, P. G.; Minkin, V. I. Chem. Heterocycl. Compd. 2014, 50, 26. [Khim. Geterotsikl. Soedin. 2014, 32.]
Minkin, V. I.; Tkachuk, A. V.; Kletskii, M. E.; Steglenko, D. V.; Voronina, V. A.; Kurbatov, S. V. Rus. Chem. Bull., Int. Ed. 2013, 62, 464. [Izv. Akad. Nauk, Ser. Khim. 2013, 464.]
Ramsden, C. A. Tetrahedron 2013, 69, 4146.
Parr, R. G.; Szentpály, L.V.; Liu, S. J. Am. Chem. Soc. 1999, 121, 1922.
Woodford, K. J.; Howell, R. M.; Usdin, K. J. Biol. Chem. 1994, 269, 27029.
Qi, H.; Lin, C.-P.; Fu, X.; Wood, L. M.; Liu, A. A.; Tsai, Y.-C.; Chen, Y.; Barbieri, C. M.; Pilch, D. S.; Liu, L. F. Cancer Res. 2006, 66, 11808.
Hurley, L. H. Nat. Rev. Cancer 2002, 2, 188.
Weinstein, I. B.; Joe, A. K. Nat. Clin. Pract. Oncol. 2006 , 3, 448.
Hounsou, C; Guittat, L.; Monchaud, D.; Jourdan, M.; Saettel, N.; Mergny, J.-L.; Teulade-Fichou, M.-P. ChemMedChem 2007, 2, 655.
Burge, S.; Parkinson, G. N.; Hazel, P.; Todd, A. K.; Neidle, S. Nucleic Acids Res. 2006, 34, 5402.
Phan, A. T.; Kuryavyi, V.; Patel, D. J. Curr. Opin. Struct. Biol. 2006, 16, 288.
Davis, J. T. Angew. Chem., Int. Ed. 2004, 43, 668.
Campbell, N. H.; Abd Karim, N. H.; Parkinson, G. N.; Gunaratnam, M.; Petrucci, V.; Todd, A. K.; Vilar, R.; Neidle, S. J. Med. Chem. 2012, 55(1), 209.
Collie, G. W.; Sparapani, S.; Parkinson, G. N.; Neidle, S. J. Am. Chem. Soc. 2011, 133, 2721.
Gomez, D.; Aouali, N.; Londoño-Vallejo, A.; Lacroix, L.; Mégnin-Chanet, F.; Lemarteleur, T.; Douarre, C.; Shin-ya, K.; Mailliet, P.; Trentesaux, C.; Morjani, H.; Mergny, J.-L.; Riou, J.-F. J. Biol. Chem. 2003, 278, 50554.
Burger, A. M.; Dai, F.; Schultes, C. M.; Reszka, A. P.; Moore, M. J.; Double, J. A.; Neidle, S. Cancer Res. 2005, 65, 1489.
Kim, N. W.; Piatyszek, M. A.; Prowse, K. R.; Harley, C. B.; West, M. D.; Ho, P. L.; Coviello, G. M.; Wright, W. E.; Weinrich, S. L.; Shay, J. W. Science 1994, 266, 2011.
Shay, J. W.; Bacchetti, S. Eur. J. Cancer 1997, 33, 787.
Neidle, S. FEBS J. 2010, 277, 1118.
Boyer, R.; Spencer, E. Y.; Wright, G. F. Сan. J. Res. 1946, 24B, 200.
Norris, W. P.; Chafin, A.; Spear, R. J.; Read, R. W. Heterocycles 1984, 22, 271.
Sharnin, G. P.; Levinson, F. S.; Akimova, S. A.; Khasanov, R. Kh. USSR Author's Certificate 627129; Byul. Izobret. 1978, (37).
Becke, A. D. Phys. Rev. A 1988, 38, 3098.
Becke, A. D. J. Chem. Phys. 1993, 98, 5648.
Acknowledgements
This study was supported by a grant from the Russian Science Foundation (project 14-13-00103).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, 2017, 53(3), 335–340
Rights and permissions
About this article
Cite this article
Burov, O.N., Kurbatov, S.V., Kletskii, M.E. et al. Synthesis and structure of dihydroberberine nitroaryl derivatives – potential ligands for G-quadruplexes. Chem Heterocycl Comp 53, 335–340 (2017). https://doi.org/10.1007/s10593-017-2055-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10593-017-2055-3