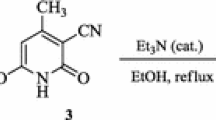
2-Acyl-1,1,3,3-tetracyanopropenides undergo cascade heterocyclization under the action of mercaptoacetic esters, leading to the formation of new 3,6-diamino-4-aroyl-5-cyanothieno[2,3-b]pyridine-2-carboxylates. The fast and convenient synthetic way to highly substituted thieno[2,3-b]pyridine scaffold is described.

Similar content being viewed by others
References
(a) Willemann, C.; Grünert, R.; Bednarski, P. J.; Troschütz, R. Bioorg. Med. Chem. 2009, 17, 4406. (b) Zeng, X.-X.; Zheng, R.-L.; Zhou, T.; He, H.-Y.; Liu, J.-Y.; Zheng, Y.; Tong, A.-P.; Xiang, M.-L.; Song, X.-R.; Yang, S.-Y.; Yu, L.-T.; Wei, Y.-Q.; Zhao, Y.-L.; Yang, L. Bioorg. Med. Chem. Lett. 2010, 20, 6282. (c) Sleebs, B. E.; Levit, A.; Street, I. P.; Falk, H.; Hammonds, T.; Wong, A. C.; Charles, M. D.; Olson, M. F.; Baell, J. B. MedChemComm 2011, 2, 977. (d) Pevet, I.; Brulé, C.; Tizot, A.; Gohier, A.; Cruzalegui, F.; Boutin, J. A.; Goldstein, S. Bioorg. Med. Chem. 2011, 19, 2517. (e) Dai, X.-Y.; Zeng, X.-X.; Peng, F.; Han, Y.-Y.; Lin, H.-J.; Xu, Y.-Z.; Zhou, T.; Xie, G.; Deng, Y.; Mao, Y.-Q.; Yu, L.-T.; Yang, L.; Zhao, Y.-L. Cell. Physiol. Biochem. 2012, 29, 281. (f) Feng, L.; Reynisdóttir, I.; Reynisson, J. Eur. J. Med. Chem. 2012, 54, 463. (g) Romagnoli, R.; Baraldi, P. G.; Salvador, M. K.; Preti, D.; Tabrizi, M. A.; Bassetto, M.; Brancale, A.; Hamel, E.; Castagliuolo, I.; Bortolozzi, R.; Basso, G.; Viola, G. J. Med. Chem. 2013, 56, 2606. (h) Arabshahi, H. J.; Leung, E.; Barker, D.; Reynisson, J. MedChemComm 2014, 5, 186. (i) Hung, J. M.; Arabshahi, H. J., Leung, E.; Reynisson, J.; Barker, D. Eur. J. Med. Chem. 2014, 86, 420. (j) Leung, E.; Hung, J. M.; Barker, D.; Reynisson, J. MedChemComm 2014, 5, 99. (k) Arabshahi, H. J.; van Rensburg, M.; Pilkington, L. I.; Jeon, C. Y.; Song, M.; Gridel, L.-M.; Leung, E.; Barker, D.; Vuica-Ross, M.; Volcho, K. P.; Zakharenko, A. L.; Lavrik, O. I.; Reynisson, J. MedChemComm 2015, 6, 1987.
(a) Tumey, L. N.; Boschelli, D. H.; Lee, J.; Chaudhary, D. Bioorg. Med. Chem. Lett. 2008, 18, 4420. (b) Boschelli, D. H.; Wu, B.; Barrios Sosa, A. C.; Chen, J.; Asselin, M.; Cole, D. C.; Lee, J.; Yang, X.; Chaudhary, D. Bioorg. Med. Chem. Lett. 2008, 18, 2850. (c) Madhusudana, K.; Shireesha, B.; Modi Naidu, V. G.; Ramakrishna, S.; Narsaiah, B.; Rao, A. R.; Diwan, P. V. Eur. J. Pharmacol. 2012, 678, 48. (d) Liu, H.; Li, Y.; Wang, X.-Y.; Wang, B.; He, H.-Y.; Liu, J.-Y.; Xiang, M.-L.; He, J.; Wu, X.-H.; Yang, L. Bioorg. Med. Chem. Lett. 2013, 23, 2349.
(a) Schnute, M. E.; Anderson, D. J.; Brideau, R. J.; Ciske, F. L.; Collier, S. A.; Cudahy, M. M.; Eggen, M.; Genin, M. J.; Hopkins, T. A.; Judge, T. M.; Kim, E. J.; Knechtel, M. L.; Nair, S. K.; Nieman, J. A.; Oien, N. L.; Scott, A.; Tanis, S. P.; Vaillancourt, V. A.; Wathen, M. W.; Wieber, J. L. Bioorg. Med. Chem. Lett. 2007, 17, 3349. (b) Wang, N.-Y.; Zuo, W.-Q.; Xu, Y.; Gao, C.; Zeng, X.-X.; Zhang, L.-D.; You, X.-Y.; Peng, C.-T.; Shen, Y.; Yang, S.-Y.; Wei, Y.-Q.; Yu, L.-T. Bioorg. Med. Chem. Lett. 2014, 24, 1581.
(a) Rolim Bernardino, A. M.; da Silva Pinheiro, L. C.; Rangel Rodrigues, C..; Loureiro, N. I.; Castro, H. C.; Lanfredi-Rangel, A.; Sabatini-Lopes, J.; Borges, J. C.; Carvalho, J. M.; Alves Romeiro, G.; Ferreira, V. F.; Frugulhetti, I. C. P. P.; Vannier-Santos, M. A. Bioorg. Med. Chem. 2006, 14, 5765. (b) Al-Trawneh, S. A.; El-Abadelah, M. M.; Zahra, J. A.; Al-Taweel, S. A.; Zani, F.; Incerti, M.; Cavazzoni, A.; Vicini, P. Bioorg. Med. Chem. 2011, 19, 2541.
(a) Bahekar, R. H.; Jain, M. R.; Goel, A.; Patel, D. N.; Prajapati, V. M.; Gupta, A. A.; Jadav, P. A.; Patel, P. R. Bioorg. Med. Chem. 2007, 15, 3248. (b) Bahekar, R. H.; Jain, M. R.; Jadav, P. A.; Prajapati, V. M.; Patel, D. N.; Gupta, A. A.; Sharma, A.; Tom, R.; Bandyopadhya, D.; Modi, H.; Patel, P. R. Bioorg. Med. Chem. 2007, 15, 6782.
(a) Ohba, S.; Nakajima, K.; Komiyama, Y.; Kugimiya, F.; Igawa, K.; Itaka, K.; Moro, T.; Nakamura, K.; Kawaguchi, H.; Takato, T.; Chung, U. Biochem. Biophys. Res. Commun. 2007, 357, 854. (b) Saito, K.; Nakao, A.; Shinozuka, T.; Shimada, K.; Matsui, S.; Oizumi, K.; Yano, K.; Ohata, K.; Nakai, D.; Nagai, Y.; Naito, S. Bioorg. Med. Chem. 2013, 21, 1628.
(a) Le, U.; Melancon, B. J.; Bridges, T. M.; Vinson, P. N.; Utley, T. J.; Lamsal, A.; Rodriguez, A. L.; Venable, D.; Sheffler, D. J.; Jones, C. K.; Blobaum, A. L.; Wood, M. R.; Daniels, J. S.; Conn, P. J.; Niswender, C. M.; Lindsley, C. W.; Hopkins, C. R. Bioorg. Med. Chem. Lett. 2013, 23, 346. (b) Szabo, M.; Herenbrink, C. K.; Christopoulos, A.; Lane, J. R.; Capuano, B. J. Med. Chem. 2014, 57, 4924. (c) Szabo, M.; Huynh, T.; Valant, C.; Lane, J. R.; Sexton, P. M.; Christopoulos, A.; Capuano, B. MedChemComm 2015, 6, 1998.
Mohler, E. G.; Shacham, S.; Noiman, S.; Lezoualc'h, F.; Robert, S.; Gastineau, M.; Rutkowski, J.; Marantz, Y.; Dumuis, A.; Bockaert, J.; Gold, P. E.; Ragozzino, M. E. Neuropharmacol. 2007, 53, 563.
(a) Leonczak, P.; Gao, L.-J.; Ramadori, A. T.; Lescrinier, E.; Rozenski, J.; De Jonghe, S.; Herdewijn, P. ChemMedChem. 2014, 9, 2587. (b) Gao, L.-J.; Kovackova, S.; Šála, M.; Ramadori, A. T.; De Jonghe, S.; Herdewijn, P. J. Med. Chem. 2014, 57, 7624.
Krauze, A.; Grinberga, S.; Krasnova, L.; Adlere, I.; Sokolova, E.; Domracheva, I.; Shestakova, I.; Andzans, Z.; Duburs, G. Bioorg. Med. Chem. 2014, 22, 5860.
Nógrádi, K.; Wágner, G.; Domány, G.; Bobok, A.; Magdó, I.; Kolok, S.; Mikó-Bakk, M.; Vastag, M.; Sághy, K.; Gyertyán, I.; Kóti, J.; Gál, K.; Farkas, S.; Keserű, G. M.; Greiner, I.; Szombathelyi, Z. Bioorg. Med. Chem. Lett. 2015, 25, 1724.
Bondarenko, S. P.; Zhitnetskyi, I. V.; Semenov, S. V.; Frasinyuk, M. S. Chem. Heterocycl. Compd. 2016, 52, 262. [Khim. Geterotsikl. Soedin. 2016, 262.]
Karpov, S. V.; Grigor'ev, A. A.; Kayukov, Ya. S.; Karpova, I. V.; Nasakin, O. E.; Tafeenko, V. A. J. Org. Chem. 2016, 81, 6402.
Kayukov, Ya. S.; Bardasov, I. N.; Karpov, S. V.; Ershov, O. V.; Nasakin, O. E.; Kayukova, O. V.; Tafeenko, V. A. Russ. J. Org. Chem. 2012, 48, 1447. [Zh. Org. Khim. 2012, 48, 1463.]
Grigor'ev, A. A.; Karpov, S. V.; Kayukov, Ya. S.; Belikov, M. Yu.; Nasakin, O. E. Tetrahedron Lett. 2015, 56, 6279.
Karpov, S. V.; Kayukov, Ya. S.; Bardasov, I. N.; Ershov, O. V.; Nasakin, O. E.; Kayukova, O. V. Russ. J. Org. Chem. 2011, 47, 1161. [Zh. Org. Khim. 2011, 47, 1144.]
(a) Karpov, S. V.; Kayukov, Ya. S.; Bardasov, I. N.; Kayukova, O. V.; Lipin, K. V.; Nasakin, O. E. Russ. J. Org. Chem. 2011, 47, 1492. [Zh. Org. Khim. 2011, 47, 1467.] (b) Kayukov, Ya. S.; Karpov, S. V.; Kayukova, O. V.; Ershov, O. V.; Nasakin, O. E. Russ. J. Org. Chem. 2014, 50, 1097. [Zh. Org. Khim. 2014, 50, 1116.]
Sheldrick, G. M. Acta Crystallogr., Sect. A: Found. Crystallogr. 2008, A64, 112.
Putz, H.; Brandenburg, K. Diamond – Crystal and Molecular Structure Visualization; Release 2.1d; Crystal Impact –GbR: Bonn, 2000. http://www.crystalimpact.com/diamond
This work was supported by the Russian Foundation for Basic Research (research project No. 15-33-21087 'mol_a_ved'). The X-ray study was supported in part by Lomonosov Moscow State University Program of Development.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Khimiya Geterotsiklicheskikh Soedinenii, 2017, 53(2), 230–235
Rights and permissions
About this article
Cite this article
Grigor’ev, A.A., Karpov, S.V., Kayukov, Y.S. et al. A new route to highly substituted thieno[2,3-b]pyridines via cascade heterocyclization of 2-acyl-1,1,3,3-tetracyanopropenide salts. Chem Heterocycl Comp 53, 230–235 (2017). https://doi.org/10.1007/s10593-017-2044-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10593-017-2044-6