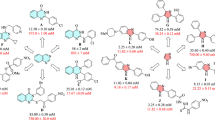
A new series of 2-substituted quinazolin-4(3H)-one derivatives including coumarin nucleus has been synthesized and screened for their lipase and α-glucosidase inhibition properties. Among the synthesized compounds, N'-{2-[2-(3,4-dichlorobenzyl)-4-oxoquinazolin-3(4H)-yl]acetyl}-2-oxo-2H-chromene-3-carbohydrazide and N'-{2-[2-(4-bromobenzyl)-4-oxoquinazolin-3(4H)-yl]acetyl}-2-oxo-2H-chromene-3-carbohydrazide showed the best inhibitory effect against α-glucosidase with IC50 values of 6.11 ± 0.40 and 7.34 ± 0.37 μM,respectively. These compounds also showed strong anti-lipase activity (IC50 3.52 ± 0.49 and 2.85 ± 0.27 μM, respectively).
Similar content being viewed by others
References
Kumakura, S.; Onoda, K.; Hirose, M. J. Infect. Chemother. 2014, 20, 194.
Hirose, M.; Ando, T.; Shofiqur, R.; Umeda, K.; Kodama, Y.; Nguyen, S. V.; Goto, T.; Shimada, M.; Nagaoka, S. Nutr. Metab. 2013, 10, 70.
Mukherjee, M. J. Mol. Catal. B: Enzym. 2003, 22, 369.
Birari, R. B.; Bhutani, K. K. Drug Discovery Today 2007, 12, 879.
Ballinger, A.; Peikin, S. R. Eur. J. Pharmacol. 2002, 440, 109.
Filippatos, T. D.; Derdemezis, C. S.; Gazi, I. F.; Nakou, E. S.; Mikhailidis, D. P.; Elisaf, M. S. Drug Saf. 2008, 31, 53.
Wang, Q. Q.; Cheng, N.; Yi, W. B.; Peng, S. M.; Zou, X. Q. Bioorg. Med. Chem. 2014, 22, 1515.
Casirola, D. M.; Ferraris, R. P. Metabolism 2006, 55, 832.
Panahi, F.; Yousefi, R.; Mehraban, M. H.; Khalafi-Nezhad, A. Carbohydr. Res. 2013, 380, 81.
King, H.; Rewers, M. Diabetes Care 1993, 16, 157.
Alapati, V.; Noolvi, M. N.; Manjula, S. N.; Pallavi, K. J.; Patel, H. M.; Tippeswamy, B. S.; Satyanarayana, S. V. Eur. Rev. Med. Pharmacol. Sci. 2012, 16, 1753.
Widemann, B. C.; Balis, F. M.; Godwin, K. S.; McCully, C.; Adamson, P. C. Cancer Chemother. Pharmacol. 1999, 44, 439.
Kobayashi, Y.; Nakano, Y.; Kizaki, M.; Hoshikuma, K.; Yokoo, Y.; Kamiya, T. Planta Med. 2001, 67, 628.
Clissold, S. P.; Beresford, R.; Proquazone, A. Drugs 1987, 33, 478.
Khadka, D. B.; Tran, G. H.; Shin, S.; Nguyen, H. T.; Cao, H. T.; Zhao, C.; Jin, Y.; Van, H. T.; Chau, M. V.; Kwon, Y.; Le, T. N.; Cho, W. J. Eur. J. Med. Chem. 2015, 103, 69.
Rakesh, K. P.; Manukumar, H. M.; Gowda, D. C. Bioorg. Med. Chem. Lett. 2015, 25, 1072.
Kayser, O.; Kolodziej, H. Planta Med. 1997, 63, 508.
Hoult, J. R.; Paya, M. Gen. Pharmacol. 1996, 27, 713.
Zaha, A. A.; Hazem, A. New Microbiol. 2002, 25, 213.
Kontogiorgis, C.; Hadjipavlou-Litina, D. J. Enzyme Inhib. Med. Chem. 2003, 18, 63.
Khode, S.; Maddi, V.; Aragade, P.; Palkar, M.; Ronad, P. K.; Mamledesai, S.; Thippeswamy, A. H. M.; Satyanarayana, D. Eur. J. Med. Chem. 2009, 44, 1682.
Kontogiorgis, C. A.; Hadjipavlou-Litina, D. J. J. Med. Chem. 2005, 48, 6400.
Wang, C. J.; Hsieh, Y. J.; Chu, C. Y.; Lin, Y. L.; Tseng, T. H. Cancer Lett. 2002, 183, 163.
Kostova, I. Mini-Rev. Med. Chem. 2006, 6, 365.
Chimichi, S.; Boccalini, M.; Cosimelli, B.; Viola, G.; Vedaldi, D.; Dall'Acqua, F. Tetrahedron Lett. 2002, 43, 7473.
Gouda, M. A.; Berghot, M. A.; Baz, E. A.; Hamama, W. S. Med. Chem. Res. 2012, 21, 1062.
Khan, K. M.; Rahim, F.; Wadood, A.; Kosar, N.; Taha, M.; Lalani, S.; Khan, A.; Fakhri, M. I.; Junaid, M.; Rehman, W.; Khan, M.; Perveen, S.; Sajid, M.; Choudhary, M. I. Eur. J. Med. Chem. 2014, 81, 245.
Wang, S. J.; Yan, J. F.; Wang, X. Y.; Yang, Z.; Lin, F. W.; Zhang, T. J. Eur. J. Med. Chem. 2010, 45, 1250.
Islam, M. N.; Jung, H. A.; Sohn, H. S.; Kim, H. M.; Choi, J. S. Arch. Pharm. Res. 2013, 36, 542.
Shen, Q.; Shao, J.; Peng, Q.; Zhang, W.; Ma, L.; Chan, A. S. C.; Gu, L. J. Med. Chem. 2010, 53, 8252.
Kahveci, B.; Yılmaz, F.; Menteşe, E.; Ülker, S. Chem. Heterocycl. Compd. 2015, 51, 447. [Khim. Geterotsikl. Soedin. 2015, 51, 447.]
Kahveci, B. Molecules 2005, 10, 376.
Kahveci, B.; Ikizler, A. A. Turk. J. Chem. 2000, 24, 343.
Thati, B.; Noble, A.; Rowan, R.; Creaven, B. S.; Walsh, M.; McCann, M.; Egan, D.; Kavanagh, K. Toxicol. In Vitro 2007, 21, 801.
Katritzky, A. R.; Narindoshvili, T.; Angrish, P. Synthesis 2008, 13, 2013.
Chandrika, P. M.; Yakaiah, T.; Rao, A. R. R.; Narsaiah, B.; Reddy, N. C.; Sridhar, V.; Rao, J. V. Eur. J. Med. Chem. 2008, 43, 846.
Grover, G.; Kini, S. G. Eur. J. Med. Chem. 2006, 41, 256.
Casagrande, M.; Barteselli, A.; Basilico, N.; Parapini, S.; Taramelli, D.; Sparatore, A. Bioorg. Med. Chem. 2012, 20, 5965.
Kashaw, S. K.; Kashaw, V.; Mishra, P.; Jain, N. K.; Stables, J. P. Eur. J. Med. Chem. 2009, 44, 4335.
Decker, M. Eur. J. Med. Chem. 2005, 40, 305.
Shen, Y.; Han, C.; Cai, S. Y.; Lu, P.; Wang, Y. G. Tetrahedron Lett. 2012, 53, 5671.
Choudhary, M. I.; Adhikari, A.; Rasheed, S.; Marasini, B. P.; Hussain, N.; Kaleem, W. A. Phytochem. Lett. 2011, 4, 404.
Kurihara, H.; Asami, S.; Shibata, H.; Fukami, H.; Tanaka, T. Biol. Pharm. Bull. 2003, 26, 383.
This work was supported by the Scientific and Technological Research Council of Turkey (TUBITAK) under the project number of 115Z885. The authors gratefully thank TUBITAK for financial support.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Khimiya Geterotsiklicheskikh Soedinenii, 2016, 52(12), 1017–1024
Rights and permissions
About this article
Cite this article
Menteşe, E., Karaali, N., Akyüz, G. et al. Synthesis and Evaluation of α-Glucosidase and Pancreatic Lipase Inhibition by Quinazolinone-Coumarin Hybrids. Chem Heterocycl Comp 52, 1017–1024 (2016). https://doi.org/10.1007/s10593-017-2002-3
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10593-017-2002-3