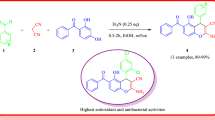
A series of alkyl 2-amino-4-aryl-4H-chromene-3-carboxylates was synthesized by an efficient, solvent-free one-pot three-component cyclocondensation of resorcinol, aromatic aldehydes, and ethyl or methyl cyanoacetate using sodium carbonate as catalyst. The present methodology offers several advantages, such as a simple procedure with ease of handling, short reaction time, high yields, and the absence of any volatile and hazardous organic solvents. The products were characterized on the basis of IR, 1H NMR, and 13C NMR spectral and microanalytical data and evaluated for their antibacterial activity against Gram-positive bacteria (Staphylococcus epidermidis and Staphylococcus aureus) and Gram-negative bacteria (Escherichia coli) using paper disc diffusion technique.

Similar content being viewed by others
References
Raj, T.; Bhatia, R. K.; Sharma, M.; Saxena, A.; Ishar, M. Eur. J. Med. Chem. 2010, 45, 790.
Conti, C.; Monaco, L. P.; Desideri, N. Bioorg. Med. Chem. 2014, 22, 1201.
Gourdeau, H.; Leblond, L.; Hamelin, B.; Desputeau, C.; Dong, K.; Kianicka, I.; Custeau, D.; Boudreau, C.; Geerts, L.; Cai, S.-X.; Drewe, J.; Labrecque, D.; Kasibhatla, S.; Tseng, B. Mol. Can. Ther. 2004, 3, 1375.
Ashok, D.; Lakshmi1, B. V.; Ravi, S.; Ganesh, A.; Adam, S. Chem. Heterocycl. Compd. 2015, 51, 462. [Khim Geterotsikl. Soedin. 2015, 462.]
Johannes, C. W.; Visser, M. S.; Weatherhead, G. S.; Hoveyda, A. H. J. Am. Chem. Soc. 1998, 120, 8340.
Bhat, M. A.; Siddiqui, N.; Khan, S. A. Acta Pol. Pharm. 2008, 65, 235.
Cheng, J.-F.; Ishikawa, A.; Ono, Y.; Arrhenius, T.; Nadzan, A. Bioorg. Med. Chem. Lett. 2003, 13, 3647.
Ellis, G. In The Chemistry of Heterocyclic of Compounds. Chromenes, Chromanes and Chromones; Weissberger, A., Taylor, EC, Eds.; John Wiley: New York, 1977., Chapter II, p. 11.
Hafez, E. A. A.; Elnagdi, M. H.; Elagamey, A. G. A.; El-Taweel, F. M. A. A. Heterocycles 1987, 26, 903.
Joulain, D.; Tabacchi, R. Phytochemistry 1994, 37, 1769.
Nolan, K. A.; Zhao, H.; Faulder, P. F.; Frenkel, A. D.; Timson, D. J.; Siegel, D.; Ross, D.; Burke Jr, T. R.; Stratford, I. J.; Bryce, R. A. J. Med. Chem. 2007, 50, 6316.
Doshi, J. M.; Tian, D.; Xing, C. J. Med. Chem. 2006, 49, 7731.
Kemnitzer, W.; Drewe, J.; Jiang, S.; Zhang, H.; Crogan-Grundy, C.; Labreque, D.; Bubenick, M.; Attardo, G.; Denis, R.; Lamothe, S.; Gourdeau, H.; Tseng, B.; Kasibhatla, S.; Cai, S. X. J. Med. Chem. 2008, 51, 417.
Zhang, G.; Zhang, Y.; Yan, J.; Chen, R.; Wang, S.; Ma, Y.; Wang, R. J. Org. Chem. 2012, 77, 878.
Kidwai, M.; Saxena, S.; Rahman Khan, M. K.; Thukral, S. S. Bioorg. Med. Chem. Lett. 2005, 15, 4295.
Shah, N. K.; Shah, N. M.; Patel, M. P.; Patel, R. G. J. Chem. Sci. 2013, 125, 525.
He, Y.; Hu, R.; Tong, R.; Li, F.; Shi, J.; Zhang, M. Molecules 2014, 19, 19253.
Zonouzi, A.; Mirzazadeh, R.; Safavi, M.; Ardestani, S. K.; Emami, S.; Foroumadi, A. Iranian J. Pharm. Res. 2013, 12, 679.
Mohammed, F. K.; Soliman, A. Y.; Ssawy, A.; Badre, M. G. J. Chem. Pharm. Res. 2009, 1, 213.
Kolla, S. R.; Lee, Y. R. Tetrahedron 2011, 67, 8271.
Anderson, D. R.; Hegde, S.; Reinhard, E.; Gomez, L.; Vernier, W. F.; Lee, L.; Liu, S.; Sambandam, A.; Snider, P. A.; Masih, L. Bioorg. Med. Chem. Lett. 2005, 15, 1587.
Caruana, L.; Mondatori, M.; Corti, V.; Morales, S.; Mazzanti, A.; Fochi, M.; Bernardi, L. Chem.–Eur. J. 2015, 21, 6037.
Fujiwara, M.; Sakamoto, M.; Komeyama, K.; Yoshida, H.; Takaki, K. J. Heterocycl. Chem. 2015, 52, 59.
Khaksar, S.; Rouhollahpour, A.; Talesh, S. M. J. Fluorine Chem. 2012, 141, 11.
Habibi-Khorassani, S. M.; Hazeri, N.; Shahraki, M.; Abbasi, M.; Karima, M.; Ali, M. Iran. J. Org. Chem. 2013, 5, 1163.
Dekamin, M. G.; Eslami, M.; Maleki, A. Tetrahedron 2013, 69, 1074.
Qareaghaj, O. H.; Mashkouri, S.; Naimi-Jamal, M. R.; Kaupp, G. RSC Adv. 2014, 4, 48191.
Safari, J.; Zarnegar, Z.; Heydarian, M. Bull. Chem. Soc. Jpn. 2012, 85, 1332.
Makarem, S.; Mohammadi, A. A.; Fakhari, A. R. Tetrahedron Lett. 2008, 49, 7194.
Kundu, S. K.; Mondal, J.; Bhaumik, A. Dalton Trans. 2013, 42, 10515.
Shekhar, A. C.; Kumar, A. R.; Sathaiah, G.; Raju, K.; Rao, P. S.; Sridhar, M.; Narsaiah, B.; Srinivas, P. V. S. S.; Sridhar, B. Helv. Chim. Acta 2012, 95, 502.
Al-Mousawi, S. M.; Elkholy, Y. M.; Mohammad, M. A.; Elnagdi, M. H. Org. Prep. Proced. Int. 1999, 31, 305.
Radwan, S. M.; Bakhite, E. A.; Kamal El-Dean, A. M. Phosphorus, Sulfur Silicon Relat. Elem. 1995, 101, 207.
Davoodnia, A.; Bakavoli, M.; Vahedinia, A.; Rahimizadeh, M.; Roshani, M. Heterocycles 2006, 68, 801.
Davoodnia, A.; Bakavoli, M.; Soleimany, M.; Tavakoli- Hoseini, N. Monatsh. Chem. 2009, 140, 355.
Davoodnia, A.; Bakavoli, M.; Moloudi, R.; Khashi, M.; Tavakoli-Hoseini, N. Chin. Chem. Lett. 2010, 21, 1–4.
Davoodnia, A.; Khashi, M.; Tavakoli-Hociseini, N.; Moloudi, R.; Zamani, H. A. Monatsh. Chem. 2013, 144, 677.
Khashi, M.; Davoodnia, A.; Chamani, J. Phosphorus, Sulfur Silicon Relat. Elem. 2014, 189, 839.
Bauer, A. W.; Kirby, W. M. M.; Sherris, J. C.; Turck, M. Am. J. Clin. Pathol. 1966, 45, 493.
The authors express their gratitude to the Islamic Azad University, Mashhad Branch for its financial support.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Khimiya Geterotsiklicheskikh Soedinenii, 2015, 51(9), 808–813
Rights and permissions
About this article
Cite this article
Gholipour, S., Davoodnia, A. & Nakhaei-Moghaddam, M. Synthesis, characterization, and antibacterial evaluation of new alkyl 2-amino-4-aryl-4H-chromene-3-carboxylates. Chem Heterocycl Comp 51, 808–813 (2015). https://doi.org/10.1007/s10593-015-1779-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10593-015-1779-1