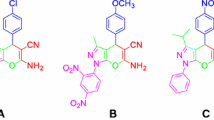
An environmentally friendly method was applied to prepare anthranilic hydrazones and quinazolin-4(1H)-ones from anthranilic hydrazides and a number of aldehydes in solventless medium. The condensations proceeded smoothly under ball milling conditions at room temperature, and the products were obtained in high purity and yield.
Similar content being viewed by others
References
A. V Milyutin, N. V. Safonova, V. P. Chesnokov, F. Y. Nazmedtinov, E. V. Voronina, I. V. Krylova, Yu. S. Andreichikov, V. E. Kolla, and Yu. V. Kozhevnikov, Pharm. Chem. J., 30, 310 (1996). [Khim.-Farm. Zh., 30, № 5, 26 (1996).]
G. Verma, A. Marella, M. Shaquiquzzaman, M. Akhtar, M. R. Ali, and M. M. Alam, J. Pharm. BioAllied Sci., 6, 69 (2014).
N. T. Patil, P. G. V. V. Lakshmi, B. Sridhar, S. Patra, M. P. Bhadra, and C. R. Patra, Eur. J. Org. Chem., 1790 (2012).
D.-S. Chen, G.-L. Dou, Y.-L. Li, Y. Liu, and X.-S. Wang, J. Org. Chem., 78, 5700 (2013).
K. S. Kumar, P. M. Kumar, V. S. Rao, A. A. Jafar, C. L. T. Meda, R. Kapavarapu, K. V. L. Parsa, and M. Pal, Org. Biomol. Chem., 10, 3098 (2012).
X.-S. Wang, J. Sheng, L. Lu, K. Yang, and Y.-L. Li, ACS Comb. Sci., 13, 196 (2011).
A. A. Mohammadi, H. Rohi, and A. A. Soorki, J. Heterocycl. Chem., 50, 1129 (2013).
Q.-S. Ding, J.-L. Zhang, J.-X. Chen, M.-C. Liu, J.-C. Ding, and H.-Y. Wu, J. Heterocycl. Chem., 49, 375 (2012).
F. Miklós and F. Fülöp, Eur. J. Org. Chem., 959 (2010).
M. Ferguson, N. Giri, X. Huang, D. Apperley, and S. T. James, Green Chem., 16, 1374 (2014).
M. Gopalakrishnan, J. Thanusu, and V. Kanagarajan, J. Korean Chem. Soc., 52, 523 (2008).
F. Miklós, V. Hum, and F. Fülöp, ARKIVOC, vi, 25 (2014).
A. Stolle, T. Szuppa, S. E. S. Leonhard, and B. Ondruschka, Chem. Soc. Rev., 40, 2317 (2011).
K. Neuvonen, F. Fülöp, H. Neuvonen, M. Simeonov, and K. Pihlaja, J. Phys. Org. Chem., 10, 55 (1997).
K. Neuvonen, F. Fülöp, H. Neuvonen, A. Koch, and E. Kleinpeter, J. Phys. Org. Chem., 21, 173 (2008).
A. Yu. Ershov, N. A. Lovushkina, I. V. Lagoda, S. I. Yakimovich, I. V. Zerova, V. V. Pakal’nis, and V. V. Shamanin, Chem. Heterocycl. Compd., 45, 965 (2009). [Khim. Geterotsikl. Soedin., 1214 (2009).]
N. P. Peet and E. W. Huber, Heterocycles, 35, 315 (1993).
D. Tan, V. Štrukil, C. Mottillo, and T. Friščić, Chem. Commun., 50, 5248 (2014).
F. Fülöp, M. Simeonov, and K. Pihlaja, Tetrahedron, 48, 531 (1992).
G. Bonola and E. Sianesi, J. Med. Chem., 13, 329 (1970).
P. P. Reddy, C. K. Reddy, and P. S. Reddy, Bull. Chem. Soc. Jpn., 59, 1575 (1986).
K.-C. Liu and M.-K. Hu, Arch. Pharm. (Weinheim, Ger.), 320, 166 (1987).
We are grateful to the Hungarian Research Foundation (OTKA NK81371) and TÁMOP-4.2.2/A-11/1/KONV-2012-0035 for financial support.
Author information
Authors and Affiliations
Corresponding author
Additional information
*Dedicated to Professor Gunars Duburs on the occasion of his 80th birthday.
Published in Khimiya Geterotsiklicheskikh Soedinenii, No. 10, pp. 1590-1595, October, 2014.
Rights and permissions
About this article
Cite this article
Magyar, T., Miklós, F., Lázár, L. et al. Application of a Ball Milling Technique for the Condensation of Anthranilic Hydrazides with Aromatic Aldehydes Towards 4-Quinazolinone Derivatives*. Chem Heterocycl Comp 50, 1464–1470 (2015). https://doi.org/10.1007/s10593-014-1611-3
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10593-014-1611-3