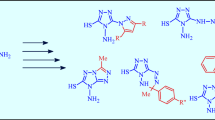
N-Alkyl(aryl)-3-amino-5H,6H,7H-pyrrolo[1,2-a]imidazole-2-carboxamides were obtained by the interaction of 5-alkyl(aryl)amino-2-(3-phthalimidopropyl)-1,3-oxazole-4-carbonitriles with hydrazine hydrate and have been used for the synthesis of substituted 3H,4H,6H,7H,8H-pyrrolo[2,1-h]purin-4-ones, their thione analogs, and also 1,2,3,6,7,8-hexahydro-4H-pyrrolo[2′,1′:2,3]imidazo[4,5-d]-[1,3,2]diazaphosphinine derivatives.
Similar content being viewed by others
References
S. A. Chumachenko, O. V. Shablykin, A. N. Vasilenko, and V. S. Brovarets, Khim. Geterotsikl. Soedin., 1238 (2011). [Chem. Heterocycl. Compd., 47, 1020 (2011).]
J. M. Fevig, J. Cacciola, J. Buriak, K. A. Rossi, R. M. Knabb, J. M. Luettgen, P. C. Wong, S. A. Bai, R. R. Wexler, and P. Y. S. Lam, Bioorg. Med. Chem. Lett., 16, 3755 (2006).
D. B. Nilov, A. V. Kadushkin, N. P. Solov'eva, Yu. N. Sheinker, and V. G. Granik, Khim. Geterotsikl. Soedin., 113 (2004). [Chem. Heterocycl. Compd., 40, 106 (2004).]
B. S. Drach, E. P. Sviridov, A. A. Kisilenko, and A. V. Kirsanov, Zh. Org. Khim., 9, 1818 (1973).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 12, pp. 1956–1962, December, 2012.
Rights and permissions
About this article
Cite this article
Chumachenko, S.A., Shablykin, O.V. & Brovarets, V.S. Application of the Recyclization Products of 5-Alkyl(aryl)amino-2-(3-phthalimidopropyl)-1,3-oxazole-4-carbonitriles to the Synthesis of Condensed Tricyclic Nitrogenous Structures. Chem Heterocycl Comp 48, 1832–1838 (2013). https://doi.org/10.1007/s10593-013-1216-2
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10593-013-1216-2
Keywords
- N-alkyl(aryl)-3-amino-5H,6H,7H-pyrrolo[1,2-a]imidazole-2-carboxamides
- 3-alkyl-2-mercapto-1,2,3,6,7,8-hexahydro-4H-pyrrolo[2′,1′:2,3]imidazo[4,5-d][1,3,2]diazaphosphinine-4-thione 2-sulfides
- 3-alkyl(aryl)-3H,4H,6H,7H,8H-pyrrolo[2,1-h]purin-4-ones
- 3-aryl-3H,4H,6H,7H,8H-pyrrolo-[2,1-h]purine-4-thiones
- recyclization.