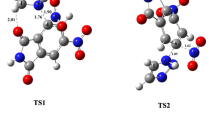
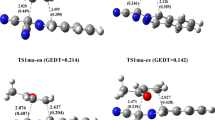
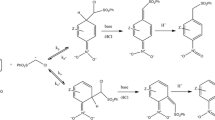
Existing experimental data on positional selectivity in electrophilic substitution reactions of π-excessive heterocycles are classified. These data are discussed basing on the results of the authors' quantum-chemical calculations [RHF/6-31G(d), MP2/6-31G(d), and B3LYP/6-31G(d)] of the σ-complexes formed during attack of electrophiles such as H+, Me+, Me3Si+, Br+, NO2 +, MeCO+, and SO3 at the α- and β-positions of furan, thiophene, selenophene, pyrrole and its N-substituted derivatives, N-R-pyrroles (R = Me, t-Bu, SiMe3, Si(i-Pr)3, C6H4(p-NO2), SO2Ph, CHO, CO2Me), and the corresponding α- and β-substituted electrophilic substitution products. The differences in energies of the α-and β-isomers of the σ-complexes characterize the preferred direction of electrophilic attack, while the differences in the energies of the isomeric products make it possible to assess the energy preference of one of them. Analysis of the obtained data demonstrates the effects of the studied heterocycles' structure, the nature of the electrophile, and the thermal and steric factors on the positional selectivity (α/β ratio) in electrophilic substitution reactions of π-excessive five-membered heteroaromatic compounds.















Similar content being viewed by others
References
G. Marino, Adv. Heterocycl. Chem., 13, 235 (1971).
G. Marino, Khim. Geterotsikl. Soedin., 579 (1973). [Chem. Heterocycl. Compd., 9, 537 (1973)].
L. I. Belen'kii, in III International Symposium on Furan Chemistry, Collection of Papers, Smolenice, Czechoslovakia, 1979, p. 4.
L. I. Belen'kii, Khim. Geterotsikl. Soedin., 1587 (1980). [Chem. Heterocycl. Compd., 16, 1195 (1980)].
P. Linda and G. Marino, J. Chem. Soc., 392 (1968).
Z. N. Nazarova, Zh. Obshch. Khim., 24, 575 (1954).
Ya. L. Gol'dfarb, Yu. B. Vol'kenshtein, and B. V. Lopatin, Zh. Obshch. Khim., 34, 969 (1964).
H. J. Anderson and S.-F. Lee, Can. J. Chem., 43, 409 (1965).
H. J. Anderson and L. C. Hopkins, Can. J. Chem., 42, 1279 (1964).
H. J. Anderson and L. C. Hopkins, Can. J. Chem., 44, 1831 (1966).
H. J. Anderson and C. W. Huang, Can. J. Chem., 45, 897 (1967).
C. E. Loader and H. J. Anderson, Tetrahedron, 25, 3879 (1969).
J. K. Groves, H. J. Anderson, and H. Nagy, Can. J. Chem., 49, 2427 (1971).
C. Jaureguiberry, M. C. Fournier-Zaluski, J. P. Chevallier, and B. Roques, C. R. Acad. Sci., Ser. C, 273, 276 (1971).
P. Fournari, M. Farnier, and C. Fournier, Bull. Soc. Chim. Fr., 283 (1972).
H. J. Anderson, C. R. Riche, T. G. Costello, C. E. Loader, and G. H. Barnett, Can. J. Chem., 56, 654 (1978).
P. Barker, P. Gendler and H. Rapoport, J. Org. Chem., 43, 4849 (1978).
Ya. L. Gol'dfarb and Yu. B. Vol'kenshtein, Dokl. Akad. Nauk USSR, 128, 536 (1959).
S. G. Mairanovskii, N. V. Barashkova, and Yu. B. Vol'kenshtein, Izv. Akad. Nauk USSR, Ser. Khim., 1539 (1965).
L. I. Belen'kii, Ya. L. Gol'dfarb, and G. P. Gromova, Izv. Akad. Nauk USSR, Ser. Khim., 2733 (1973).
Ya. L. Gol'dfarb, A. P. Yakubov, and L. I. Belen'kii, Izv. Akad. Nauk USSR, Ser. Khim., 185, 941 (1969).
L. I. Belen'kii, I. B. Karmanova, and Ya. L. Gol'dfarb, Zh. Org. Khim., 7, 1743 (1971).
Ya. L. Gol'dfarb, I. B. Karmanova, Yu. B. Vol'kenshtein, and L. I. Belen'kii, Khim. Geterotsikl. Soedin., 1474 (1978). [Chem. Heterocycl. Compd., 14, 1196 (1978)].
Ya. L. Gol'dfarb, E. I. Novikova, and L. I. Belen'kii, Izv. Akad. Nauk USSR, Ser. Khim., 1233 (1971).
Ya. L. Gol'dfarb, E. I. Novikova, and L. I. Belen'kii, Izv. Akad. Nauk USSR, Ser. Khim., 2841 (1971).
L. I. Belen'kii, E. I. Novikova, and Ya. L. Gol'dfarb, Khim. Geterotsikl. Soedin., 1353 (1971). [Chem. Heterocycl. Compd., 7, 1265 (1971)].
D. M. Antonov, L. I. Belen'kii, and S. Gronowitz, J. Heterocycl. Chem., 32, 53 (1995).
G. Hart, D. R. Liljegren, and K. T. Potts, J. Chem. Soc., 4267 (1961).
S. Clementi, P. Linda, and G. Marino, J. Chem. Soc., 79 (1971).
Tran Quang Minh, F. Mantovani, P. Faller, L. Christiaens, and M. Renson, Bull. Soc. Chim. Fr., 3955 (1972).
G. Olah, K. K. Laali, Q. Wang, and G. K. Surya Prakash, Onium Ions, J. Wiley & Sons, New York (1998).
H. Meerwein, in: Houben-Weyl Methoden der Organischen Chemie, 4 Aufl., Bd. VI/3, G. Thieme Verlag, Stuttgart (1965), S. 325.
J. Goerdeler, in: Houben-Weyl Methoden der Organischen Chemie, 4 Aufl., Bd. IX, G. Thieme Verlag, Stuttgart (1955), S. 174.
H. Reinboldt, in: Houben-Weyl Methoden der Organischen Chemie, 4 Aufl., Bd. IX, G. Thieme Verlag, Stuttgart (1955), S. 917.
H. Meerwein, E. Battenberg, H. Gold, E. Pfeil, and G Willfang, J. Prakt. Chem., 154, 83 (1939).
L. I. Belen'kii, I. A. Suslov, and N. D. Chuvylkin, Khim. Geterotsikl. Soedin., 38 (2003). [Chem. Heterocycl. Compd., 39, 36 (1971)].
L. I. Belen'kii and. A. Abronin, Zh. Org. Khim., 17, 1129 (1981).
L. I. Belen'kii, in: 16th International Congress of Heterocyclic Chemistry Abstracts, August 10-15, 1997, Montana State University – Boseman, USA. OP-V-20.
L. I. Belen'kii, T. G. Kim, I. A. Suslov, and N. D. Chuvylkin, Izv. Akad. Nauk, Ser. Khim., 837 (2005).
A. R. Katritzky and A. F. Pozharskii, Handbook of Heterocyclic Chemistry, 2nd еd., Pergamon, Amsterdam (2000), p. 61.
C. F. Candy, R. A. Jones, and P. H. Wright, J. Chem. Soc., 2563 (1970).
A. A. Chaikovskaya, Yu. V. Dmitriv, S. P. Ivonin, A. M. Pinchuk, and A. A. Tolmachev, Heteroatom Chem., 16, 599 (2005).
A. A. Chaikovskaya, S. P. Ivonin, Yu. V. Dmitriv, and A. M. Pinchuk, Khim. Geterotsikl. Soedin., 1102 (2005). [Chem. Heterocycl. Compd., 41, 934 (2005)].
A. R. Katritzky, K. Suzuki, S. K. Singh, and Hai-Ying He, J. Org. Chem., 68, 5720 (2003).
B. L. Bray, P. H. Mathies, R. Naef, D. R. Solas, Th. T. Tidwell, D. R. Artis, and J. M. Muchowski, J. Org. Chem., 55, 6317 (1990).
A. R. Kennedy, A. I. Khalaf, C. J. Suckling, and R. D. Waigh, Acta Crystallogr., Sect. E: Struct. Rep. Online, 62, o3282 (2006); Chem. Abstr., 146, 263001 (2007).
R. C. Foitzik, E. K. Bowen, A. M. Taylor, F. M. Pfeffer, and A. Kaynak, Synth. Met., 157, 924 (2007); Chem. Abstr., 148, 285562 (2008).
H. J. Anderson, C. A. Loader, R. X. Xu, N. Le, N. J. Gogan, R. McDonald, and L. G. Edwards, Can. J. Chem., 63, 896 (1985).
R. X. Xu, H. J. Anderson, N. J. Gogan, C. A. Loader, and R. McDonald, Tetrahedron Lett., 22, 4899 (1981).
H. M. Gilow, Y. H. Hong, P. L. Millirons, R. C. Snyder, and W. J. Casteel, Jr., J. Heterocycl. Chem., 23, 1475 (1986).
J. Rokach, P. Hamel, M. Kakushima, and G. M. Smith, Tetrahedron Lett., 22, 4901 (1981).
M. Kakushima, P. Hamel, R. Frenette, and J. Rokach, J. Org. Chem., 48, 3214 (1983).
Ch. Song, D. W. Knight, and M. A. Whatton, Tetrahedron Lett., 45, 9573 (2004).
E. Baum, L. I. Belen'kii, V. G. Kul'nevich, and T. E. Goldovskaya, Khim. Geterotsikl. Soedin., 662 (1982). [Chem. Heterocycl. Compd., 18, 499 (1982)].
I. A. Abronin, L. I. Belen'kii, and Ya. L. Gol'dfarb, in: New Trends in Heterocyclic Chemistry, R. B. Mitra, N. R. Ayyangar, V. N. Gogte, R. M. Acheson, and N. Cromwell (editors), Elsevier, Amsterdam (1979), p. 154.
M. V. Sigalov and B. A. Trofimov, Zh. Org. Khim., 31, 801 (1995).
A. P. Terent'ev and L. A. Yanovskaya, Zh. Obshch. Khim., 19, 538 (1949).
A. Mizuno, Y. Kan, H. Fukami, T. Kamei, K. Miyazaki, S. Matsuki, and Y. Oyama, Tetrahedron Lett., 41, 6605 (2000).
T. Janosik, H. Shirani, N. Wahlström, I. Malky, B. Stensland, and J. Bergman, Tetrahedron, 62, 1699 (2006).
M. Renard and L. Hevesi, J. Chem. Soc., Chem. Commun., 688 (1986).
M. W. Majchrzak and G. Simchen, Tetrahedron, 42, 1299 (1986).
K. Hrnčariková and D. Végh, Molecules, 8, 536 (2003); Chem. Abstr., 140, 321185 (2004).
J. R. Carson and N. M. Davis, J. Org. Chem., 46, 839 (1981).
V. A. Budylin, M. Del K. Pina, and Yu. G. Bundel', Khim. Geterotsikl. Soedin., 562 (1984). [Chem. Heterocycl. Compd., 20, 460 (1984)].
J. DeSales, R. Greenhouse, and J. Muchowski, J. Org. Chem., 47, 3668 (1982).
V. A. Budylin, E. D. Matveeva, and A N. Kost, Khim. Geterotsikl. Soedin., 1235 (1980). [Chem. Heterocycl. Compd., 16, 933 (1980)].
V. A. Budylin, A. N. Kost, E. D. Matveeva, and V. I. Minkin, Khim. Geterotsikl. Soedin., 68 (1972). [Chem. Heterocycl. Compd., 8, 63 (1972)]
V. A. Budylin, M. S. Ermolenko, and A. N. Kost, Khim. Geterotsikl. Soedin., 921 (1978). [Chem. Heterocycl. Compd., 14, 742 (1978)].
L. I. Belen'kii, T. G. Kim, I. A. Suslov, and N. D. Chuvylkin, ARKIVOC, xiii, 59 (2003).
I. A. Abronin, L. I. Belen'kii, G. M. Zhidomirov, and Ya. L. Gol'dfarb, Zh. Org.. Khim., 17, 1134 (1981).
L. I. Belen'kii, N. D. Chuvylkin, T. G. Kim, and I. A. Suslov, in: International Conference "Chemistry Biology Interface: Synergistic New Frontiers", November 21–26, 2004, New Delhi, India, Abstr., IL92.
L. I. Belen'kii, N. D. Chuvylkin, A. I. Serykh, and I. A. Suslov, Zh. Org.. Khim., 41, 1362 (2005).
L. I. Belen'kii, I. D. Nesterov, and N. D. Chuvylkin, Khim. Geterotsikl. Soedin., 1647 (2006). [Chem. Heterocycl. Compd., 42, 1414 (2006)].
L. I. Belen'kii, I. D. Nesterov, and N. D. Chuvylkin, Khim. Geterotsikl. Soedin., 34 (2007). [Chem. Heterocycl. Compd., 43, 28 (2007)].
N. D. Chuvylkin, I. D. Nesterov, and L. I. Belen'kii, Izv. Akad. Nauk, Ser. Khim., 1425 (2007).
H. Cerfontain, Mechanistic Aspects in Aromatic Sulfonation and Desulfonation, Intersci. Publ., New York (1968).
C. W. F. Kort and H. Cerfontain, Rec. Trav. Chim., 88, 860 (1969).
B. V. Salov and A. I. Gershenovich, in: L. I. Belen'kii (editor), Production and Properties of Organic Compounds of Sulfur [in Russian], Khimiya, Moscow (1998), p. 260.
L. I. Belen'kii, I. D. Nesterov, and N. D. Chuvylkin, Khim. Geterotsikl. Soedin., 1645 (2008). [Chem. Heterocycl. Compd., 44, 1339 (2008)].
L. I. Belen'kii, N. D. Chuvylkin, and I. D. Nesterov, Izv. Akad. Nauk, Ser. Khim., 2163 (2011).
M. Speranza, Adv. Heterocycl. Chem., 40, 25 (1986).
M. Speranza, J. Chem. Soc., Chem. Commun., 1177 (1981).
C. Angelini, C. Sparapani, and M. Speranza, J. Am. Chem. Soc., 104, 7084 (1982).
L. I. Belen'kii, Adv. Heterocycl. Chem., 99, 143 (2010).
L. I. Belen'kii, G. P. Gromova, B. V. Lichitskii, and M. M. Krayushkin, Khim. Geterotsikl. Soedin., 1477 (1997). [Chem. Heterocycl. Compd., 33, 1276 (1997)].
L. I. Belen'kii, I. D. Nesterov, and N. D. Chuvylkin, Khim. Geterotsikl. Soedin., 1801 (2008). [Chem. Heterocycl. Compd., 44, 1460 (2008)].
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 2, pp. 256-274, February, 2012.
Rights and permissions
About this article
Cite this article
Belen’kii, L.I., Chuvylkin, N.D. & Nesterov, I.D. Positional selectivity in electrophilic substitution reactions of π-excessive heterocycles (review). Chem Heterocycl Comp 48, 241–257 (2012). https://doi.org/10.1007/s10593-012-0985-3
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10593-012-0985-3