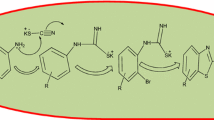
2,3-Disubstituted quinoxalines were synthesized via condensation of α-diketones and 1,2-phenylenediamines. Nano-BF3·SiO2 as a "green" and reusable solid acid was used as the catalyst for the synthesis of quinoxalines. The reaction was carried out at room temperature under sonication with high to excellent yields.
Similar content being viewed by others
References
S. D. Undevia, F. Innocenti, J. Ramirez, L. House, A. A. Desai, L. A. Skoog, D. A. Singh, T. Karrison, H. L. Kindler, and M. J. Ratain, Eur. J. Cancer, 44, 1684 (2008).
Q. Weng, D. Wang, P. Guo, L. Fang, Y. Hu, Q. He, and B. Yang, Eur. J. Pharmacol., 581, 262 (2008).
A. Budakoti, A. R. Bhat, and A. Azam, Eur. J. Med. Chem., 44, 1317 (2009).
S. Wagle, A. V. Adhikari, and N. S. Kumari, Eur. J. Med. Chem., 44, 1135 (2009).
E. Vicente, L. M. Lima, E. Bongard, S. Charnaud, R. Villar, B. Solano, A. Burguete, S. Perez-Silanes, I. Aldana, L. Vivas, and A. Monge, Eur. J. Med. Chem., 43, 1903 (2008).
A. Burguete, E. Pontiki, D. Hadjipavlou-Litina, R. Villar, E. Vicente, B. Solano, S. Ancizu, S. Pérez-Silanes, I. Aldana, and A. Monge, Bioorg. Med. Chem. Lett., 17, 6439 (2007).
X. Hui, J. Desrivot, C. Bories, P. M. Loiseau, X. Franck, R. Hocquemiller, and B. Figadère, Bioorg. Med. Chem. Lett., 16, 815 (2006).
J. Y. Jaung, Dyes and Pigments, 71, 245 (2006).
K. R. J. Thomas, M. Velusamy, J. T. Lin, C.H. Chuen, and Y. T. Tao, Chem. Mater., 17, 1860 (2005).
M. J. C. Crossley and L. A. Johnston, Chem. Commun., 1122 (2002).
S. Dailey, J. W. Feast, R. J. Peace, I. C. Sage, S. Till, and E. L. Wood, J. Mater. Chem., 11, 2238 (2001).
Z. Zhao, D. D. Wisnoski, S. E. Wolkenberg, W. H. Leister, Y. Wang, and C. W. Lindsley, Tetrahedron Lett., 45, 4873 (2004).
S. V. More, M. N. V. Sastry, C. C. Wang, and C. Yao, Tetrahedron Lett., 46, 6345 (2005).
M. M. Heravi, S. Taheri, K. Bakhtiari, and H. A. Oskooie, Catal. Commun., 8, 211 (2007).
M. M. Heravi, M. H. Tehrani, K. Bakhtiari, and H. A. Oskooie, Catal. Commun., 8, 1341 (2007).
A. Kumar, S. Kumar, A. Saxena, A. De, and S. Mozumdar, Catal. Commun., 9, 778 (2008).
J. J. Cai, J. P. Zou, X. Q. Pan, and W. Zhang, Tetrahedron Lett., 49, 7386 (2008).
T. K. Huang, R. Wang, L. Shi, and X. X. Lu, Catal. Commun., 9, 1143 (2008).
F. Dong, G. Kai, F. Zhenghao, Z. Xinli, and L. Zuliang, Catal. Commun., 9, 317 (2008).
M. M. Heravi, K. Bakhtiari, H. A. Oskooie, and S. Taheri, Heteroatom Chem., 19, 218 (2008).
S. Ajaikumar and A. Pandurangan, Appl. Catal. A, 357, 184 (2009).
B. F. Mirjalili, A. Bamoniri, and A. Akbari, J. Iran. Chem. Soc., 8, 135 (2011).
B. Sadeghi, B.F. Mirjalili, and M. M. Hashemi, Tetrahedron Lett., 49, 2575 (2008).
B. F. Mirjalili, A. Bamoniri, and A. Akbari, Tetrahedron Lett., 49, 6454 (2008).
B. Sadeghi, B. F. Mirjalili, and M. M. Hashemi, J. Iran. Chem. Soc., 5, 694 (2008).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 4, pp. 591–596, April, 2011.
Rights and permissions
About this article
Cite this article
Mirjalili, B.B.F., Bamoniri, A. & Akbari, A. Nano-BF3·SIO2: a reusable and eco-friendly catalyst for synthesis of quinoxalines. Chem Heterocycl Comp 47, 487–491 (2011). https://doi.org/10.1007/s10593-011-0785-1
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10593-011-0785-1