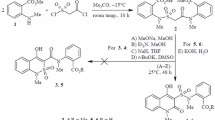
NMR spectroscopy showed that treatment of 1-allyl-3-[(arylamino)methylene]quinoline-2,4-(1H,3H)-diones with an equivalent amount of dry bromine in anhydrous acetic acid leads to the formation of 4-aryliminomethyl-2-bromomethyl-5-oxo-1,2-dihydro-5H-oxazolo[3,2-a]quinolinium bromides.
Similar content being viewed by others
References
I. V. Ukrainets, L. V. Sidorenko, A. A. Davidenko, and A. K. Yarosh, Khim. Geterotsikl. Soedin., 560 (2010). [Chem. Heterocycl. Comp., 46, 445 (2010)].
I. V. Ukrainets, Liu Yangyang, N. L. Bereznyakova, and A. V. Turov, Khim. Geterotsikl. Soedin., 1539 (2009). [Chem. Heterocycl. Comp., 45, 1235 (2009)].
I. V. Ukrainets, Liu Yanyang, A. A. Tkach, and A. V. Turov, Khim. Geterotsikl. Soedin., 1015 (2009). [Chem. Heterocycl. Comp., 45, 802 (2009)].
Author information
Authors and Affiliations
Corresponding author
Additional information
For Communication 174, see [1].
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 4, pp. 569-574, April, 2010.
Rights and permissions
About this article
Cite this article
Ukrainets, I.V., Bereznyakova, N.L., Yangyang, L. et al. 4-Hydroxy-2-quinolones. 175.*Reaction of -1-allyl-3-[(arylamino)methylene]quinoline-2,4-(1H,3H)-diones with bromine. Chem Heterocycl Comp 46, 452–456 (2010). https://doi.org/10.1007/s10593-010-0530-1
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10593-010-0530-1