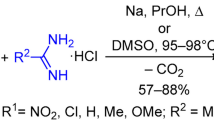
The heterocyclization of 2-acetyl-3-indolylacetic acid hydrazones and its amides, in contrast to similar derivatives of phenylacetic acid, does not lead to 2,3-dihydro-2-oxo-5-R1-1H-[1,2]diazepino-[4,5-b]indoles but rather to 2-aminoindolo[2,3-c]pyridin-3(2H)-one or azines of 2-acetyl-3-indolyl-acetic acid. 2,3-Dihydro-2-oxo-5-R1-1H-[1,2]diazepino[4,5-b]indoles were obtained by the reaction of 1-alkylaminoacetylindolo[2,3-c]pyrilium perchlorates and of methyl esters of 2-acetyl- and 2-propionyl-3-indolylacetic acid with hydrazine hydrate.
Similar content being viewed by others
References
E. J. Horvath, K. Horvath, T. Hamori, M. I. K. Fekete, S. Solyom, and M. Palkovits, Progress in Neurobiology , 60, 309 (2000).
T. Seppala, E. Palva, M. Mattila, K. Korttila, and R. Shrotriya, Psychopharmacology, 69, 209 (1980).
A. De Sarro, G. De Sarro, R. Gitto, S. Grasso, N. Micale, and M. Zappala, Farmaco, 54, 178 (1999).
G. De Sarro, R. Gitto, M. Zappala, and A. Chimirri, Farmaco, 57, 129 (2002).
A. Chimirri, R. Gitto, S. Quartarone, V. Orlando, A. De Sarro, and G. De Sarro, Farmaco, 57, 759 (2002).
A. Chimirri, G. De Sarro, A. De Sarro, R. Gitto, S. Quartarone, M. Zappala, A. Constanti, and V. Libri, J. Med. Chem., 41, 3409 (1998).
M. Zappala, R. Gitto, F. Bevacqua, S. Quartarone, A. Chimirri, M. Rizzo, G. De Sarro, and A. De Sarro, J. Med. Chem., 43, 4834 (2000).
R. Gitto, M. L. Barreca, L. De Luca, G. De Sarro, G. Ferreri, S. Quartarone, E. Russo, A. Constanti, and A. Chimirri, J. Med. Chem., 46, 197 (2003).
F. Gatta, D. Piazza, M. R. Del Giudice, and M. Massotti, Farmaco, 40, 942 (1985).
F. Bevacqua, A. Basso, R. Gitto, M. Bradly, and A. Chimirri, Tetrahedron Lett., 42, 7683 (2001).
A. Monge, M. T. Martinez, J. A. Palop, J. M. Mateo, and E. Fernandez-Alvarez, J. Heterocycl. Chem., 18, 889 (1981).
A. Monge, M. T. Martinez, J. A. Palop, T. Goni, and E. Fernandez-Alvarez, J. Heterocycl. Chem., 21, 381 (1984).
A. Monge, J. A. Palop, T. Goni, A. Martinez, and E. Fernandez-Alvarez, J. Heterocycl. Chem., 22, 1445 (1985).
V. S. Tolkunov, I. F. Perepichka, and V. I. Dulenko, J. Heterocycl. Chem., 42, 811 (2005).
V. S. Tolkunov, Yu. B. Vysotskii, O. A. Gorban’, S. V. Shishkina, O. V. Shishkin, R. I. Zubatyuk, and V. I. Dulenko, Khim. Geterotsikl. Soedin., 601 (2005). [Chem. Heterocycl. Comp., 41, 515 (2005)].
Yu. V. Zefirov and P. M. Zorkii, Usp. Khim., 58, 713 (1989).
G. M. Sheldrick, SHELXTL PLUS. PC Version. A system of Computer Programs for the Determination of Crystal Structure from X-ray Diffraction Data, Rev. 5.1 (1998).
H. Plieninger, W. Müller, and K. Weinert, Chem. Ber., 97, 667 (1964).
S. V. Tolkunov, M. N. Kal’nitskii, and E. A. Zemskaya, Khim. Geterotsikl. Soedin., 1552 (1991). [Chem. Heterocycl. Comp., 27, 1253 (1991)].
G. N. Dorofeenko, C. V. Krivun, and E. I. Sadekova, Khim. Geterotsikl. Soedin., 730 (1971). [Chem. Heterocycl. Comp., 7, 681 (1971)].
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 6, pp. 907–917, June, 2009.
Rights and permissions
About this article
Cite this article
Tolkunov, V.S., Eresko, A.B., Khizhan, A.I. et al. Heterocyclization of 2-acyl-3-indolylacetic acids using hydrazine. Synthesis of 2,3-dihydro-2-oxo-5-R1-1H-[1,2]diazepino[4,5-b]indoles. Chem Heterocycl Comp 45, 726–734 (2009). https://doi.org/10.1007/s10593-009-0322-7
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10593-009-0322-7