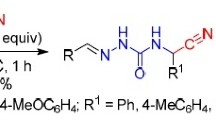
It has been shown that bromination of 5-aryl-5-benzylidene-3,5-dihydro-4H-2-methylthioimidazol-4-one in methylene chloride at room temperature leads to the addition of bromine at the double bond, but the the reaction of 5-aryl-3,5-dihydro-4H-2-methylthio-5-(2-pyridylmethylene)imidazol-4-one gave complex compound of the starting materials. After boiling both of the compounds obtained in ethanol the corresponding 5-[bromo(aryl)methylene]imidazolidine-2,4-diones were obtained.
Similar content being viewed by others
References
J. R. Lewis, Nat. Prod. Rep., 17, 57 (2000).
T. Lindel and H. Hoffmann, Tetrahedron Lett., 38, 8935 (1997).
J. Charton, A. C. Gassiot, S. Girault-Mizzi, M.-A. Debreu-Fontaine, P. Melnyk, and Ch. Sergheraert, Bioorg. Med. Chem. Lett., 15, 4833 (2005).
A. Degterev, Z. Huang, M. Boyce, Y. Li, P. Jagtap, N. Mizushima, G. D. Cuny, T. Mitchison, M. Moskowitz, and J. Yuan, Nat. Chem. Biol., 1, 112 (2005).
A. I. Khodair, Carbohydr. Res., 331, 445 (2001).
D. A. Hahn, M. J. McLean, and H. T. Murphy, J. Am. Chem. Soc., 60, 1927 (1938).
M. J. McLean and D. R. Seeger, J. Am. Chem. Soc., 66, 2020 (1944).
A. G. Mazhuga, E. K. Beloglazkina, S. Z. Vatsadze, N. A. Frolova, and N. V. Zyk, Izv. AN, Ser. Khim., 2734 (2004).
G. G. Muccioli, J. H. Poupaert, J. Wouters, B. Norberg, W. Poppitz, G. K. E. Scriba, and D. M. Lambert, Tetrahedron, 59, 1301 (2003).
Author information
Authors and Affiliations
Corresponding author
Additional information
Dedicated to Academician B. A. Trofimov on his 70th jubilee.
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 9, 1406–1408, September, 2008.
Rights and permissions
About this article
Cite this article
Antipin, R.L., Beloglazkina, E.K., Magouga, A.G. et al. A new method for the synthesis of 5-bromo(aryl)methylene-substituted hydantoins. Chem Heterocycl Comp 44, 1135–1137 (2008). https://doi.org/10.1007/s10593-008-0167-5
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10593-008-0167-5