Abstract
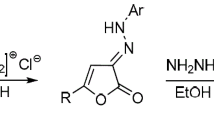
The reaction of 2,3-dihydro-2,3-benzo[b]furandione with the triphenylphosphazines of diazo compounds leads to substituted 2-methylenehydrazono-2,3-dihydro-3-benzo[b]furanones, which are hydrolyzed in an acidic medium to the substituted methylenehydrazides of o-hydroxyphenylglyoxalic acid and react with amines to form the products of addition at the activated CH=N bond of the side chain or 2-hydrazono-2,3-dihydro-3-benzo[b]furanone respectively.
Similar content being viewed by others
References
V. V. Zalesov, N. A. Pulina, and Yu. S. Andreichuk, Zh. Org. Chim., 25, 1054 (1989).
V. P. Kruglenko, V. P. Gnidets, N. A. Klyuev, and M. V. Povstyanoi, Khim. Geterotsikl. Soed. 533 (1987). [Chem. Heterocycl. Comp., 23, 444 (1987)].
H. J. Bestmann and H. Fritzsche, Chem. Ber., 94, 2477 (1961).
A. Schonberg and K. Sunahans, Chem. Ber., 97, 2539 (1964).
F. R. Hewgill, D. L. Hewitt, L. B. Howie, and W. L. Spencer, Austral. J. Chem., 30, 1971 (1977).
K. Fries and W. Pfaffendorf, Ber., 45, 154 (1912).
A. B. Tomchin, I. S. Ioffe, and E. A. Rusakov, Zh. Org. Chim., 10, 604 (1974).
Author information
Authors and Affiliations
Corresponding author
Additional information
__________
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 6, pp. 817–822, June, 2007.
Rights and permissions
About this article
Cite this article
Pulina, N.A., Zalesov, V.V. Synthesis and properties of substituted 2-methylene-hydrazono-2,3-dihydro-3-benzo[b]furanones. Chem Heterocycl Compd 43, 685–689 (2007). https://doi.org/10.1007/s10593-007-0111-0
Received:
Issue Date:
DOI: https://doi.org/10.1007/s10593-007-0111-0