Abstract
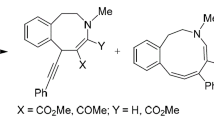
The transformations of 1-substituted tetrahydro-β-carbolines by the action of activated alkynes were studied. The action of dimethyl acetylenedicarboxylate in methanol gives products of the opening of the tetrahydropyridine fragment, namely, 2-methoxyalkylindoles. The action of ethyl propiolate in ethanol and of tosylacetylene in methanol gives mixtures of azocino[5,4-b]indoles and 2-alkoxyindoles. The action of ethyl propiolate in acetonitrile gives azocinoindoles.
Similar content being viewed by others
References
I. G. Voskressensky, T. N. Borisova, L. N. Kulikova, A. V. Varlamov, M. Catto, C. Altomare, and A. Carotti, Eur. J. Org. Chem., 3128 (2004).
I. G. Voskressensky, T. N. Borisova, T. A. Soklakova, L. N. Kulikova, 4. R. S. Borisov, and A. V. Varlamov, Lett. Org. Chem., 2, 18 (2005).
P. D. Bailey and S. P. Hollinshead, J. Chem. Soc., Perkin Trans. 1, 739 (1988).
A. V. Varlamov, T. N. Borisova, I. G. Voskressensky, B. Nsabimane, and A. I. Chernyshev, Heterocycl. Commun., 7, 461 (2001).
Author information
Authors and Affiliations
Additional information
__________
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 5, pp. 703–715, May, 2007.
Rights and permissions
About this article
Cite this article
Voskressensky, L.G., Borisova, T.N., Kulikova, L.N. et al. Reaction of 1-substituted tetrahydro-β-carbolines with activated alkynes-a new original approach to the synthesis of tetrahydroazocino[5,4-b]indoles. Chem Heterocycl Compd 43, 587–598 (2007). https://doi.org/10.1007/s10593-007-0093-y
Received:
Issue Date:
DOI: https://doi.org/10.1007/s10593-007-0093-y