Abstract
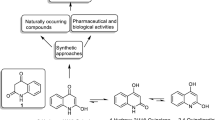
A modified method is proposed for preparation and purification, and the special features of the spatial structure have been studied for the ethyl ester of 1-hydroxy-3-oxo-5,6-dihydro-3H-pyrrolo[3,2,1-i,j]-quinoline-2-carboxylic acid.
Similar content being viewed by others
References
I. V. Ukrainets, L. V. Sidorenko, O. V. Gorokhov, and N. A. Jaradat, Khim. Geterotsikl. Soedin., 542 (2006).
P. Molnar and S. L. Erdo, Eur. J. Pharmacol., 311, 311 (1996).
C. A. Hicks, M. A. Ward, N. Ragumoorthy, S. J. Ambler, C. P. Dell, D. Dobson, and M. J. O’Neill, Brain. Res., 819, 65 (1999).
M. Rowley, J. J. Kulagowski, A. P. Watt, D. Rathbone, G. I. Stevenson, R. W. Carling, R. Baker, G. R. Marshall, J. A. Kemp, A. C. Foster, S. Grimwood, R. Hargreaves, C. Hurley, K. L. Saywell, M. D. Tricklebank, and P. D. Leeson, J. Med. Chem., 40, 4053 (1997).
I. V. Ukrainets, P. A. Bezuglyi, O. V. Gorokhova, V. I. Treskach, and A. V. Turov, Khim. Geterotsikl. Soedin., 105 (1993).
X. Collin, J. M. Robert, M. Duflos, G. Wielgosz, G. Le Baut, C. Robin-Dubigeon, N. Grimaud, F. Lang, and J. Y. Petit, J. Pharm. Pharmacol., 53, 417 (2001).
I. V. Ukrayinecz and P. A. Bezuhliy, US Patent 6340692 (2002); http://ep.espacenet.com.
K. Tsuji, G. W. Spears, K. Nakamura, T. Tojo, N. Seki, A. Sugiyama, and M. Matsuo, Bioorg. Med. Chem. Lett., 12, 85 (2002)
I. V. Ukrainets, S. A. El Kayal, O. V. Gorokhova, L. V. Sidorenko, and T. V. Alekseeva, Vestn. Farm., No. 1 (37), 12 (2004).
T. Kappe, C. Nuebling, K.-O. Westphalen, U. Kardorff, W. Deyn, M. Gerber, and H. Walter, DE Patent 4138820 (1993); http://ep.espacenet.com.
S. R. Khan, A. Mhaka, R. Pili, and J. T. Isaacs, Bioorg. Med. Chem. Lett., 11, 451 (2001).
I. V. Ukrainets, O. V. Gorokhova, S. G. Taran, P. A. Bezuglyi, A. V. Turov, N. A. Marusenko, and O. A. Evtifeeva, Khim. Geterotsikl. Soedin., 958 (1994).
H. Hayashi, Y. Miwa, S. Ichikawa, N. Yoda, I. Miki, A. Ishii, M. Kono, T. Yasuzawa, and F. Suzuki, J. Med. Chem., 36, 617 (1993).
S. Jönsson, G. Andersson, T. Fex, T. Fristedt, G. Hedlund, K. Jansson, L. Abramo, I. Fritzson, O. Pekarski, A. Runström, H. Sandin, I. Thuvesson, and A. Björk, J. Med. Chem., 47, 2075 (2004).
M. Rowley, P. D. Leeson, G. I. Stevenson, A. M. Moseley, I. Stansfield, I. Sanderson, L. Robinson, R. Baker, J. A. Kemp, G. R. Marshall, A. C. Foster, S. Grimwood, M. D. Tricklebank, and K. L. Saywell, J. Med. Chem., 36, 3386 (1993).
A. Kutyrev and T. Kappe, J. Heterocycl. Chem., 34, 969 (1997).
H.-B. Burgi and J. D. Dunitz, Struct. Correl., Vol. 2, VCH Weinheim (1994), p. 741.
Yu. V. Zefirov and P. M. Zorkii, Usp. Khim., 58, 713 (1989).
V. A. Strel’tsov and V. E. Zavodnik, Kristallografiya, 34, 1369 (1989).
G. M. Sheldrick, SHELX97. PC Version. A System of Computer Programs for Crystal Structure Solution and Refinement. Rev. 2 (1998).
Author information
Authors and Affiliations
Additional information
__________
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 5, pp. 718–723, May, 2006.
Rights and permissions
About this article
Cite this article
Ukrainets, I.V., Sidorenko, L.V., Gorokhova, O.V. et al. 4-hydroxy-2-quinolones. 94. Improved synthesis and structure of 1-hydroxy-3-oxo-5,6-dihydro-3h-pyrrolo[3,2,1-i,j]-quinoline-2-carboxylic acid ethyl ester. Chem Heterocycl Compd 42, 631–635 (2006). https://doi.org/10.1007/s10593-006-0138-7
Received:
Issue Date:
DOI: https://doi.org/10.1007/s10593-006-0138-7