Abstract
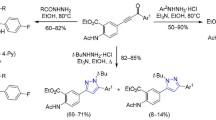
A convenient preparative two-stage method was developed for the isomerization of 2-phenacyl-1H-benzimidazole arylhydrazones to previously unknown 5-(o-aminoanilino)-1-aryl-3-phenylpyrazoles. The transformation scheme includes recyclization during acylation with trifluoroacetic anhydride leading to the formation of 1-aryl-3-phenyl-5-(o-trifluoroacetylaminoanilino)pyrazoles, which then undergo hydrazinolysis.
Similar content being viewed by others
REFERENCES
O. P. Shvaika and V. N. Artemov, Zh. Obshch. Khim., 41, 1788 (1972).
H. C. Van der Plas, Ring Transformation of Heterocycles, Wiley, New York, etc. (1972), Vol. 1, 2.
N. Vivona, S. Buscemi, V. Frenna, and G. Gusmano, in: A. R. Katritzky (editor), Advances in Heterocyclic Chemistry, Academic Press, New York (1993), Vol. 56, p. 49.
E. V. Babaev and N. S. Zefirov, Khim. Geterotsikl. Soedin., 1564 (1996).
G. Hajos, Z. Riedl, and G. Kollenz, Eur. J. Org. Chem., 18, 3405 2001).
I. B. Dzvinchuk and M. O. Lozinskii, Khim. Geterotsikl. Soedin., 206 (2005).
I. B. Dzvinchuk, A. V. Vypirailenko, and M. O. Lozinskii, Zh. Org. Khim., 32, 1759 (1996).
I. B. Dzvinchuk, A. V. Vypirailenko, and M. O. Lozinskii, Zh. Org. Khim., 33, 116 (1997).
Author information
Authors and Affiliations
Additional information
__________
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 7, pp. 1002–1006, July, 2005.
Rights and permissions
About this article
Cite this article
Dzvinchuk, I.B., Lozinskii, M.O. Isomerization of 2-Phenacyl-1H-benzimidazole Arylhydrazones to 1-Aryl-5-(o-aminoanilino)-3-phenylpyrazoles by Trifluoroacetylation and Hydrazinolysis. Chem Heterocycl Compd 41, 845–849 (2005). https://doi.org/10.1007/s10593-005-0236-y
Received:
Issue Date:
DOI: https://doi.org/10.1007/s10593-005-0236-y