Abstract
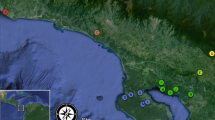
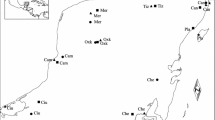
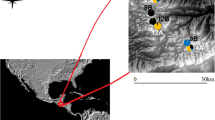
The prospects for persistence of bees living in fragmented landscapes is a topic of considerable interest due to bees’ importance as pollinators of agricultural crops and wild plants, coupled with the ubiquity of native habitat loss and evidence that bees may be declining worldwide. Population persistence in fragmented areas depends on dispersal potential and maintenance of gene flow among fragments of habitat. Here we used population genetic techniques to characterize, for two equally abundant orchid bee species that differ in their physiology and ecology, levels of genetic differentiation among fragments of tropical forest in southeastern Costa Rica in a ~200 km2 landscape. We measured population differentiation with ϕPT (an analogue to the traditional summary statistic Fst), as well as two measures that may more accurately reflect the level of differentiation when highly variable loci are used: G’st and Dest. We also calculated pairwise genetic distances among individuals and conducted Mantel tests to test the correlation of genetic and geographic distance, for each species. We found strong differences in genetic structure between the species. Contrary to our expectations, each measure of genetic structure revealed that the larger-bodied species, Eulaema bombiformis, had higher levels of differentiation than the smaller species, Euglossa championi. Furthermore, for Eulaema bombiformis there was a significant positive correlation of genetic and geographic distance while for Euglossa championi there was no significant positive correlation. Our results demonstrate that bee species can have strikingly different levels of gene flow in fragmented habitats, and that body size may not always act as a useful proxy for dispersal, even in closely related taxa.



Similar content being viewed by others
References
Bawa KS (1990) Plant-pollinator interactions in tropical rain-forests. Ann Rev Ecol Syst 21:399–422
Becker P (1991) More about euglossine bees in Amazonian forest fragments. Biotropica 23:586–591
Benjamini Y, Yekutieli D (2001) The control of the false discovery rate in multiple testing under dependency. Annals Stat 29:1165–1188
Biesmeijer JC, Roberts SPM, Reemer M et al (2006) Parallel declines in pollinators and insect-pollinated plants in Britain and the Netherlands. Science 313:351–354
Brosi B (2009) The effects of forest fragmentation on euglossine bee communities (Hymenoptera: Apidae: Euglossini). Biol Conserv 142:414–423
Burd M (1994) Bateman’s principle and plant reproduction: the role of pollen limitation in fruit and seed set. Bot Rev 60:83–139
Clement R, Horn S (2001) Pre-Columbian land-use history in Costa Rica: a 3000-year record of forest clearance, agriculture and fires from Laguna Zoncho. Holocene 11:419–426
Crawford NG (2010) smogd: software for the measurement of genetic diversity. Mol Ecol Res 10:556–557
Darveau CA, Hochachka PW, Welch KC Jr, Roubik DW, Suarez RK (2005) Allometric scaling of flight energetics in panamanian orchid bees: a comparative phylogenetic approach. J Exp Biol 208:3581–3591
Darvill B, Ellis JS, Lye GC, Goulson D (2006) Population structure and inbreeding in a rare and declining bumblebee, Bombus muscorum (Hymenoptera: Apidae). Mol Ecol 15:601–611
Darvill B, O’Connor S, Lye GC et al (2010) Cryptic differences in dispersal lead to differential sensitivity to habitat fragmentation in two bumblebee species. Mol Ecol 19:53–63
Dick CW, Roubik DW, Gruber KF, Bermingham E (2004) Long-distance gene flow and cross-Andean dispersal of lowland rainforest bees (Apidae: Euglossini) revealed by comparative mitochondrial DNA phylogeography. Mol Ecol 13:3775–3785
Ellis JS, Knight ME, Darvill B, Goulson D (2006) Extremely low effective population sizes, genetic structuring and reduced genetic diversity in a threatened bumblebee species, Bombus sylvarum (Hymenoptera: Apidae). Mol Ecol 15:4375–4386
Ewers RM, Didham RK (2006) Confounding factors in the detection of species responses to habitat fragmentation. Biol Rev 81:117–142
Excoffier L, Smouse PE, Quattro JM (1992) Analysis of molecular variance inferred from metric distances among DNA haplotypes: application to human mitochondrial DNA restriction data. Genetics 131:479–491
Exeler N, Kratochwil A, Hochkirch A (2008) Strong genetic exchange among populations of a specialist bee Andrena vaga (Hymenoptera: Andrenidae). Conserv Genet 9:1233–1241
Exeler N, Kratochwil A, Hochkirch A (2010) Does recent habitat fragmentation affect the population genetics of a heathland specialist, Andrena fuscipes (Hymenoptera: Andrenidae)? Conserv Genet 11:1679–1687
Goulson D, Lye GC, Darvill B (2008) Decline and conservation of bumble bees. Ann Rev Ent 53:191–208
Goulson D, Kaden JC, Lepais O, Lye GC, Darvill B (2011) Population structure, dispersal and colonization history of the garden bumblebee Bombus hortorum in the Western Isles of Scotland. Conserv Genet 12:869–879. doi:10.1007/s10592-011-0190-4
Greenleaf SS, Williams NM, Winfree R, Kremen C (2007) Bee foraging ranges and their relationship to body size. Oecologia 153:589–596
Grixti JC, Wong LT, Cameron SA, Favret C (2009) Decline of bumble bees (Bombus) in the North American Midwest. Biol Conserv 142:75–84
Haskell JP, Ritchie ME, Olff H (2002) Fractal geometry predicts varying body size scaling relationships for mammal and bird home ranges. Nature 418:527–530
Hedrick P (2005) A standardized genetic differentiation measure. Evolution 59:1633–1638
Heller R, Siegismund HR (2009) Relationship between three measures of genetic differentiation GST, DEST and G’ST: how wrong have we been? Mol Ecol 18:2080–2083
Hoehn M, Sarre D, Henle K (2007) The tales of two geckos: does dispersal prevent extinction in recently fragmented populations? Mol Ecol 16:3299–3312
Huff D, Peakall R, Smouse PE (1993) RAPD variation within and among natural populations of outcrossing buffalograss Buchloe dactyloides. Theor Appl Genet 86:927–934
Janzen D (1971) Euglossine bees as long-distance pollinators of tropical plants. Science 171:203–205
Jost LOU (2008) GST and its relatives do not measure differentiation. Mol Ecol 17:4015–4026
Kimura M, Crow J (1964) The number of alleles maintained in a finite population. Genetics 49:725–738
Kosior A, Celary W, Olejniczak P et al (2007) The decline of the bumble bees and cuckoo bees (Hymenoptera: Apidae: Bombini) of Western and Central Europe. Oryx 41:79–88
Matzkin L, Mutsaka K, Johnson S, Markow T (2009) Metabolic pools differ among ecologically diverse Drosophila species. J Insect Physiol 55:1145–1150
Narum SR (2006) Beyond Bonferroni: less conservative analyses for landscape genetics. Conserv Genet 7:783–787
Ollerton J, Winfree R, Tarrant S (2010) How many flowering plants are pollinated by animals? Oikos 120:321–326. doi:10.111/j.1600-0706.2010.18644.x
Paxton RJ, Zobel MU, Steiner J, Zillikens A (2009) Microsatellite loci for Euglossa annectans (Hymenoptera: Apidae) and their variability in other orchid bees. Mol Ecol Resour 9:1221–1223
Peakall ROD, Smouse PE (2006) genalex 6: genetic analysis in Excel. Population genetic software for teaching and research. Mol Ecol Notes 6:288–295
Powell A, Powell G (1987) Population dynamics of male euglossine bees in Amazonian forest fragments. Biotropica 19:176–179
Raw A (1989) The dispersal of euglossine bees between isolated patches of Eastern Brazilian wet forest (Hymenoptera, Apidae). Rev Bras Entomol 33:103–107
Roubik D (1989) Ecology and natural history of tropical bees. Cambridge University Press, Cambridge
Roubik D, Hanson P (2004) Orchid bees: biology and field guide. InBio Press, Heredia
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning: a laboratory manual, 2nd edn. Cold Spring Harbor Press, Cold Spring Harbor
Souza RO, Cervini M, Del Lama MA, Paxton RJ (2007) Microsatellite loci for euglossine bees (Hymenoptera: Apidae). Mol Ecol Notes 7:1352–1356
Stow A, Silberbauer L, Beattie AJ, Briscoe DA (2007) Fine-Scale genetic structure and fire-created habitat patchiness in the australian allodapine bee, exoneura nigrescens (Hymenoptera: Apidae). J Hered 98:60–66
Taylor A (2003) Assessing the consequences of inbreeding for population fitness: past challenges and future prospects. Cambridge University Press, Cambridge
Tonhasca A, Blackmer JL, Albuquerque GS (2002a) Abundance and diversity of euglossine bees in the fragmented landscape of the Brazilian Atlantic forest. Biotropica 34:416–422
Tonhasca A Jr, Blackmer JL, Albuquerque GS (2002b) Within-habitat heterogeneity of euglossine bee populations: a re-evaluation of the evidence. J Trop Ecol 18:929–933
Tonhasca AJ, Albuquerque GS, Blackmer JL (2003) Dispersal of euglossine bees between fragments of the Brazilian Atlantic forest. J Trop Ecol 19:99–102
Vamosi JC, Knight TM, Steets JA et al (2006) Pollination decays in biodiversity hotspots. Proc Natl Acad Sci 103:956–961
Whitehorn PR, Tinsley MC, Brown MJF, Darvill B, Goulson D (2011) Genetic diversity, parasite prevalence in wild bumblebees. Proc R Soc Lond B Biol Sci 278:1195–1202
Whitlock MC (2011) G’st and D do not replace Fst. Mol Ecol 20:1083–1091
Wikelski M, Moxley J, Eaton-Mordas A et al (2010) Large-range movements of neotropical orchid bees observed via radio telemetry. PLOS ONE 5:e10738
Winfree R, Aguilar R, Vazquez DP, LeBuhn G, Aizen MA (2009) A meta-analysis of bees’ responses to anthropogenic disturbance. Ecology 90:2068–2076
Woolnough DA, Downing JA, Newton TJ (2009) Fish movement and habitat use depends on water body size and shape. Ecol Fresh Fish 18:83–91
Wright S (1951) The genetical structure of natural populations. Ann Eugen 15:323–354
Zimmermann Y, Schorkopf DLP, Moritz RFA, Pemberton RW, Quezada-Euan JJG, Eltz T (2011) Population genetic structure of orchid bees (Euglossini) in anthropogenically altered landscapes. Conserv Genet. doi:10.1007/s10592-011-0221-1
Acknowledgments
We thank the staff at the Las Cruces Biological Station near San Vito, Costa Rica, J. Van Cleve and J. Lyons for helpful comments, M. Kaplan and the staff of University of Arizona’s genomics core facility for help with laboratory work, and T. Brookhart for fieldwork and cartographic assistance. SSS was supported by the Center for Insect Science at the University of Arizona (NIH grant 5 K12 GM000708). Original collection of the bee specimens by BJB was supported by grants to the Center for Conservation Biology at Stanford University from the Koret, McDonnell, Sherwood, and Winslow Foundations and Peter and Helen Bing. Additional fieldwork in Costa Rica by BJB was supported by the Woodruff Travel Fund of Emory University.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Suni, S.S., Brosi, B.J. Population genetics of orchid bees in a fragmented tropical landscape. Conserv Genet 13, 323–332 (2012). https://doi.org/10.1007/s10592-011-0284-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10592-011-0284-z