Abstract
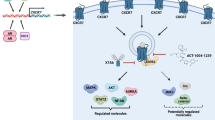
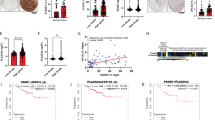
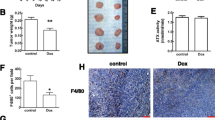
Prostate and breast cancer are major causes of death worldwide, mainly due to patient relapse upon disease recurrence through formation of metastases. Chemokines are small proteins with crucial roles in the immune system, and their regulation is finely tuned in early inflammatory responses. They are key molecules during inflammatory processes, and many studies are focusing on their regulatory functions in tumor growth and angiogenesis during metastatic cell seeding and spreading. Bindarit is an anti-inflammatory indazolic derivative that can inhibit the synthesis of MCP-1/CCL2, with a potential inhibitory function in tumor progression and metastasis formation. We show here that in vitro, bindarit can modulate cancer-cell proliferation and migration, mainly through negative regulation of TGF-β and AKT signaling, and it can impair the NF-κB signaling pathway through enhancing the expression of the NF-κB inhibitor IkB-α. In vivo administration of bindarit results in impaired metastatic disease in prostate cancer xenograft mice (PC-3M-Luc2 cells injected intra-cardially) and impairment of local tumorigenesis in syngeneic Balb/c mice injected under the mammary gland with murine breast cancer cells (4T1-Luc cells). In addition, bindarit treatment significantly decreases the infiltration of tumor-associated macrophages and myeloid-derived suppressor cells in 4T1-Luc primary tumors. Overall, our data indicate that bindarit is a good candidate for new therapies against prostate and breast tumorigenesis, with an action through impairment of inflammatory cell responses during formation of the tumor–stroma niche microenvironment.








Similar content being viewed by others
References
Havens AM et al (2008) An in vivo mouse model for human prostate cancer metastasis. Neoplasia 10(4):371–380
Spano D, Zollo M (2012) Tumor microenvironment: a main actor in the metastasis process. Clin Exp Metastasis 29(4):381–395
Pikarsky E et al (2004) NF-kappaB functions as a tumour promoter in inflammation-associated cancer. Nature 431(7007):461–466
Mizutani K et al (2009) The chemokine CCL2 increases prostate tumor growth and bone metastasis through macrophage and osteoclast recruitment. Neoplasia 11(11):1235–1242
Aggarwal MM et al (2010) Measurement of the bottom quark contribution to nonphotonic electron production in p + p collisions at sqrt[s] = 200 GeV. Phys Rev Lett 105(20):202301
Lu X, Kang Y (2009) Chemokine (C-C motif) ligand 2 engages CCR2+ stromal cells of monocytic origin to promote breast cancer metastasis to lung and bone. J Biol Chem 284(42):29087–29096
Zhang J, Lu Y, Pienta KJ (2010) Multiple roles of chemokine (C-C motif) ligand 2 in promoting prostate cancer growth. J Natl Cancer Inst 102(8):522–528
Takahashi M et al (2009) Chemokine CCL2/MCP-1 negatively regulates metastasis in a highly bone marrow-metastatic mouse breast cancer model. Clin Exp Metastasis 26(7):817–828
Nam JS et al (2006) Chemokine (C-C motif) ligand 2 mediates the prometastatic effect of dysadherin in human breast cancer cells. Cancer Res 66(14):7176–7184
Zhang J, Patel L, Pienta KJ (2010) CC chemokine ligand 2 (CCL2) promotes prostate cancer tumorigenesis and metastasis. Cytokine Growth Factor Rev 21(1):41–48
Loberg RD et al (2007) CCL2 as an important mediator of prostate cancer growth in vivo through the regulation of macrophage infiltration. Neoplasia 9(7):556–562
Molloy AP et al (2009) Mesenchymal stem cell secretion of chemokines during differentiation into osteoblasts, and their potential role in mediating interactions with breast cancer cells. Int J Cancer 124(2):326–332
Qian BZ et al (2011) CCL2 recruits inflammatory monocytes to facilitate breast-tumour metastasis. Nature 475(7355):222–225
Salcedo R et al (2000) Human endothelial cells express CCR2 and respond to MCP-1: direct role of MCP-1 in angiogenesis and tumor progression. Blood 96(1):34–40
Saji H et al (2001) Significant correlation of monocyte chemoattractant protein-1 expression with neovascularization and progression of breast carcinoma. Cancer 92(5):1085–1091
Mirolo M et al (2008) Impact of the anti-inflammatory agent bindarit on the chemokinome: selective inhibition of the monocyte chemotactic proteins. Eur Cytokine Netw 19(3):119–122
Mora E et al (2012) Bindarit: an anti-inflammatory small molecule that modulates the NFkappaB pathway. Cell Cycle 11(1):159–169
Bhatia M et al (2008) Treatment with bindarit, an inhibitor of MCP-1 synthesis, protects mice against trinitrobenzene sulfonic acid-induced colitis. Inflamm Res 57(10):464–471
Bhatia M et al (2005) Treatment with bindarit, a blocker of MCP-1 synthesis, protects mice against acute pancreatitis. Am J Physiol Gastrointest Liver Physiol 288(6):G1259–G1265
Ble A et al (2011) Antiproteinuric effect of chemokine C-C motif ligand 2 inhibition in subjects with acute proliferative lupus nephritis. Am J Nephrol 34(4):367–372
Guglielmotti A et al (2002) Amelioration of rat adjuvant arthritis by therapeutic treatment with bindarit, an inhibitor of MCP-1 and TNF-alpha production. Inflamm Res 51(5):252–258
Ialenti A et al (2011) Inhibition of in-stent stenosis by oral administration of bindarit in porcine coronary arteries. Arterioscler Thromb Vasc Biol 31(11):2448–2454
Rulli NE et al (2009) Amelioration of alphavirus-induced arthritis and myositis in a mouse model by treatment with bindarit, an inhibitor of monocyte chemotactic proteins. Arthritis Rheum 60(8):2513–2523
Rulli NE et al (2011) Protection from arthritis and myositis in a mouse model of acute chikungunya virus disease by bindarit, an inhibitor of monocyte chemotactic protein-1 synthesis. J Infect Dis 204(7):1026–1030
Zhou GX et al (2010) Protective effects of MCP-1 inhibitor on a rat model of severe acute pancreatitis. Hepatobiliary Pancreat Dis Int 9(2):201–207
Gazzaniga S et al (2007) Targeting tumor-associated macrophages and inhibition of MCP-1 reduce angiogenesis and tumor growth in a human melanoma xenograft. J Invest Dermatol 127(8):2031–2041
D’Angelo A, Garzia L, André A, Carotenuto P, Aglio V, Guardiola O, Arrigoni G, Cossu A, Palmieri G, Aravind L, Zollo M (2004) Prune cAMP phosphodiesterase binds nm23-H1 and promotes cancer metastasis. Cancer Cell 5(2):137–149
Roca H, Varsos Z, Pienta KJ (2008) CCL2 protects prostate cancer PC3 cells from autophagic death via phosphatidylinositol 3-kinase/AKT-dependent survivin up-regulation. J Biol Chem 283(36):25057–25073
Roca H et al (2008) CCL2, survivin and autophagy: new links with implications in human cancer. Autophagy 4(7):969–971
Yang L et al (2008) Abrogation of TGF beta signaling in mammary carcinomas recruits Gr-1+CD11b+ myeloid cells that promote metastasis. Cancer Cell 13(1):23–35
Mantovani A (2010) Molecular pathways linking inflammation and cancer. Curr Mol Med 10(4):369–373
Balkwill F (2004) Cancer and the chemokine network. Nat Rev Cancer 4(7):540–550
Lazennec G, Richmond A (2010) Chemokines and chemokine receptors: new insights into cancer-related inflammation. Trends Mol Med 16(3):133–144
Monti P et al (2003) The CC chemokine MCP-1/CCL2 in pancreatic cancer progression: regulation of expression and potential mechanisms of antimalignant activity. Cancer Res 63(21):7451–7461
Fujimoto H et al (2009) Stromal MCP-1 in mammary tumors induces tumor-associated macrophage infiltration and contributes to tumor progression. Int J Cancer 125(6):1276–1284
Ueno T et al (2000) Significance of macrophage chemoattractant protein-1 in macrophage recruitment, angiogenesis, and survival in human breast cancer. Clin Cancer Res 6(8):3282–3289
Spano D , Heckc C, De Antonellisa P, Christofori G, Zollo M (2012). Molecular Networks that Regulate Cancer Metastasis. Semin Cancer Biol. doi:10.1016/j.semcancer.2012.03.006. Accessed 30 March 2012
Soria G, Ben-Baruch A (2008) The inflammatory chemokines CCL2 and CCL5 in breast cancer. Cancer Lett 267(2):271–285
Li X et al (2009) A destructive cascade mediated by CCL2 facilitates prostate cancer growth in bone. Cancer Res 69(4):1685–1692
Lu Y et al (2009) Activation of MCP-1/CCR2 axis promotes prostate cancer growth in bone. Clin Exp Metastasis 26(2):161–169
Soria G et al (2008) Concomitant expression of the chemokines RANTES and MCP-1 in human breast cancer: a basis for tumor-promoting interactions. Cytokine 44(1):191–200
Lu Y et al (2006) Monocyte chemotactic protein-1 (MCP-1) acts as a paracrine and autocrine factor for prostate cancer growth and invasion. Prostate 66(12):1311–1318
Lu Y et al (2007) CCR2 expression correlates with prostate cancer progression. J Cell Biochem 101(3):676–685
Lu Y et al (2007) Monocyte chemotactic protein-1 mediates prostate cancer-induced bone resorption. Cancer Res 67(8):3646–3653
Izhak L et al (2012) Dissecting the autocrine and paracrine roles of the CCR2-CCL2 axis in tumor survival and angiogenesis. PLoS One 7(1):e28305
van Golen KL et al (2008) CCL2 induces prostate cancer transendothelial cell migration via activation of the small GTPase Rac. J Cell Biochem 104(5):1587–1597
Lu Y et al (2007) PTHrP-induced MCP-1 production by human bone marrow endothelial cells and osteoblasts promotes osteoclast differentiation and prostate cancer cell proliferation and invasion in vitro. Int J Cancer 121(4):724–733
Qian DZ et al (2010) CCL2 is induced by chemotherapy and protects prostate cancer cells from docetaxel-induced cytotoxicity. Prostate 70(4):433–442
Hembruff SL et al (2010) Loss of transforming growth factor-beta signaling in mammary fibroblasts enhances CCL2 secretion to promote mammary tumor progression through macrophage-dependent and -independent mechanisms. Neoplasia 12(5):425–433
Loberg RD et al (2007) Targeting CCL2 with systemic delivery of neutralizing antibodies induces prostate cancer tumor regression in vivo. Cancer Res 67(19):9417–9424
Zoja C et al (1998) Bindarit retards renal disease and prolongs survival in murine lupus autoimmune disease. Kidney Int 53(3):726–734
Guglielmotti A et al (1993) Chronic inflammatory response in the rat can be blocked by bindarit. Biochem Mol Biol Int 29(4):747–756
Cioli V et al (1992) A new protein antidenaturant agent, bindarit, reduces secondary phase of adjuvant arthritis in rats. J Rheumatol 19(11):1735–1742
Craig MJ, Loberg RD (2006) CCL2 (monocyte chemoattractant protein-1) in cancer bone metastases. Cancer Metastasis Rev 25(4):611–619
Rodrigues LR et al (2007) The role of osteopontin in tumor progression and metastasis in breast cancer. Cancer Epidemiol Biomarkers Prev 16(6):1087–1097
Rittling SR, Chambers AF (2004) Role of osteopontin in tumour progression. Br J Cancer 90(10):1877–1881
Solinas G et al (2010) Tumor-conditioned macrophages secrete migration-stimulating factor: a new marker for M2-polarization, influencing tumor cell motility. J Immunol 185(1):642–652
DuPre SA, Redelman D, Hunter KW Jr (2007) The mouse mammary carcinoma 4T1: characterization of the cellular landscape of primary tumours and metastatic tumour foci. Int J Exp Pathol 88(5):351–360
Morales JK et al (2010) GM-CSF is one of the main breast tumor-derived soluble factors involved in the differentiation of CD11b-Gr1- bone marrow progenitor cells into myeloid-derived suppressor cells. Breast Cancer Res Treat 123(1):39–49
Roland CL et al (2009) Inhibition of vascular endothelial growth factor reduces angiogenesis and modulates immune cell infiltration of orthotopic breast cancer xenografts. Mol Cancer Ther 8(7):1761–1771
Melancon MP et al (2010) Targeted imaging of tumor-associated M2 macrophages using a macromolecular contrast agent PG-Gd-NIR813. Biomaterials 31(25):6567–6573
Polfliet MM et al (2006) The rat macrophage scavenger receptor CD163: expression, regulation and role in inflammatory mediator production. Immunobiology 211(6–8):419–425
van Dongen M et al (2010) Anti-inflammatory M2 type macrophages characterize metastasized and tyrosine kinase inhibitor-treated gastrointestinal stromal tumors. Int J Cancer 127(4):899–909
Sanchez-Martin L et al (2011) The chemokine CXCL12 regulates monocyte-macrophage differentiation and RUNX3 expression. Blood 117(1):88–97
Shabo I et al (2008) Breast cancer expression of CD163, a macrophage scavenger receptor, is related to early distant recurrence and reduced patient survival. Int J Cancer 123(4):780–786
Asano K et al (2011) CD169-positive macrophages dominate antitumor immunity by crosspresenting dead cell-associated antigens. Immunity 34(1):85–95
Lau SK, Chu PG, Weiss LM (2004) CD163: a specific marker of macrophages in paraffin-embedded tissue samples. Am J Clin Pathol 122(5):794–801
Barral P et al (2010) CD169(+) macrophages present lipid antigens to mediate early activation of iNKT cells in lymph nodes. Nat Immunol 11(4):303–312
Allavena P et al (2008) The Yin-Yang of tumor-associated macrophages in neoplastic progression and immune surveillance. Immunol Rev 222:155–161
Roy PG, Thompson AM (2006) Cyclin D1 and breast cancer. Breast 15(6):718–727
Fu M et al (2004) Minireview: cyclin D1: normal and abnormal functions. Endocrinology 145(12):5439–5447
Neumeister P et al (2003) Cyclin D1 governs adhesion and motility of macrophages. Mol Biol Cell 14(5):2005–2015
Tsou CL et al (2007) Critical roles for CCR2 and MCP-3 in monocyte mobilization from bone marrow and recruitment to inflammatory sites. J Clin Invest 117(4):902–909
Acknowledgments
We thank the Core Services platforms of CEINGE Laboratories, and Prof. Luigi del Vecchio, Head of the Cell Sorter Facility, and its facility for helpful and critical discussions. This study was funded by Angelini Grants ACRAF 004FA09369, 004FA09383 (MZ) and the European GRANT-FP7-Tumic HEALTH-F2-2008-201662 (MZ). VDD was supported by Fondazione San Paolo (IM) and Tumic EU-FP7, DS was supported by the Dipartimento di Biochimica e Biotecnologie Mediche, ‘Federico II’ University of Naples, DMD was supported by Dottorato in Medicina Molecolare e Genetica, ‘Federico II’ University of Naples, and LL was supported by Scuola di Specializzazione in Genetica Medica, ‘Federico II’ University of Naples, Italy.
Author information
Authors and Affiliations
Corresponding author
Additional information
Valeria Di Dato and Daniela Spano contributed equally to the work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zollo, M., Di Dato, V., Spano, D. et al. Targeting monocyte chemotactic protein-1 synthesis with bindarit induces tumor regression in prostate and breast cancer animal models. Clin Exp Metastasis 29, 585–601 (2012). https://doi.org/10.1007/s10585-012-9473-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10585-012-9473-5