Abstract
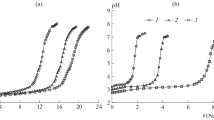
The method was developed allowing quantitative extraction of fulvic acids from soil samples in the form close to native. Formation of gel-like and soluble forms of iron(III) fulvates in the systems containing equivalent amounts of iron(III) and fulvic acid was studied within pH range 1–9. The molecular structure of fulvates and the mechanism of transformation of gel to soluble forms was analyzed by IR spectroscopy. Distribution of microamounts of europium between gel-like iron fulvates and their solutions was studied. The correlation between phase distribution of europium and solubilization of iron(III) fulvates was analyzed.
Similar content being viewed by others
References
L. N. Aleksandrova:Organic Substance of Soil and Processes of its Transformation, Nauka, Leningrad, 1980.
V. V. Ponomareva:Theory of Podzol Formation, Nauka, Moscow, 1964.
D. S. Orlov:Humic Acids of Soil and General Theory of Humification, Mosk. Gos. Univ., Moscow, 1980.
A. I. Karpukhin:Application of Gel Chromatography to Study of Fulvic Acids and Iron Fulvate Compounds. Features of Soil Processes in Soddy-Podzolic Soils, Moscow, 1977.
M. M. Kononova and N. A. Titova: Pochvovedenie (1951) No. 11, 81.
V. F. Kovalev and I. A. Generalova: Pochvovedenie (1967) No. 9, 135.
A. E. Vozbutskaya:The Chemistry of Soils, Moscow, 1968.
Yu. P. Davydov:Speciation of Radionuclides in Solutions, Nauka i Tekhnika, Minsk, 1978.
E. K. Legin, Yu. I. Trifonov, M. L. Khokhlov, and D. N. Suglobov: Radiokhimiya37 (1995) 275.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Legin, E.K., Trifonov, Y.I., Khokhlov, M.L. et al. Phase behavior of Eu(III) in fulvic acid-Fe(III)−H20 system at pH 1–9. Czech J Phys 49 (Suppl 1), 151–157 (1999). https://doi.org/10.1007/s10582-999-0021-8
Issue Date:
DOI: https://doi.org/10.1007/s10582-999-0021-8