Abstract
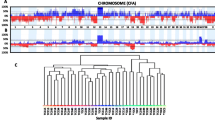
Urothelial carcinoma (UC), also referred to as transitional cell carcinoma (TCC), is the most common bladder malignancy in both human and canine populations. In human UC, numerous studies have demonstrated the prevalence of chromosomal imbalances. Although the histopathology of the disease is similar in both species, studies evaluating the genomic profile of canine UC are lacking, limiting the discovery of key comparative molecular markers associated with driving UC pathogenesis. In the present study, we evaluated 31 primary canine UC biopsies by oligonucleotide array comparative genomic hybridization (oaCGH). Results highlighted the presence of three highly recurrent numerical aberrations: gain of dog chromosome (CFA) 13 and 36 and loss of CFA 19. Regional gains of CFA 13 and 36 were present in 97 % and 84 % of cases, respectively, and losses on CFA 19 were present in 77 % of cases. Fluorescence in situ hybridization (FISH), using targeted bacterial artificial chromosome (BAC) clones and custom Agilent SureFISH probes, was performed to detect and quantify these regions in paraffin-embedded biopsy sections and urine-derived urothelial cells. The data indicate that these three aberrations are potentially diagnostic of UC. Comparison of our canine oaCGH data with that of 285 human cases identified a series of shared copy number aberrations. Using an informatics approach to interrogate the frequency of copy number aberrations across both species, we identified those that had the highest joint probability of association with UC. The most significant joint region contained the gene PABPC1, which should be considered further for its role in UC progression. In addition, cross-species filtering of genome-wide copy number data highlighted several genes as high-profile candidates for further analysis, including CDKN2A, S100A8/9, and LRP1B. We propose that these common aberrations are indicative of an evolutionarily conserved mechanism of pathogenesis and harbor genes key to urothelial neoplasia, warranting investigation for diagnostic, prognostic, and therapeutic applications.








Similar content being viewed by others
Abbreviations
- AMADID:
-
Agilent MicroArray Design Identifier
- BAC:
-
Bacterial artificial chromosome
- CDKN2A:
-
Cyclin-dependent kinase inhibitor 2A
- CFA:
-
Canis familiaris (also used as a prefix to canine chromosome numbers)
- CNA:
-
Copy number aberration
- DNA:
-
Deoxyribonucleic acid
- ECCS:
-
Evolutionarily conserved chromosome segment
- FASST2:
-
Fast Adaptive States Segmentation Technique 2
- FFPE:
-
Formalin-fixed paraffin embedded
- FISH:
-
Fluorescence in situ hybridization
- GO:
-
Gene ontology
- H&E:
-
Hematoxylin and eosin
- HSA:
-
Homo sapiens (also used as a prefix to human chromosome numbers)
- oaCGH:
-
Oligo-array comparative genomic hybridization
- OR:
-
Odds ratio
- PANTHER:
-
Protein Analysis Through Evolutionary Relationships
- PBS:
-
Phosphate-buffered saline
- RR:
-
Relative risk
- UC:
-
Urothelial carcinoma
- TCC:
-
Transitional cell carcinoma
References
American Cancer Society (2014) Cancer facts and figures 2014. Society AC, Atlanta
American Joint Committee on Cancer (2002) Urinary bladder. In: Greene F, Page D, Fleming I et al (eds) Cancer staging manual. Springer-Verlag, New York, pp 335–338
Anderson W, Dunham B, King J et al (1989) Presumptive subcutaneous transplation of a urinary bladder transitional cell carcinoma of the urinary bladder in a dog. Cornell Vet 79:263–266
Angstadt AY, Motsinger-Reif A, Thomas R et al (2011) Characterization of canine osteosarcoma by array comparative genomic hybridization and RT-qPCR: signatures of genomic imbalance in canine osteosarcoma parallel the human counterpart. Genes Chromosom Cancer 50(11):859–874
Angstadt AY, Thayanithy V, Subramanian S, Modiano JF, Breen M (2012) A genome-wide approach to comparative oncology: high-resolution oligonucleotide aCGH of canine and human osteosarcoma pinpoints shared microaberrations. Cancer Genet 205(11):572–587
Bansal N, Gupta A, Sankhwar SN, Mahdi AA (2014) Low- and high-grade bladder cancer appraisal via serum-based proteomics approach. Clin Chim Acta 436:97–103
Bhatlekar S, Fields JZ, Boman BM (2014) HOX genes and their role in the development of human cancers. J Mol Med (1432–1440 (Electronic))
Cai H, Kumar N, Bagheri H, von Mering C, Robinson M, Baudis M (2014) Chromothripsis-like patterns are recurring but heterogenously distributed features in a survery of 22,347 cancer genome screens. BMC Genomics 15:82
Calin GA, Dumitru CS et al (2004) Human microRNA genes are frequently located at fragile sites and genomic regions involved in cancers. Proc Natl Acad Sci 101(9):2999–3004
Castillo-Martin M, Domingo-Domenech J, Karni-Schmidt O, Matos T, Cordon-Cardo C (2010) Molecular pathways of urothelial development and bladder tumorigenesis. Urol Oncol 28(4):401–408
Chekaluk Y, Wu CL, Rosenberg J et al (2013) Identification of nine genomic regions of amplification in urothelial carcinoma, correlation with stage, and potential prognostic and therapeutic value. PLoS One 8(4):e60927
Chen R, Feng C, Xu Y (2011) Cyclin-dependent kinase-associated protein Cks2 is associated with bladder cancer progression. J Int Med Res 39(2):533–540
Development Team R (2010) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Dhawan D, Ramos-Vara JA, Stewart JC, Zheng R, Knapp DW (2009) Canine invasive transitional cell carcinoma cell lines: in vitro tools to complement a relevant animal model of invasive urinary bladder cancer. Urol Oncol 27(3):284–292
Dobson JM (2013) Breed-predispositions to cancer in pedigree dogs. ISRN Veterinary Science 2013
Ebbing J, Mathia S, Seibert F et al (2013) Urinary calprotectin: a new diagnostic marker in urothelial carcinoma of the bladder. World J Urol 1–8
Eliseeva IA, Lyabin DN, Ovchinnikov LP (2013) Poly(A)-binding proteins: structure, domain organization, and activity regulation. Biochemistry Moscow 78(13):1377–1391
Fadl-Elmula I (2005) Chromosomal changes in uroepithelial carcinomas. Cell Chromosom 4:1
Goebell PJ, Knowles MA (2010) Bladder cancer or bladder cancers? Genetically distinct malignant conditions of the urothelium. Urol Oncol 28(4):409–428
Hall M, Frank E, Holmes G, Pfahringer B, Reutemann P, Witten IH (2009) The WEKA Data Mining Software: an update. SIGKDD Explorations 11(1)
Hedan B, Thomas R, Motsinger-Reif A et al (2011) Molecular cytogenetic characterization of canine histiocytic sarcoma: a spontaneous model for human histiocytic cancer identifies deletion of tumor suppressor genes and highlights influence of genetic background on tumor behavior. BMC Cancer (in revision)
Heidenblad M, Lindgren D, Jonson T et al (2008) Tiling resolution array CGH and high density expression profiling of urothelial carcinomas delineate genomic amplicons and candidate target genes specific for advanced tumors. BMC Med Genomics 1:3
Hosseini SA, Horton S, Saldivar JC et al (2013) Common chromosome fragile sites in human and murine epithelial cells and FHIT/FRA3B loss-induced global genome instability. Genes Chromosom Cancer 52(11):1017–1029
Karolchik D, Hinrichs AS, Furey TS et al (2004) The UCSC Table Browser data retrieval tool. Nucleic Acids Res 32(suppl 1):D493–D496
Kim WJ, Kim SK, Jeong P et al (2011) A four-gene signature predicts disease progression in muscle invasive bladder cancer. Mol Med 17(5–6):478–485
Kim WT, Kim J, Yan C et al (2014) S100A9 and EGFR gene signatures predict disease progression in muscle invasive bladder cancer patients after chemotherapy. Ann Oncol 25(5):974–979
Knapp DW, Glickman NW, Denicola DB, Bonney PL, Lin TL, Glickman LT (2000) Naturally-occurring canine transitional cell carcinoma of the urinary bladder A relevant model of human invasive bladder cancer. Urol Oncol 5(2):47–59
Knapp D, Ramos-Vara J, Moore G, Dhawan D, Bonney P, Young K (2014) Urinary bladder cancer in dogs, a naturally occurring model for cancer biology and drug development. ILAR J 55(1):100–118
Langbein S, Szakacs O, Wilhelm M et al (2002) Alteration of the LRP1B gene region is associated with high grade of urothelial cancer. Lab Investig 82(5):639–643
Ma K, Qiu L, Mrasek K et al (2012) Common fragile sites: genomic hotspots of DNA damage and carcinogenesis. Int J Mol Sci 13(9):11974–11999
Mutsaers AJ, Widmer WR, Knapp DW (2003) Canine transitional cell carcinoma. J Vet Intern Med 17(2):136–144
Oliveira PA, Arantes-Rodrigues R, Vasconcelos-Nóbrega C (2014) Animal models of urinary bladder cancer and their application to novel drug discovery. Expert Opin Drug Discov 9(5):485–503
Panani A, Roussos C (2006) Sex chromosome abnormalities in bladder cancer: Y polysomies are linked to PT1-grade III transitional cell carcinoma. Anticancer Res 26(1):319–323
Poorman K, Borst L, Moroff S et al (2014) Comparative cytogenetic characterization of primary canine melanocytic lesions using array CGH and fluorescence in situ hybridization. Chromosom Res (in press)
Prazeres H, Torres J, Rodrigues F, Pinto M (2011) Chromosomal, epigenetic and microRNA-mediated inactivation of LRP1B, a modulator of the extracellular envrionemnt of thyroid cancer cells. Oncogene 30:1302–1317
Quinlan AR, Hall IM (2010) BEDTools: a flexible suite of utilities for comparing genomic features. Bioinformatics 26(6):841–842
Richards R (2001) Fragile and unstable chromosomes in cancer: causes and consequences. Trends Genet 17(6):339–345
Richter J, Beffa L, Wagner U et al (1998) Patterns of chromosomal imbalances in advanced urinary bladder cancer detected by comparative genomic hybridization. Am J Pathol 153(5):1615–1621
Sahrief Y, Reich C, Bonar R (1980) Polyploidy in mammalian urothelial cells. Urol Res 8:153–161
Schulz W (2006) Understanding urothelial carcinoma through cancer pathways. Int J Cancer 119:1513–1518
Shearin AL, Ostrander EA (2010) Leading the way: canine models of genomics and disease. Dis Model Mech 3(1–2):27–34
Simoneau M, LaRue H, Aboulkassim T, Meyer F, Moore L, Fradet Y (2000) Chromosome 9 deletions and recurrence of superficial bladder cancer: identification of four regions of prognostic interest. Oncogene 19(54):6317–6323
Stephens P, Greenman C, Fu B, Yang F et al (2011) Massive genomic rearrangement required in a single catastrophic event during cancer development. Cell 144(1):27–40
The Cancer Genome Atlas Research Network (2014) Comprehensive molecular characterization of urothelial bladder carcinoma. Nature 507(7492):315–322
Thomas PD, Campbell MJ, Kejariwal A et al (2003a) PANTHER: a library of protein families and subfamilies indexed by function. Genome Res 13(9):2129–2141
Thomas PD, Kejariwal A, Campbell MJ et al (2003b) PANTHER: a browsable database of gene products organized by biological function, using curated protein family and subfamily classification. Nucleic Acids Res 31(1):334–341
Thomas R, Smith KC, Ostrander EA, Galibert F, Breen M (2003c) Chromosome aberrations in canine multicentric lymphomas detected with comparative genomic hybridisation and a panel of single locus probes. Br J Cancer 89(8):1530–1537
Thomas R, Seiser EL, Motsinger-Reif A et al (2011) Refining tumor-associated aneuploidy through 'genomic recoding' of recurrent DNA copy number aberrations in 150 canine non-Hodgkin lymphomas. Leuk Lymphoma 52(7):1321–1335
Thomas R, Borst L, Rotroff D et al (2014) Genomic profiling reveals extensive heterogeneity in somatic DNA copy number aberrations of canine hemangiosarcoma. Chromosom Res 1–15
van Duin M, van Marion R, Vissers K et al (2005) High-resolution array comparative genomic hybridization of chromosome arm 8q: evaluation of genetic progression markers for prostate cancer. Genes Chromosom Cancer 44(4):438–449
Xiao H, Li H, Yu G et al (2014) MicroRNA-10b promotes migration and invasion through KLF4 and HOXD10 in human bladder cancer. Oncol Rep 31(4):1832–1838
Acknowledgments
The canine oaCGH and FISH data generated in this study were funded by the NCSU-CVM Cancer Genomics Fund (MB). SS was supported in part by a Graduate Fellowship from the NCSU Comparative Biomedical Sciences Graduate Program, an NIH-T35 grant, a Triangle Community Foundation award, a George Hitchings New Investigator Award in Health Research, and the NCSU-CVM Cancer Genomics Fund (MB). We gratefully acknowledge support of Skippy Frank Fund for Life Sciences and Translational Research/ Rockefeller Philanthropy Advisors (awarded to MB/JS), whose funding supported SR as a Skippy Frank Translational Postdoctoral Fellow. J.D.S. holds the Edward B. Clark, MD Chair in Pediatric Research, and is supported through the Primary Children’s Hospital (PCH) Pediatric Cancer Program funded by the Intermountain Healthcare Foundation and the Primary Children’s Hospital Foundation. We thank Rachael Thomas for assistance with humanization of canine CGH data and Clint Mason for valuable informatics advice.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Conly Rieder.
Electronic supplementary material
Below is the link to the electronic supplementary material.
SOM Fig. 1
Gene ontology analysis highlighted gene function categories frequency affected by shared copy number aberration in human and canine UC. A GO analysis was performed as a part of the pathway analysis done in PANTHER. Conserved copy number gains and losses are shown as the inner and outer donut plots, respectively, with number of genes affected shown in each category. These data indicated that genes associated with metabolic processes (GO:0008152), cell processes (GO:0009987), and biological regulation (GO:0065007) were the most prominent among human and canine UC. Each of these three processes is highlighted in the corresponding donut plot. (PDF 802 kb)
Rights and permissions
About this article
Cite this article
Shapiro, S.G., Raghunath, S., Williams, C. et al. Canine urothelial carcinoma: genomically aberrant and comparatively relevant. Chromosome Res 23, 311–331 (2015). https://doi.org/10.1007/s10577-015-9471-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10577-015-9471-y