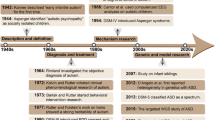
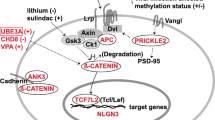
Abstract
The Autism Spectrum Disorder (ASD) consists of a prevalent and heterogeneous group of neurodevelopmental diseases representing a severe burden to affected individuals and their caretakers. Despite substantial improvement towards understanding of ASD etiology and pathogenesis, as well as increased social awareness and more intensive research, no effective drugs have been successfully developed to resolve the main and most cumbersome ASD symptoms. Hence, finding better treatments, which may act as “disease-modifying” agents, and novel biomarkers for earlier ASD diagnosis and disease stage determination are needed. Diverse mutations of core components and consequent malfunctions of several cell signaling pathways have already been found in ASD by a series of experimental platforms, including genetic associations analyses and studies utilizing pre-clinical animal models and patient samples. These signaling cascades govern a broad range of neurological features such as neuronal development, neurotransmission, metabolism, and homeostasis, as well as immune regulation and inflammation. Here, we review the current knowledge on signaling pathways which are commonly disrupted in ASD and autism-related conditions. As such, we further propose ways to translate these findings into the development of genetic and biochemical clinical tests for early autism detection. Moreover, we highlight some putative druggable targets along these pathways, which, upon further research efforts, may evolve into novel therapeutic interventions for certain ASD conditions. Lastly, we also refer to the crosstalk among these major signaling cascades as well as their putative implications in therapeutics. Based on this collective information, we believe that a timely and accurate modulation of these prominent pathways may shape the neurodevelopment and neuro-immune regulation of homeostatic patterns and, hopefully, rescue some (if not all) ASD phenotypes.






Similar content being viewed by others
Abbreviations
- β-TrCP:
-
Beta-transducin repeat-containing protein
- ADH:
-
Alcohol dehydrogenase
- AKT:
-
Protein kinase B (PKB)
- ALDH:
-
Retinaldehyde dehydrogenase
- ALDH1A2:
-
ALDH isoform 1A2
- AMPA:
-
α-Amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid
- ASD:
-
Autism spectrum disorder
- APC:
-
Adenomatous polyposis coli
- ATP:
-
Adenosine triphosphate
- BCL2:
-
B-cell CLL/lymphoma 2 apoptosis regulator
- BCL9:
-
B-cell CLL/lymphoma 9 transcription co-activator
- BDNF:
-
Brain-derived neurotrophic factor
- BMP:
-
Bone morphogenetic protein
- BRAF :
-
Proto-oncogene B-Raf encoding gene
- CBP:
-
CREB(cAMP response element)-binding protein
- CDC:
-
Centers for Disease Control and Prevention (USA)
- CDH7/8/9:
-
Chromodomain helicase DNA binding protein 7/8/9
- CEP290:
-
Centrosomal protein of 290 kDa
- CK1α:
-
Casein kinase 1α
- CNS:
-
Central nervous system
- CSF:
-
Cerebrospinal fluid
- CTNF:
-
Ciliary neurotrophic factor
- CTNNB1 :
-
Catenin beta 1 gene (β-catenin gene)
- Cyp26:
-
Cytochrome P450 enzyme family 26
- Dhh:
-
Desert Hedgehog
- DISC1:
-
Disrupted in schizophrenia 1
- DNMT:
-
DNA methyltransferase
- DKK:
-
Dickkopf
- DSM-5:
-
Diagnostic and statistical manual of mental disorders 5th edition
- Dvl:
-
Disheveled
- DYRK1B:
-
Dual specificity tyrosine phosphorylation regulated kinase 1B
- EIF4E :
-
Eukaryotic translation initiation factor 4E gene
- EGF:
-
Epidermal growth factor
- EGFR:
-
Epidermal growth factor receptor
- EPHA7 :
-
Ephrin type-A receptor 7 gene
- ERK/MAPK/MEK:
-
Mitogen-activated protein kinase
- FDA:
-
Food and Drug Administration
- FMRP:
-
Fragile X mental retardation protein
- FXS:
-
Fragile X syndrome
- FZD:
-
Frizzled (family of G protein-coupled receptors)
- GABA:
-
Gamma-amino butyric acid
- GDF:
-
Growth and differentiation factor
- Gli:
-
Glioma-associated oncogene/transcription factor
- GM-CSF:
-
Granulocyte–macrophage colony-stimulating factor
- Grb2:
-
Growth factor receptor-bound protein
- GSK-3β:
-
Glycogen synthase kinase 3β
- Hh:
-
Hedgehog
- HOX:
-
Homeobox
- HRAS :
-
HRas GTPase gene
- HTR2C :
-
Hydroxytryptamine (serotonin) receptor (human, X chromosomal) gene
- IκB:
-
Inhibitor of nuclear factor kappa-B
- I-SMADs:
-
Inhibitory SMADs
- IGF-1:
-
Insulin-like growth factor 1
- IFN-γ:
-
Interferon gamma
- IL:
-
Interleukin
- Ihh:
-
Indian Hedgehog
- IKKα:
-
IκB kinase alpha
- IKKβ:
-
IκB kinase beta
- IKKγ:
-
IκB kinase gamma
- iNOS:
-
Inducible nitric oxide synthase
- iPSC:
-
Induced pluripotent stem cell
- JAK:
-
Janus kinase
- KRAS:
-
Kirsten rat sarcoma viral oncogene homolog
- LEF:
-
Lymphoid enhancer factor
- LOF:
-
Loss of function
- LRP5/6:
-
Lipoprotein receptor-related protein 5/6
- MeCP2:
-
Methyl-CpG-binding protein 2
- MEK1/2 :
-
Mitogen-activated protein kinase 1 or 2 gene
- mTOR:
-
Mammalian target of rapamycin
- mTORC1:
-
MTOR Complex 1
- mTORC2:
-
MTOR Complex 2
- NFI:
-
Neurofibromatosis Type I
- NF-κB:
-
Nuclear factor kappa B
- NIK:
-
NF-κB inducing kinase
- NMDA:
-
N-Methyl-d-aspartate
- NPC:
-
Neural precursor/progenitor cell
- p300:
-
E1A binding protein p300, transcription co-activator
- Ptch1:
-
Patched 1
- PCDH20 :
-
Protocadherin-20 gene
- PI3K:
-
Phosphoinositide 3-kinase
- PIP2:
-
Phosphatidylinositol 4,5-bisphosphate
- PIP3:
-
Phosphatidylinositol 3,4,5-bisphosphate
- PKC:
-
Protein kinase C
- PTEN:
-
Phosphatase and tensin homolog
- RA:
-
Retinoic acid
- RAI1:
-
Retinoic acid inducible
- RAR:
-
Retinoic acid receptor
- RARE:
-
Retinoic acid response element
- RAS:
-
Rat sarcoma viral oncogene homolog
- RAF1 :
-
RAF proto-oncogene serine/threonine-protein kinase gene
- RDH:
-
Retinol dehydrogenases
- RelA:
-
Nuclear factor NF-kappa B p65 subunit
- RelB:
-
RELB proto-oncogene, NF-κB Subunit
- RERE :
-
Arginine-glutamic acid dipeptide repeats
- RERE:
-
Arginine-glutamic acid dipeptide repeats encoded nuclear receptor coregulator
- RHD:
-
Rel homology domain
- RHEB:
-
Ras homolog enriched in brain
- ROR:
-
Retinoic acid-related orphan receptors
- RORA :
-
Retinoic acid-related orphan receptor α gene
- RORE:
-
ROR response elements
- R-SMADs:
-
Receptor-regulated SMADs
- RXR:
-
Retinoid X receptor
- S6K:
-
Ribosomal protein S6 kinase
- SEMA3A :
-
Semaphorin-3A gene
- SFARI:
-
Simons foundation autism research initiative
- SFRP:
-
Secreted frizzled-related proteins
- SGK1:
-
Serine/threonine-protein kinase
- Shh:
-
Sonic Hedgehog
- SLOS:
-
Smith-Lemli-Opitz syndrome
- SMO:
-
Smoothened
- SNP:
-
Single-nucleotide polymorphism
- SOS:
-
Son of sevenless
- SOCS:
-
Suppressors of cytokine signaling proteins
- SSRI:
-
Selective serotonin reuptake inhibitor
- STAT:
-
Signal transducer and activator of transcription
- SuFu:
-
Suppressor of fused
- TAK1:
-
Mitogen-activated protein kinase 7 (MAP3K7)
- TCF:
-
T-cell factor
- TET:
-
Ten eleven translocation
- TF:
-
Transcription factor
- Th:
-
T-helper cell
- TGF-β:
-
Transforming growth factor beta
- TGFBR1:
-
Type 1 TGF-β receptor
- TGFBR2:
-
Type 2 TGF-β receptor
- TLR:
-
Toll-like receptor
- TNF:
-
Tumor necrosis factor
- TNFR:
-
Tumor necrosis factors receptors
- Treg:
-
Regulatory T-cell
- TSC1/2:
-
Tuberous sclerosis complex 1 or 2
- Tyk2:
-
Tyrosine kinase 2
- UBE3A:
-
Ubiquitin-protein Ligase E3 = E6AP ubiquitin-protein ligase
- VA:
-
Valproic acid
- Wnt:
-
Wingless-type/Int-1 (known as: INT1 and related gene products)
- WIF:
-
Wnt inhibiting factor
References
Adler BA, Wink LK, Early M et al (2015) Drug-refractory aggression, self-injurious behavior, and severe tantrums in autism spectrum disorders: a chart review study. Autism 19:102–106. https://doi.org/10.1177/1362361314524641
Ahmad SF, Nadeem A, Ansari MA et al (2017a) Upregulation of IL-9 and JAK-STAT signaling pathway in children with autism. Prog Neuropsychopharmacol Biol Psychiatry 79:472–480. https://doi.org/10.1016/j.pnpbp.2017.08.002
Ahmad SF, Zoheir KMA, Ansari MA et al (2017b) Dysregulation of Th1, Th2, Th17, and T regulatory cell-related transcription factor signaling in children with autism. Mol Neurobiol 54:4390–4400. https://doi.org/10.1007/s12035-016-9977-0
Ahmad SF, Ansari MA, Nadeem A et al (2018) Resveratrol attenuates pro-inflammatory cytokines and activation of JAK1-STAT3 in BTBR T(+) Itpr3(tf)/J autistic mice. Eur J Pharmacol 829:70–78. https://doi.org/10.1016/j.ejphar.2018.04.008
Ahmad SF, Ansari MA, Nadeem A et al (2020) Inhibition of tyrosine kinase signaling by tyrphostin AG126 downregulates the IL-21/IL-21R and JAK/STAT pathway in the BTBR mouse model of autism. Neurotoxicology 77:1–11. https://doi.org/10.1016/j.neuro.2019.12.003
Al-Ayadhi LY (2012) Relationship between sonic hedgehog protein, brain-derived neurotrophic factor and oxidative stress in autism spectrum disorders. Neurochem Res 37:394–400. https://doi.org/10.1007/s11064-011-0624-x
Al-Ayadhi L, Alhowikan AM, Halepoto DM (2018) Impact of auditory integrative training on transforming growth factor-beta1 and its effect on behavioral and social emotions in children with autism spectrum disorder. Med Princ Pract 27:23–29. https://doi.org/10.1159/000486572
Alvarez-Buylla A, Ihrie RA (2014) Sonic hedgehog signaling in the postnatal brain. Semin Cell Dev Biol 33:105–111. https://doi.org/10.1016/j.semcdb.2014.05.008
American Psychiatric Publishing (2013) Diagnostic and statistical manual of mental disorders, 5th edn. American Psychiatric Publishing, New York
Aoki CA, Borchers AT, Li M et al (2005) Transforming growth factor beta (TGF-beta) and autoimmunity. Autoimmunol Rev 4:450–459. https://doi.org/10.1016/j.autrev.2005.03.006
Ashwood P, Enstrom A, Krakowiak P et al (2008) Decreased transforming growth factor beta1 in autism: a potential link between immune dysregulation and impairment in clinical behavioral outcomes. J Neuroimmunol 204:149–153. https://doi.org/10.1016/j.jneuroim.2008.07.006
Ashwood P, Krakowiak P, Hertz-Picciotto I et al (2011) Elevated plasma cytokines in autism spectrum disorders provide evidence of immune dysfunction and are associated with impaired behavioral outcome. Brain Behav Immunol 25:40–45. https://doi.org/10.1016/j.bbi.2010.08.003
Baio J, Wiggins L, Christensen DL et al (2018) Prevalence of Autism spectrum disorder among children aged 8 years—autism and developmental disabilities monitoring network, 11 sites, United States, 2014. MMWR Surveill Summ 67:1–23. https://doi.org/10.15585/mmwr.ss6706a1
Bashir S, Halepoto DM, Al-Ayadhi L (2014) Serum level of desert hedgehog protein in autism spectrum disorder: preliminary results. Med Princ Pract 23:14–17. https://doi.org/10.1159/000354295
Belgacem YH, Hamilton AM, Shim S et al (2016) The many hats of sonic hedgehog signaling in nervous system development and disease. J Dev Biol. https://doi.org/10.3390/jdb4040035
Benitez-Burraco A, Lattanzi W, Murphy E (2016) Language impairments in ASD resulting from a failed domestication of the human brain. Front Neurosci 10:373. https://doi.org/10.3389/fnins.2016.00373
Besedovsky HO, del Rey A, Klusman I et al (1991) Cytokines as modulators of the hypothalamus-pituitary-adrenal axis. J Steroid Biochem Mol Biol 40:613–618. https://doi.org/10.1016/0960-0760(91)90284-c
Biever A, Valjent E, Puighermanal E (2015) Ribosomal protein S6 phosphorylation in the nervous system: From regulation to function. Front Mol Neurosci 8:75. https://doi.org/10.3389/fnmol.2015.00075
Bilder DA, Bakian AV, Stevenson DA et al (2016) Brief report: the prevalence of neurofibromatosis type 1 among children with autism spectrum disorder identified by the autism and developmental disabilities monitoring network. J Autism Dev Disord 46:3369–3376. https://doi.org/10.1007/s10803-016-2877-3
Blassberg R, Macrae JI, Briscoe J, Jacob J (2016) Reduced cholesterol levels impair smoothened activation in Smith-Lemli-Opitz syndrome. Hum Mol Genet 25:693–705. https://doi.org/10.1093/hmg/ddv507
Boland MR, Tatonetti NP (2016) Investigation of 7-dehydrocholesterol reductase pathway to elucidate off-target prenatal effects of pharmaceuticals: a systematic review. Pharmacogenomics J 16:411–429. https://doi.org/10.1038/tpj.2016.48
Boyle SC, Kim M, Valerius MT et al (2011) Notch pathway activation can replace the requirement for Wnt4 and wnt9b in mesenchymal-to-epithelial transition of nephron stem cells. Development 138:4245–4254. https://doi.org/10.1242/dev.070433
Bozdagi O, Tavassoli T, Buxbaum JD (2013) Insulin-like growth factor-1 rescues synaptic and motor deficits in a mouse model of autism and developmental delay. Mol Autism 4:9. https://doi.org/10.1186/2040-2392-4-9
Bozzi Y, Provenzano G, Casarosa S (2018) Neurobiological bases of autism–epilepsy comorbidity: a focus on excitation/inhibition imbalance. Eur J Neurosci 47:534–548. https://doi.org/10.1111/ejn.13595
Breder CD, Dinarello CA, Saper CB (1988) Interleukin-1 immunoreactive innervation of the human hypothalamus. Science 240:321–324. https://doi.org/10.1126/science.3258444
Caban C, Khan N, Hasbani D, Crino PB (2016) Genetics of tuberous sclerosis complex: implications for clinical practice. Appl Clin Genet 10:1–8. https://doi.org/10.2147/TACG.S90262
Carballo GB, Honorato JR, de Lopes GPF, de Spohr TCL (2018) A highlight on sonic hedgehog pathway. Cell Commun Signal 16:11. https://doi.org/10.1186/s12964-018-0220-7
Cargnello M, Roux PP (2011) Activation and function of the MAPKs and their substrates, the MAPK-activated protein kinases. Microbiol Mol Biol Rev 75:50–83. https://doi.org/10.1128/MMBR.00031-10
Cataldo I, Azhari A, Esposito G (2018) A review of oxytocin and arginine-vasopressin receptors and their modulation of autism spectrum disorder. Front Mol Neurosci 11:27. https://doi.org/10.3389/fnmol.2018.00027
Chatterjee S, Sil PC (2019) Targeting the crosstalks of Wnt pathway with Hedgehog and Notch for cancer therapy. Pharmacol Res 142:251–261. https://doi.org/10.1016/j.phrs.2019.02.027
Chen L, Tao Y, Song F et al (2016) Evidence for genetic regulation of mRNA expression of the dosage-sensitive gene retinoic acid induced-1 (RAI1) in human brain. Sci Rep 6:19010. https://doi.org/10.1038/srep19010
Cheng B, Zhu J, Yang T et al (2020) Vitamin A deficiency increases the risk of gastrointestinal comorbidity and exacerbates core symptoms in children with autism spectrum disorder. Pediatr Res. https://doi.org/10.1038/s41390-020-0865-y
Chenn A, Walsh CA (2002) Regulation of cerebral cortical size by control of cell cycle exit in neural precursors. Science 297:365–369. https://doi.org/10.1126/science.1074192
Choi GB, Yim YS, Wong H et al (2016) The maternal interleukin-17a pathway in mice promotes autism-like phenotypes in offspring. Science 351:933–939. https://doi.org/10.1126/science.aad0314
Chopko TC, Lindsley CW (2018) Classics in chemical neuroscience: risperidone. ACS Chem Neurosci 9:1520–1529. https://doi.org/10.1021/acschemneuro.8b00159
Clapcote SJ, Lipina TV, Millar JK et al (2007) Behavioral phenotypes of Disc1 missense mutations in mice. Neuron 54:387–402. https://doi.org/10.1016/j.neuron.2007.04.015
Clevers H, Nusse R (2012) Wnt/β-catenin signaling and disease. Cell 149:1192–1205. https://doi.org/10.1016/j.cell.2012.05.012
Coghlan S, Horder J, Inkster B et al (2012) GABA system dysfunction in autism and related disorders: from synapse to symptoms. Neurosci Biobehav Rev 36:2044–2055. https://doi.org/10.1016/j.neubiorev.2012.07.005
Correa RG, Tergaonkar V, Ng JK et al (2004) Characterization of NF-kappa B/I kappa B proteins in zebra fish and their involvement in notochord development. Mol Cell Biol 24:5257–5268. https://doi.org/10.1128/MCB.24.12.5257-5268.2004
Croen LA, Grether JK, Yoshida CK et al (2011) Antidepressant use during pregnancy and childhood autism spectrum disorders. Arch Gen Psychiatry 68:1104–1112. https://doi.org/10.1001/archgenpsychiatry.2011.73
Cukier HN, Dueker ND, Slifer SH et al (2014) Exome sequencing of extended families with autism reveals genes shared across neurodevelopmental and neuropsychiatric disorders. Mol Autism 5:1. https://doi.org/10.1186/2040-2392-5-1
Dawson G, Jones EJH, Merkle K et al (2012) Early behavioral intervention is associated with normalized brain activity in young children with autism. J Am Acad Child Adolesc Psychiatry 51:1150–1159. https://doi.org/10.1016/j.jaac.2012.08.018
de la Torre-Ubieta L, Won H, Stein JL, Geschwind DH (2016) Advancing the understanding of autism disease mechanisms through genetics. Nat Med 22:345–361. https://doi.org/10.1038/nm.4071
del Rey A, Besedovsky HO, Sorkin E et al (1981) Immunoregulation mediated by the sympathetic nervous system, II. Cell Immunol 63:329–334. https://doi.org/10.1016/0008-8749(81)90012-5
Delbroek H, Steyaert J, Legius E (2011) An 8.9 year old girl with autism and Gorlin syndrome. Eur J Paediatr Neurol 15:268–270. https://doi.org/10.1016/j.ejpn.2010.12.001
Deverman BE, Patterson PH (2009) Cytokines and CNS development. Neuron 64:61–78. https://doi.org/10.1016/j.neuron.2009.09.002
DiCarlo GE, Aguilar JI, Matthies HJ et al (2019) Autism-linked dopamine transporter mutation alters striatal dopamine neurotransmission and dopamine-dependent behaviors. J Clin Investig 129:3407–3419. https://doi.org/10.1172/JCI127411
Dong F, Jiang J, McSweeney C et al (2016) Deletion of CTNNB1 in inhibitory circuitry contributes to autism-associated behavioral defects. Hum Mol Genet 25:2738–2751. https://doi.org/10.1093/hmg/ddw131
Durak O, Gao F, Kaeser-Woo YJ et al (2016) Chd8 mediates cortical neurogenesis via transcriptional regulation of cell cycle and Wnt signaling. Nat Neurosci 19:1477–1488. https://doi.org/10.1038/nn.4400
Ehninger D, Han S, Shilyansky C et al (2008) Reversal of learning deficits in a Tsc2+/- mouse model of tuberous sclerosis. Nat Med 14:843–848. https://doi.org/10.1038/nm1788
Eissa N, Al-Houqani M, Sadeq A et al (2018) Current enlightenment about etiology and pharmacological treatment of autism spectrum disorder. Front Neurosci. https://doi.org/10.3389/fnins.2018.00304
El Gohary TM, El Aziz NA, Darweesh M, Sadaa ES (2015) Plasma level of transforming growth factor β 1 in children with autism spectrum disorder. Egypt J Ear, Nose, Throat Allied Sci 16:69–73. https://doi.org/10.1016/j.ejenta.2014.12.002
El Hokayem J, Weeber E, Nawaz Z (2018) Loss of Angelman syndrome protein E6AP disrupts a novel antagonistic estrogen-retinoic acid transcriptional crosstalk in neurons. Mol Neurobiol 55:7187–7200. https://doi.org/10.1007/s12035-018-0871-9
El-Ansary A, Al-Ayadhi L (2012) Neuroinflammation in autism spectrum disorders. J Neuroinflamm 9:265. https://doi.org/10.1186/1742-2094-9-265
Elenkov IJ, Wilder RL, Chrousos GP, Vizi ES (2000) The sympathetic nerve—an integrative interface between two supersystems: the brain and the immune system. Pharmacol Rev 52:595–638
Ellis H, Ma CX (2019) PI3K inhibitors in breast cancer therapy. Curr Oncol Rep 12:342
Emanuele E, Orsi P, Boso M et al (2010) Low-grade endotoxemia in patients with severe autism. Neurosci Lett 471:162–165. https://doi.org/10.1016/j.neulet.2010.01.033
Enstrom AM, Onore CE, Water JA, Ashwood P (2010) Differential monocyte responses to TLR ligands in children with autism spectrum disorders. Brain Behav Immunol 24:64–71. https://doi.org/10.1016/j.bbi.2009.08.001
Fakhoury M (2015) Autistic spectrum disorders: a review of clinical features, theories and diagnosis. Int J Dev Neurosci 43:70–77. https://doi.org/10.1016/j.ijdevneu.2015.04.003
Fang WQ, Chen WW, Jiang L et al (2014) Overproduction of upper-layer neurons in the neocortex leads to autism-like features in mice. Cell Rep 9:1635–1643. https://doi.org/10.1016/j.celrep.2014.11.003
Faravelli I, Bucchia M, Rinchetti P et al (2014) Motor neuron derivation from human embryonic and induced pluripotent stem cells: experimental approaches and clinical perspectives. Stem Cell Res Ther 5:87. https://doi.org/10.1186/scrt476
Faridar A, Jones-Davis D, Rider E et al (2014) Mapk/Erk activation in an animal model of social deficits shows a possible link to autism. Mol Autism 5:57. https://doi.org/10.1186/2040-2392-5-57
Farmer C, Thurm A, Grant P (2013) Pharmacotherapy for the core symptoms in autistic disorder: current status of the research. Drugs 73:303–314. https://doi.org/10.1007/s40265-013-0021-7
Fatemi SH, Halt AR, Stary JM et al (2002) Glutamic acid decarboxylase 65 and 67 kDa proteins are reduced in autistic parietal and cerebellar cortices. Biol Psychiatry 52:805–810. https://doi.org/10.1016/s0006-3223(02)01430-0
Fragoso YD, Stoney PN, Shearer KD et al (2015) Expression in the human brain of retinoic acid induced 1, a protein associated with neurobehavioural disorders. Brain Struct Funct 220:1195–1203. https://doi.org/10.1007/s00429-014-0712-1
Fraser MM, Bayazitov IT, Zakharenko SS, Baker SJ (2008) Phosphatase and tensin homolog, deleted on chromosome 10 deficiency in brain causes defects in synaptic structure, transmission and plasticity, and myelination abnormalities. Neuroscience 151:476–488. https://doi.org/10.1016/j.neuroscience.2007.10.048
Fregeau B, Kim BJ, Hernandez-Garcia A et al (2016) De novo mutations of RERE cause a genetic syndrome with features that overlap those associated with proximal 1p36 deletions. Am J Hum Genet 98:963–970. https://doi.org/10.1016/j.ajhg.2016.03.002
Fuccillo M, Joyner AL, Fishell G (2006) Morphogen to mitogen: the multiple roles of hedgehog signalling in vertebrate neural development. Nat Rev Neurosci 7:772–783. https://doi.org/10.1038/nrn1990
Funamoto S, Meili R, Lee S et al (2002) Spatial and temporal regulation of 3-phosphoinositides by PI 3-kinase and PTEN mediates chemotaxis. Cell 109:611–623. https://doi.org/10.1016/S0092-8674(02)00755-9
Furmanski AL, Saldana JI, Ono M et al (2013) Tissue-derived hedgehog proteins modulate Th differentiation and disease. J Immunol 190:2641–2649. https://doi.org/10.4049/jimmunol.1202541
Garg S, Brooks A, Burns A et al (2017) Autism spectrum disorder and other neurobehavioural comorbidities in rare disorders of the Ras/MAPK pathway. Dev Med Child Neurol 59:544–549. https://doi.org/10.1111/dmcn.13394
Ghanizadeh A (2012) Malondialdehyde, Bcl-2, superoxide dismutase and glutathione peroxidase may mediate the association of sonic hedgehog protein and oxidative stress in autism. Neurochem Res 37:899–901. https://doi.org/10.1007/s11064-011-0667-z
Ghyselinck NB, Duester G (2019) Retinoic acid signaling pathways. Development 146:167502. https://doi.org/10.1242/dev.167502
Giaccone G, Bazhenova LA, Nemunaitis J et al (2015) A phase III study of belagenpumatucel-L, an allogeneic tumour cell vaccine, as maintenance therapy for non-small cell lung cancer. Eur J Cancer 51:2321–2329. https://doi.org/10.1016/j.ejca.2015.07.035
Goines PE, Croen LA, Braunschweig D et al (2011) Increased midgestational IFN-gamma, IL-4 and IL-5 in women bearing a child with autism: a case-control study. Mol Autism 2:13. https://doi.org/10.1186/2040-2392-2-13
Goldani AAS, Downs SR, Widjaja F et al (2014) Biomarkers in autism. Front Psychiatry 5:100. https://doi.org/10.3389/fpsyt.2014.00100
Gottfried C, Bambini-Junior V, Francis F et al (2015) The impact of neuroimmune alterations in autism spectrum disorder. Front Psychiatry 6:121. https://doi.org/10.3389/fpsyt.2015.00121
Greenblatt EJ, Spradling AC (2018) Fragile X mental retardation 1 gene enhances the translation of large autism-related proteins. Science 361:709–712. https://doi.org/10.1126/science.aas9963
Guo M, Zhu J, Yang T et al (2018) Vitamin A improves the symptoms of autism spectrum disorders and decreases 5-hydroxytryptamine (5-HT): a pilot study. Brain Res Bull 137:35–40. https://doi.org/10.1016/j.brainresbull.2017.11.001
Guo M, Zhu J, Yang T et al (2019) Vitamin A and vitamin D deficiencies exacerbate symptoms in children with autism spectrum disorders. Nutr Neurosci 22:637–647. https://doi.org/10.1080/1028415X.2017.1423268
Halepoto DM, Bashir S, Zeina R, Al-Ayadhi LY (2015) Correlation between hedgehog (Hh) PROTEIN family and brain-derived neurotrophic factor (BDNF) in autism spectrum disorder (ASD). J Coll Phys Surg Pak 25:882–885
Hayward P, Kalmar T, Arias AM (2008) Wnt/Notch signalling and information processing during development. Development 135:411–424. https://doi.org/10.1242/dev.000505
Hellings JA, Arnold LE, Han JC (2017) Dopamine antagonists for treatment resistance in autism spectrum disorders: review and focus on BDNF stimulators loxapine and amitriptyline. Expert Opin Pharmacother 18:581–588. https://doi.org/10.1080/14656566.2017.1308483
Hill SA, Blaeser AS, Coley AA et al (2019) Sonic hedgehog signaling in astrocytes mediates cell type-specific synaptic organization. Elife. https://doi.org/10.7554/eLife.45545
Hirsch MM, Deckmann I, Santos-Terra J et al (2020) Effects of single-dose antipurinergic therapy on behavioral and molecular alterations in the valproic acid-induced animal model of autism. Neuropharmacology. https://doi.org/10.1016/j.neuropharm.2019.107930
Hormozdiari F, Penn O, Borenstein E, Eichler EE (2015) The discovery of integrated gene networks for autism and related disorders. Genome Res. https://doi.org/10.1101/gr.178855.114.142
Hu Y, Chen W, Wu L et al (2019) TGF-β1 restores hippocampal synaptic plasticity and memory in Alzheimer model via the PI3K/Akt/Wnt/β-catenin signaling pathway. J Mol Neurosci 67:142–149. https://doi.org/10.1007/s12031-018-1219-7
Huang W-H, Guenthner CJ, Xu J et al (2016) Molecular and neural functions of Rai1, the causal gene for smith-magenis syndrome. Neuron 92:392–406. https://doi.org/10.1016/j.neuron.2016.09.019
Ikeda H, Hideshima T, Fulciniti M et al (2010) PI3K/p110δ is a novel therapeutic target in multiple myeloma. Blood 116:1460–1468. https://doi.org/10.1182/blood-2009-06-222943
Inoki K, Li Y, Zhu T et al (2002) TSC2 is phosphorylated and inhibited by Akt and suppresses mTOR signalling. Nat Cell Biol 4:648–657. https://doi.org/10.1038/ncb839
Jaramillo TC, Speed HE, Xuan Z et al (2017) Novel Shank3 mutant exhibits behaviors with face validity for autism and altered striatal and hippocampal function. Autism Res 10:42–65. https://doi.org/10.1002/aur.1664
Jyonouchi H, Sun S, Le H (2001) Proinflammatory and regulatory cytokine production associated with innate and adaptive immune responses in children with autism spectrum disorders and developmental regression. J Neuroimmunol 120:170–179. https://doi.org/10.1016/s0165-5728(01)00421-0
Kahn M (2014) Can we safely target the WNT pathway? Nat Rev Drug Discov 13:513–532. https://doi.org/10.1038/nrd4233
Kalkman HO (2012) A review of the evidence for the canonical Wnt pathway in autism spectrum disorders. Mol Autism 3:1–12. https://doi.org/10.1186/2040-2392-3-10
Kaminska B, Gozdz A, Zawadzka M et al (2009) MAPK signal transduction underlying brain inflammation and gliosis as therapeutic target. Anat Rec (Hoboken) 292:1902–1913. https://doi.org/10.1002/ar.21047
Karunaweera N, Raju R, Gyengesi E, Münch G (2015) Plant polyphenols as inhibitors of NF-κB induced cytokine production—a potential anti-inflammatory treatment for Alzheimer’s disease? Front Mol Neurosci 8:24
Kashima R, Hata A (2018) The role of TGF-beta superfamily signaling in neurological disorders. Acta Biochim Biophys Sin (Shanghai) 50:106–120. https://doi.org/10.1093/abbs/gmx124
Katoh Y, Katoh M (2009) Hedgehog target genes: mechanisms of carcinogenesis induced by aberrant hedgehog signaling activation. Curr Mol Med 9:873–886. https://doi.org/10.2174/156652409789105570
Kaur N, Lu B, Monroe RK et al (2005) Inducers of oxidative stress block ciliary neurotrophic factor activation of Jak/STAT signaling in neurons. J Neurochem 92:1521–1530. https://doi.org/10.1111/j.1471-4159.2004.02990.x
Kawashima N, Nakayama K, Itoh K et al (2010) Reversible dimerization of EGFR revealed by single-molecule fluorescence imaging using quantum dots. Chem A Eur J 16:1186–1192. https://doi.org/10.1002/chem.200902963
Kerbeshian J, Burd L, Avery K (2001) Pharmacotherapy of autism: a review and clinical approach. J Dev Phys Disabil 13:199–228. https://doi.org/10.1023/A:1016686802786
Kern JK, Geier DA, King PG et al (2015) Shared brain connectivity issues, symptoms, and comorbidities in autism spectrum disorder, attention deficit/hyperactivity disorder, and tourette syndrome. Brain Connect 5:321–335. https://doi.org/10.1089/brain.2014.0324
Kern JK, Geier DA, Sykes LK, Geier MR (2016) Relevance of neuroinflammation and encephalitis in autism. Front Cell Neurosci 9:519. https://doi.org/10.3389/fncel.2015.00519
Khakzad MR, Salari F, Javanbakht M et al (2015) Transforming growth factor beta 1 869T/C and 915G/C polymorphisms and risk of autism spectrum disorders. Rep Biochem Mol Biol 3:82–88
Khatri N, Man H-Y (2019) The autism and angelman syndrome protein Ube3A/E6AP: the gene, E3 ligase ubiquitination targets and neurobiological functions. Front Mol Neurosci 12:109
Khaw P, Grehn F, Holló G et al (2007) A phase III study of subconjunctival human anti-transforming growth factor beta(2) monoclonal antibody (CAT-152) to prevent scarring after first-time trabeculectomy. Ophthalmology 114:1822–1830. https://doi.org/10.1016/j.ophtha.2007.03.050
Kilander MBC, Wang C-H, Chang C-H et al (2018) A rare human CEP290 variant disrupts the molecular integrity of the primary cilium and impairs Sonic Hedgehog machinery. Sci Rep 8:17335. https://doi.org/10.1038/s41598-018-35614-x
Klaus A, Birchmeier W (2008) WNT signalling and its impact on development and cancer. Nat Rev Cancer 8:387
Klein SD, Nguyen DC, Bhakta V et al (2019) Mutations in the sonic hedgehog pathway cause macrocephaly-associated conditions due to crosstalk to the PI3K/AKT/mTOR pathway. Am J Med Genet A 179:2517–2531. https://doi.org/10.1002/ajmg.a.61368
Kolevzon A, Bush L, Wang AT et al (2014) A pilot controlled trial of insulin-like growth factor-1 in children with Phelan-McDermid syndrome. Mol Autism 5:54. https://doi.org/10.1186/2040-2392-5-54
Komiya Y, Habas R (2008) Wnt signal transduction pathways. Organogenesis 4:68–75. https://doi.org/10.4161/org.4.2.5851
Koopmans T, Eilers R, Menzen M et al (2017) β-catenin directs nuclear factor-κB p65 output via CREB-binding protein/p300 in human airway smooth muscle. Front Immunol. https://doi.org/10.3389/fimmu.2017.01086
Kumar V, Zhang M-X, Swank MW et al (2005) Regulation of dendritic morphogenesis by Ras-PI3K-Akt-mTOR and Ras-MAPK signaling pathways. J Neurosci 25:11288–11299. https://doi.org/10.1523/JNEUROSCI.2284-05.2005
Kumar S, Reynolds K, Ji Y et al (2019) Impaired neurodevelopmental pathways in autism spectrum disorder: a review of signaling mechanisms and crosstalk. J Neurodev Disord 11:1–14. https://doi.org/10.1186/s11689-019-9268-y
Kumawat K, Menzen MH, Bos IST et al (2013) Noncanonical WNT-5A signaling regulates TGF-β-induced extracellular matrix production by airway smooth muscle cells. FASEB J Off Publ Fed Am Soc Exp Biol 27:1631–1643. https://doi.org/10.1096/fj.12-217539
Kwan V, Unda BK, Singh KK (2016) Wnt signaling networks in autism spectrum disorder and intellectual disability. J Neurodev Disord 8:1–10. https://doi.org/10.1186/s11689-016-9176-3
Lai M-C, Lombardo MV, Baron-Cohen S (2014) Autism. Lancet 383:896–910. https://doi.org/10.1016/S0140-6736(13)61539-1
Lai X, Wu X, Hou N et al (2018) Vitamin A deficiency induces autistic-like behaviors in rats by regulating the RARβ-CD38-oxytocin axis in the hypothalamus. Mol Nutr Food Res 62:1700754. https://doi.org/10.1002/mnfr.201700754
Lawrence T (2009) The nuclear factor NF-kappaB pathway in inflammation. Cold Spring Harb Perspect Biol 1:a001651–a001651. https://doi.org/10.1101/cshperspect.a001651
Lee RWY, Tierney E (2011) Hypothesis: the role of sterols in autism spectrum disorder. Autism Res Treat 2011:653570. https://doi.org/10.1155/2011/653570
Lee Y, Kim H, Kim J-E et al (2018) Excessive D1 dopamine receptor activation in the dorsal striatum promotes autistic-like behaviors. Mol Neurobiol 55:5658–5671. https://doi.org/10.1007/s12035-017-0770-5
Li Q, Verma IM (2002) NF-κB regulation in the immune system. Nat Rev Immunol 2:725–734. https://doi.org/10.1038/nri910
Li X, Deng W, Lobo-Ruppert SM, Ruppert JM (2007) Gli1 acts through snail and E-cadherin to promote nuclear signaling by β-catenin. Oncogene 26:4489–4498. https://doi.org/10.1038/sj.onc.1210241
Li X, Chauhan A, Sheikh AM et al (2009) Elevated immune response in the brain of autistic patients. J Neuroimmunol 207:111–116. https://doi.org/10.1016/j.jneuroim.2008.12.002
Li M, Shin J, Risgaard RD et al (2020) Identification of FMR1-regulated molecular networks in human neurodevelopment. Genome Res 30:361–374. https://doi.org/10.1101/gr.251405.119
Liu J, Pan S, Hsieh MH et al (2013) Targeting Wnt-driven cancer through the inhibition of porcupine by LGK974. Proc Natl Acad Sci USA 110:20224–20229. https://doi.org/10.1073/pnas.1314239110
Long JM, LaPorte P, Paylor R, Wynshaw-Boris A (2004) Expanded characterization of the social interaction abnormalities in mice lacking Dvl1. Genes Brain Behav 3:51–62. https://doi.org/10.1046/j.1601-183x.2003.00045.x
Lord C, Elsabbagh M, Baird G, Veenstra-Vanderweele J (2018) Autism spectrum disorder. Lancet (London, England) 392:508–520. https://doi.org/10.1016/S0140-6736(18)31129-2
Lu Z, Xu S (2006) ERK1/2 MAP kinases in cell survival and apoptosis. IUBMB Life 58:621–631. https://doi.org/10.1080/15216540600957438
Ma B, Hottiger MO (2016) Crosstalk between wnt/β-catenin and NF-κB signaling pathway during inflammation. Front Immunol. https://doi.org/10.3389/fimmu.2016.00378
Madden KS, Sanders VM, Felten DL (1995) Catecholamine influences and sympathetic neural modulation of immune responsiveness. Annu Rev Pharmacol Toxicol 35:417–448. https://doi.org/10.1146/annurev.pa.35.040195.002221
Madrid LV, Mayo MW, Reuther JY, Baldwin AS (2001) Akt stimulates the transactivation potential of the RelA/p65 subunit of NF-κB through utilization of the IκB kinase and activation of the mitogen-activated protein kinase p38. J Biol Chem 276:18934–18940. https://doi.org/10.1074/jbc.M101103200
Maes M, Anderson G, Betancort Medina SR et al (2019) Integrating autism spectrum disorder pathophysiology: mitochondria, vitamin A, CD38, oxytocin, serotonin and melatonergic alterations in the placenta and gut. Curr Pharm Des 25:4405–4420. https://doi.org/10.2174/1381612825666191102165459
Maestroni GJM (2006) Sympathetic nervous system influence on the innate immune response. Ann N Y Acad Sci 1069:195–207. https://doi.org/10.1196/annals.1351.017
Magnuson B, Ekim B, Fingar DC (2011) Regulation and function of ribosomal protein S6 kinase (S6K) within mTOR signalling networks. Biochem J 441:1–21. https://doi.org/10.1042/BJ20110892
Malek H, Ebadzadeh MM, Safabakhsh R et al (2015) Dynamics of the HPA axis and inflammatory cytokines: insights from mathematical modeling. Comput Biol Med 67:1–12. https://doi.org/10.1016/j.compbiomed.2015.09.018
Malik M, Sheikh AM, Wen G et al (2011a) Expression of inflammatory cytokines, Bcl2 and cathepsin D are altered in lymphoblasts of autistic subjects. Immunobiology 216:80–85. https://doi.org/10.1016/j.imbio.2010.03.001
Malik M, Tauqeer Z, Sheikh AM et al (2011b) NF-κB signaling in the brain of autistic subjects. Mediat Inflamm 2011:785265. https://doi.org/10.1155/2011/785265
Markham A (2017) Copanlisib: first global approval. Drugs 77:2057–2062. https://doi.org/10.1007/s40265-017-0838-6
Maximo JO, Cadena EJ, Kana RK (2014) The implications of brain connectivity in the neuropsychology of autism. Neuropsychol Rev 24:16–31. https://doi.org/10.1007/s11065-014-9250-0
McCubrey JA, May WS, Duronio V, Mufson A (2000) Serine/threonine phosphorylation in cytokine signal transduction. Leukemia 14:9–21. https://doi.org/10.1038/sj.leu.2401657
McDuffie A, Thurman AJ, Hagerman RJ, Abbeduto L (2015) Symptoms of autism in males with fragile X syndrome: a comparison to nonsyndromic ASD using current ADI-R scores. J Autism Dev Disord 45:1925–1937. https://doi.org/10.1007/s10803-013-2013-6
Mefford H, Sharp A, Baker C et al (2008) Recurrent rearrangements of chromosome 1q21.1 and variable pediatric phenotypes. N Engl J Med 359:1685–1699. https://doi.org/10.1056/NEJMoa0805384
Mendoza MC, Er EE, Blenis J (2011) The Ras-ERK and PI3K-mTOR pathways: cross-talk and compensation. Trends Biochem Sci 36:320–328. https://doi.org/10.1016/j.tibs.2011.03.006
Mila M, Alvarez-Mora MI, Madrigal I, Rodriguez-Revenga L (2018) Fragile X syndrome: an overview and update of the FMR1 gene. Clin Genet 93:197–205. https://doi.org/10.1111/cge.13075
Moreno-Ramos OA, Olivares AM, Haider NB et al (2015) Whole-exome sequencing in a south american cohort links ALDH1A3, FOXN1 and retinoic acid regulation pathways to autism spectrum disorders. PLoS ONE 10:e0135927. https://doi.org/10.1371/journal.pone.0135927
Morin PJ (1999) Β-catenin signaling and cancer. BioEssays 21:1021–1030. https://doi.org/10.1002/(SICI)1521-1878(199912)22:1%3c1021:AID-BIES6%3e3.0.CO;2-P
Mousa A, Bakhiet M (2013) Role of cytokine signaling during nervous system development. Int J Mol Sci 14:13931–13957. https://doi.org/10.3390/ijms140713931
Nadeem A, Ahmad SF, Attia SM et al (2018) Activation of IL-17 receptor leads to increased oxidative inflammation in peripheral monocytes of autistic children. Brain Behav Immunol 67:335–344. https://doi.org/10.1016/j.bbi.2017.09.010
Nagatomi R, Kaifu T, Okutsu M et al (2000) Modulation of the immune system by the autonomic nervous system and its implication in immunological changes after training. Exerc Immunol Rev 6:54–74
Naik US, Gangadharan C, Abbagani K et al (2011) A study of nuclear transcription factor-kappa B in childhood autism. PLoS ONE 6:e19488–e19488. https://doi.org/10.1371/journal.pone.0019488
Neul JL (2012) The relationship of Rett syndrome and MECP2 disorders to autism. Dialog Clin Neurosci 14:253–262
Nevison C, Blaxill M, Zahorodny W (2018) California autism prevalence trends from 1931 to 2014 and comparison to national ASD data from IDEA and ADDM. J Autism Dev Disord 48:4103–4117. https://doi.org/10.1007/s10803-018-3670-2
Nguyen A, Rauch TA, Pfeifer GP, Hu VW (2010) Global methylation profiling of lymphoblastoid cell lines reveals epigenetic contributions to autism spectrum disorders and a novel autism candidate gene, RORA, whose protein product is reduced in autistic brain. FASEB J Off Publ Fed Am Soc Exp Biol 24:3036–3051. https://doi.org/10.1096/fj.10-154484
Niehrs C (2012) The complex world of WNT receptor signalling. Nat Rev Mol Cell Biol 13:767–779. https://doi.org/10.1038/nrm3470
Nightingale S (2012) Autism spectrum disorders. Nat Rev Drug Discov 11:745–746. https://doi.org/10.1016/B978-0-12-800685-6.00016-3
Nikoletopoulou V, Sidiropoulou K, Kallergi E et al (2017) Modulation of autophagy by BDNF underlies synaptic plasticity. Cell Metab 26:230–242.e5. https://doi.org/10.1016/j.cmet.2017.06.005
Niu M, Han Y, Dy ABC et al (2017) Autism symptoms in fragile X syndrome. J Child Neurol 32:903–909. https://doi.org/10.1177/0883073817712875
Niyaz M, Khan MS, Mudassar S (2019) Hedgehog signaling: an achilles’ heel in cancer. Transl Oncol 12:1334–1344. https://doi.org/10.1016/j.tranon.2019.07.004
Ohja K, Gozal E, Fahnestock M et al (2018) Neuroimmunologic and neurotrophic interactions in autism spectrum disorders: relationship to neuroinflammation. Neuromol Med 20:161–173. https://doi.org/10.1007/s12017-018-8488-8
Okada K, Hashimoto K, Iwata Y et al (2007) Decreased serum levels of transforming growth factor-beta1 in patients with autism. Prog Neuropsychopharmacol Biol Psychiatry 31:187–190. https://doi.org/10.1016/j.pnpbp.2006.08.020
Okamoto H, Voleti B, Banasr M et al (2010) Wnt2 expression and signaling is increased by different classes of antidepressant treatments. Biol Psychiatry 68:521–527. https://doi.org/10.1016/j.biopsych.2010.04.023
Oztan O, Garner JP, Partap S et al (2018) Cerebrospinal fluid vasopressin and symptom severity in children with autism. Ann Neurol 84:611–615. https://doi.org/10.1002/ana.25314
Parker-Athill E, Luo D, Bailey A et al (2009) Flavonoids, a prenatal prophylaxis via targeting JAK2/STAT3 signaling to oppose IL-6/MIA associated autism. J Neuroimmunol 217:20–27. https://doi.org/10.1016/j.jneuroim.2009.08.012
Patel SS, Tomar S, Sharma D et al (2017) Targeting sonic hedgehog signaling in neurological disorders. Neurosci Biobehav Rev 74:76–97. https://doi.org/10.1016/j.neubiorev.2017.01.008
Paul A, Edwards J, Pepper C, Mackay S (2018) Inhibitory-κB kinase (IKK) α and nuclear factor-κB (NFκB)-inducing kinase (NIK) as anti-cancer drug targets. Cells 7:176. https://doi.org/10.3390/cells7100176
Paval D (2017) A dopamine hypothesis of autism spectrum disorder. Dev Neurosci 39:355–360. https://doi.org/10.1159/000478725
Peltonen S, Kallionpaa RA, Peltonen J (2017) Neurofibromatosis type 1 (NF1) gene: beyond cafe au lait spots and dermal neurofibromas. Exp Dermatol 26:645–648. https://doi.org/10.1111/exd.13212
Pelullo M, Zema S, Nardozza F et al (2019) Wnt, Notch, and TGF-β pathways impinge on hedgehog signaling complexity: an open window on cancer. Front Genet 10:1–16. https://doi.org/10.3389/fgene.2019.00711
Plata-Salaman CR, Oomura Y, Kai Y (1988) Tumor necrosis factor and interleukin-1 beta: suppression of food intake by direct action in the central nervous system. Brain Res 448:106–114. https://doi.org/10.1016/0006-8993(88)91106-7
Platt RJ, Zhou Y, Slaymaker IM et al (2017) Chd8 mutation leads to autistic-like behaviors and impaired striatal circuits. Cell Rep 19:335–350. https://doi.org/10.1016/j.celrep.2017.03.052
Porta C, Paglino C, Mosca A (2014) Targeting PI3K/Akt/mTOR signaling in cancer. Front Oncol 4:64. https://doi.org/10.3389/fonc.2014.00064
Pucilowska J, Vithayathil J, Pagani M et al (2018) Pharmacological inhibition of ERK signaling rescues pathophysiology and behavioral phenotype associated with 16p11.2 chromosomal deletion in mice. J Neurosci 38:6640–6652. https://doi.org/10.1523/JNEUROSCI.0515-17.2018
Puighermanal E, Biever A, Pascoli V et al (2017) Ribosomal protein S6 phosphorylation is involved in novelty-induced locomotion, synaptic plasticity and mRNA translation. Front Mol Neurosci 10:419. https://doi.org/10.3389/fnmol.2017.00419
Purcell AE, Jeon OH, Zimmerman AW et al (2001) Postmortem brain abnormalities of the glutamate neurotransmitter system in autism. Neurology 57:1618–1628. https://doi.org/10.1212/wnl.57.9.1618
Qiu S, Li Y, Li Y et al (2018) Association between SHANK3 polymorphisms and susceptibility to autism spectrum disorder. Gene 651:100–105. https://doi.org/10.1016/j.gene.2018.01.078
Quattrocki E, Friston K (2014) Autism, oxytocin and interoception. Neurosci Biobehav Rev 47:410–430. https://doi.org/10.1016/j.neubiorev.2014.09.012
Rauen KA (2007) HRAS and the costello syndrome. Clin Genet 71:101–108. https://doi.org/10.1111/j.1399-0004.2007.00743.x
Rauen KA (2013) The RASopathies. Annu Rev Genomics Hum Genet 14:355–369. https://doi.org/10.1146/annurev-genom-091212-153523
Rhinn M, Dolle P (2012) Retinoic acid signalling during development. Development 139:843–858. https://doi.org/10.1242/dev.065938
Riobó NA, Lu K, Ai X et al (2006) Phosphoinositide 3-kinase and Akt are essential for Sonic Hedgehog signaling. Proc Natl Acad Sci USA 103:4505–4510. https://doi.org/10.1073/pnas.0504337103
Roak BJO, Vives L, Fu W et al (2012) Multiplex targeted sequencing identifies recurrently mutated genes in autism spectrum disorders. Science 23:1619–1623
Rodriguez JI, Kern JK (2011) Evidence of microglial activation in autism and its possible role in brain underconnectivity. Neuron Glia Biol 7:205–213. https://doi.org/10.1017/S1740925X12000142
Rodriguez-Martinez G, Velasco I (2012) Activin and TGF-beta effects on brain development and neural stem cells. CNS Neurol Disord Drug Targets 11:844–855. https://doi.org/10.2174/1871527311201070844
Rubenstein JLR, Merzenich MM (2003) Model of autism: increased ratio of excitation/inhibition in key neural systems. Genes Brain Behav 2:255–267. https://doi.org/10.1034/j.1601-183x.2003.00037.x
Russo FB, Brito A, de Freitas AM et al (2019) The use of iPSC technology for modeling autism spectrum disorders. Neurobiol Dis 130:104483. https://doi.org/10.1016/j.nbd.2019.104483
Sancheti H, Akopian G, Yin F et al (2013) Age-dependent modulation of synaptic plasticity and insulin mimetic effect of lipoic acid on a mouse model of Alzheimer’s disease. PLoS ONE 8:e69830. https://doi.org/10.1371/journal.pone.0069830
Sánchez-Alegría K, Flores-León M, Avila-Muñoz E et al (2018) PI3K signaling in neurons: a central node for the control of multiple functions. Int J Mol Sci 19:3725. https://doi.org/10.3390/ijms19123725
Sanders SJ, Murtha MT, Gupta AR et al (2012) De novo mutations revealed by whole-exome sequencing are strongly associated with autism. Nature 485:237–241. https://doi.org/10.1038/nature10945
Sawicka K, Zukin RS (2012) Dysregulation of mTOR signaling in neuropsychiatric disorders: therapeutic implications. Neuropsychopharmacol Off Publ Am Coll Neuropsychopharmacol 37:305–306. https://doi.org/10.1038/npp.2011.210
Sayad A, Noroozi R, Omrani MD et al (2017) Retinoic acid-related orphan receptor alpha (RORA) variants are associated with autism spectrum disorder. Metab Brain Dis 32:1595–1601. https://doi.org/10.1007/s11011-017-0049-6
Schneider T, Przewłocki R (2005) Behavioral alterations in rats prenatally to valproic acid: animal model of autism. Neuropsychopharmacology 30:80–89. https://doi.org/10.1038/sj.npp.1300518
Senftleben U, Karin M (2002) The IKK/NF-kappaB pathway. Crit Care Med 30:S18–S26
Shah RR, Bird AP (2017) MeCP2 mutations: progress towards understanding and treating Rett syndrome. Genome Med 9:17. https://doi.org/10.1186/s13073-017-0411-7
Shah S, Molinaro G, Liu B et al (2020) FMRP control of ribosome translocation promotes chromatin modifications and alternative splicing of neuronal genes linked to autism. Cell Rep 30:4459–4472.e6. https://doi.org/10.1016/j.celrep.2020.02.076
Shcheglovitov A, Shcheglovitova O, Yazawa M et al (2013) SHANK3 and IGF1 restore synaptic deficits in neurons from 22q13 deletion syndrome patients. Nature 503:267–271. https://doi.org/10.1038/nature12618
Shi Z-M, Han Y-W, Han X-H et al (2016) Upstream regulators and downstream effectors of NF-kappaB in Alzheimer’s disease. J Neurol Sci 366:127–134. https://doi.org/10.1016/j.jns.2016.05.022
Sikora DM, Pettit-Kekel K, Penfield J et al (2006) The near universal presence of autism spectrum disorders in children with Smith-Lemli-Opitz syndrome. Am J Med Genet A 140:1511–1518. https://doi.org/10.1002/ajmg.a.31294
Simonoff E, Pickles A, Charman T et al (2008) Psychiatric disorders in children with autism spectrum disorders: prevalence, comorbidity, and associated factors in a population-derived sample. J Am Acad Child Adolesc Psychiatry 47:921–929. https://doi.org/10.1097/CHI.0b013e318179964f
Singh VK (1996) Plasma increase of interleukin-12 and interferon-gamma Pathological significance in autism. J Neuroimmunol 66:143–145. https://doi.org/10.1016/0165-5728(96)00014-8
Singh R, Dhanyamraju PK, Lauth M (2017) DYRK1B blocks canonical and promotes non-canonical Hedgehog signaling through activation of the mTOR/AKT pathway. Oncotarget 8:833–845. https://doi.org/10.18632/oncotarget.13662
Siveen KS, Nguyen AH, Lee JH et al (2014) Negative regulation of signal transducer and activator of transcription-3 signalling cascade by lupeol inhibits growth and induces apoptosis in hepatocellular carcinoma cells. Br J Cancer 111:1327–1337. https://doi.org/10.1038/bjc.2014.422
Sizemore N, Leung S, Stark GR (1999) Activation of phosphatidylinositol 3-Kinase in response to interleukin-1 leads to phosphorylation and activation of the NF-κB p65/RelA subunit. Mol Cell Biol 19:4798–4805. https://doi.org/10.1128/mcb.19.7.4798
Song L, Li ZY, Liu WP, Zhao MR (2015) Crosstalk between Wnt/β-catenin and Hedgehog/Gli signaling pathways in colon cancer and implications for therapy. Cancer Biol Ther 16:1–7. https://doi.org/10.4161/15384047.2014.972215
Sousa KM, Carlos Villaescusa J, Cajanek L et al (2010) Wnt2 regulates progenitor proliferation in the developing ventral midbrain. J Biol Chem 285:7246–7253. https://doi.org/10.1074/jbc.M109.079822
Spanjer AIR, Baarsma HA, Oostenbrink LM et al (2016) TGF-β-induced profibrotic signaling is regulated in part by the WNT receptor Frizzled-8. FASEB J Off Publ Fed Am Soc Exp Biol 30:1823–1835. https://doi.org/10.1096/fj.201500129
Stanton BZ, Peng LF (2010) Small-molecule modulators of the Sonic Hedgehog signaling pathway. Mol Biosyst 6:44–54. https://doi.org/10.1039/b910196a
Stefanatos GA (2008) Regression in autistic spectrum disorders. Neuropsychol Rev 18:305–319. https://doi.org/10.1007/s11065-008-9073-y
Subramanian M, Timmerman CK, Schwartz JL et al (2015) Characterizing autism spectrum disorders by key biochemical pathways. Front Neurosci 9:1–18. https://doi.org/10.3389/fnins.2015.00313
Sun S-C (2017) The non-canonical NF-kappaB pathway in immunity and inflammation. Nat Rev Immunol 17:545–558. https://doi.org/10.1038/nri.2017.52
Sun J, Nan G (2017) The extracellular signal-regulated kinase 1/2 pathway in neurological diseases: a potential therapeutic target (review). Int J Mol Med 39:1338–1346. https://doi.org/10.3892/ijmm.2017.2962
Suzuki K, Matsuzaki H, Iwata K et al (2011) Plasma cytokine profiles in subjects with high-functioning autism spectrum disorders. PLoS ONE 6:e20470. https://doi.org/10.1371/journal.pone.0020470
Sweetman DU, O’Donnell SM, Lalor A et al (2019) Zinc and vitamin A deficiency in a cohort of children with autism spectrum disorder. Child Care Health Dev 45:380–386. https://doi.org/10.1111/cch.12655
Sztainberg Y, Zoghbi HY (2016) Lessons learned from studying syndromic autism spectrum disorders. Nat Neurosci 19:1408–1418. https://doi.org/10.1038/nn.4420
Tergaonkar V, Correa RG, Ikawa M, Verma IM (2005) Distinct roles of IκB proteins in regulating constitutive NF-κB activity. Nat Cell Biol 7:921–923. https://doi.org/10.1038/ncb1296
Theoharides TC, Tsilioni I, Patel AB, Doyle R (2016) Atopic diseases and inflammation of the brain in the pathogenesis of autism spectrum disorders. Transl Psychiatry 6:e844–e844. https://doi.org/10.1038/tp.2016.77
Thomas GM, Huganir RL (2004) MAPK cascade signalling and synaptic plasticity. Nat Rev Neurosci 5:173–183. https://doi.org/10.1038/nrn1346
Tilot AK, Frazier TW 2nd, Eng C (2015) Balancing proliferation and connectivity in pten-associated autism spectrum disorder. Neurotherapeutics 12:609–619. https://doi.org/10.1007/s13311-015-0356-8
Trifonova EA, Klimenko AI, Mustafin ZS et al (2019) The mTOR signaling pathway activity and vitamin D availability control the expression of most autism predisposition genes. Int J Mol Sci. https://doi.org/10.3390/ijms20246332
Tsai PT, Hull C, Chu Y et al (2012) Autistic-like behaviour and cerebellar dysfunction in Purkinje cell Tsc1 mutant mice. Nature 488:647–651. https://doi.org/10.1038/nature11310
van der Poest CE, Jansen FE, Braun KPJ, Peters JM (2020) Update on drug management of refractory epilepsy in tuberous sclerosis complex. Pediatr Drugs 22:73–84. https://doi.org/10.1007/s40272-019-00376-0
Varela-Nallar L, Inestrosa NC (2013) Wnt signaling in the regulation of adult hippocampal neurogenesis. Front Cell Neurosci 7:1–11. https://doi.org/10.3389/fncel.2013.00100
Veeraragavan S, Wan Y-W, Connolly DR et al (2016) Loss of MeCP2 in the rat models regression, impaired sociability and transcriptional deficits of Rett syndrome. Hum Mol Genet 25:3284–3302. https://doi.org/10.1093/hmg/ddw178
Vithayathil J, Pucilowska J, Landreth GE (2018) ERK/MAPK signaling and autism spectrum disorders. Prog Brain Res 241:63–112. https://doi.org/10.1016/bs.pbr.2018.09.008
Volkmar F, Chawarska K, Klin A (2005) Autism in infancy and early childhood. Annu Rev Psychol 56:315–336. https://doi.org/10.1146/annurev.psych.56.091103.070159
Waltes R, Gfesser J, Haslinger D et al (2014) Common EIF4E variants modulate risk for autism spectrum disorders in the high-functioning range. J Neural Transm 121:1107–1116. https://doi.org/10.1007/s00702-014-1230-2
Wang Z, Xu L, Zhu X et al (2010) Demethylation of specific Wnt/β-catenin pathway genes and its upregulation in rat brain induced by prenatal valproate exposure. Anat Rec 293:1947–1953. https://doi.org/10.1002/ar.21232
Wang Y, Ding Q, Yen C-J et al (2012) The crosstalk of mTOR/S6K1 and Hedgehog pathways. Cancer Cell 21:374–387. https://doi.org/10.1016/j.ccr.2011.12.028
Wang Y, Billon C, Walker JK, Burris TP (2016) Therapeutic effect of a synthetic RORalpha/gamma agonist in an animal model of autism. ACS Chem Neurosci 7:143–148. https://doi.org/10.1021/acschemneuro.5b00159
Wei H, Zou H, Sheikh AM et al (2011) IL-6 is increased in the cerebellum of autistic brain and alters neural cell adhesion, migration and synaptic formation. J Neuroinflamm 8:52. https://doi.org/10.1186/1742-2094-8-52
Wen Y, Alshikho MJ, Herbert MR (2016) Pathway Network analyses for autism reveal multisystem involvement, major overlaps with other diseases and convergence upon MAPK and calcium signaling. PLoS ONE 11:e0153329. https://doi.org/10.1371/journal.pone.0153329
Wennerberg K, Rossman KL, Der CJ (2005) The Ras superfamily at a glance. J Cell Sci 118:843–846. https://doi.org/10.1242/jcs.01660
Wexler EM, Geschwind DH, Palmer TD (2008) Lithium regulates adult hippocampal progenitor development through canonical Wnt pathway activation. Mol Psychiatry 13:285–292. https://doi.org/10.1038/sj.mp.4002093
Whitmarsh AJ (2007) Regulation of gene transcription by mitogen-activated protein kinase signaling pathways. Biochim Biophys Acta Mol Cell Res 1773:1285–1298. https://doi.org/10.1016/j.bbamcr.2006.11.011
Woiciechowsky C, Asadullah K, Nestler D et al (1998) Sympathetic activation triggers systemic interleukin-10 release in immunodepression induced by brain injury. Nat Med 4:808–813. https://doi.org/10.1038/nm0798-808
Wyss-Coray T, Borrow P, Brooker MJ, Mucke L (1997) Astroglial overproduction of TGF-beta 1 enhances inflammatory central nervous system disease in transgenic mice. J Neuroimmunol 77:45–50. https://doi.org/10.1016/s0165-5728(97)00049-0
Xu H, Zhuang X (2019) Atypical antipsychotics-induced metabolic syndrome and nonalcoholic fatty liver disease: a critical review. Neuropsychiatr Dis Treat 15:2087–2099. https://doi.org/10.2147/NDT.S208061
Xu N, Li X, Zhong Y (2015) Inflammatory cytokines: potential biomarkers of immunologic dysfunction in autism spectrum disorders. Mediators Inflamm 2015:531518. https://doi.org/10.1155/2015/531518
Xu Q, Liu Y-Y, Wang X et al (2018a) Autism-associated CHD8 deficiency impairs axon development and migration of cortical neurons. Mol Autism 9:65. https://doi.org/10.1186/s13229-018-0244-2
Xu X, Li C, Gao X et al (2018b) Excessive UBE3A dosage impairs retinoic acid signaling and synaptic plasticity in autism spectrum disorders. Cell Res 28:48–68. https://doi.org/10.1038/cr.2017.132
Ya W (2017) Involvement of calcium, Ras, MAPK, PI3K-Akt and mTOR signaling pathways in autism spectrum disorders. Neurol Neurother Open Access J. https://doi.org/10.23880/NNOAJ-16000110
Yabut OR, Pleasure SJ (2018) Sonic Hedgehog signaling rises to the surface: emerging roles in neocortical development. Brain Plast (Amsterdam, Netherlands) 3:119–128. https://doi.org/10.3233/BPL-180064
Young AMH, Campbell E, Lynch S et al (2011) Aberrant NF-kappaB expression in autism spectrum condition: a mechanism for neuroinflammation. Front Psychiatry 2:27. https://doi.org/10.3389/fpsyt.2011.00027
Yu JSL, Cui W (2016) Proliferation, survival and metabolism: the role of PI3K/AKT/mTOR signalling in pluripotency and cell fate determination. Development 143:3050–3060. https://doi.org/10.1242/dev.137075
Zhan T, Rindtorff N, Boutros M (2017) Wnt signaling in cancer. Oncogene 36:1461–1473. https://doi.org/10.1038/onc.2016.304
Zhang Y, Hu W (2012) NFκB signaling regulates embryonic and adult neurogenesis. Front Biol (Beijing). https://doi.org/10.1007/s11515-012-1233-z
Zhang Y, Pizzute T, Pei M (2014) A review of crosstalk between MAPK and Wnt signals and its impact on cartilage regeneration. Cell Tissue Res 358:633–649. https://doi.org/10.1007/s00441-014-2010-x
Zhang Q, Lenardo MJ, Baltimore D (2017a) 30 years of NF-κB: a blossoming of relevance to human pathobiology. Cell 168:37–57. https://doi.org/10.1016/j.cell.2016.12.012
Zhang X, He X, Li Q et al (2017b) PI3K/AKT/mTOR signaling mediates valproic acid-induced neuronal differentiation of neural stem cells through epigenetic modifications. Stem cell reports 8:1256–1269. https://doi.org/10.1016/j.stemcr.2017.04.006
Zhou W, Li S (2018) Decreased levels of serum retinoic acid in chinese children with autism spectrum disorder. Psychiatry Res 269:469–473. https://doi.org/10.1016/j.psychres.2018.08.091
Zhou L, An N, Haydon RC et al (2003) Tyrosine kinase inhibitor STI-571/Gleevec down-regulates the beta-catenin signaling activity. Cancer Lett 193:161–170. https://doi.org/10.1016/s0304-3835(03)00013-2
Acknowledgements
We are grateful to the Department of Biochemistry of the Institute of Chemistry (IQ) of the University of São Paulo (USP, São Paulo, Brazil) for the academic support and opportunity to write this review.
Funding
R.G.C. was supported by a Visiting Professor grant (Edital 02/2019, PrInt USP/CAPES) from University of São Paulo (USP) and CAPES (Federal Agency for Superior Education and Training), Brazil.
Author information
Authors and Affiliations
Contributions
Manuscript conceptualization: RGC. Drafting the text: JB, GD, MCSB, RBA, RRA, ALPA, DP. Preparing figures: JB, GD, MCSB. Manuscript editing and formatting: JB, GD, MCS, RGC, HU. Manuscript revision: RGC, MCS, HU.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Baranova, J., Dragunas, G., Botellho, M.C.S. et al. Autism Spectrum Disorder: Signaling Pathways and Prospective Therapeutic Targets. Cell Mol Neurobiol 41, 619–649 (2021). https://doi.org/10.1007/s10571-020-00882-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-020-00882-7