Abstract
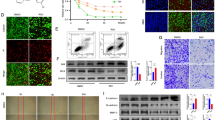
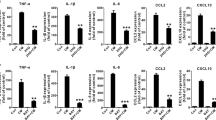
Activation of inflammasome leads to the formation of an inflammatory microenvironment which plays an important role in the process of cancer development. Beta-hydroxybutyrate (BHB) is a ketone body that has recently been reported to exert anti-inflammatory effects via inhibition of NOD-like receptor pyrin domain-containing 3 (NLRP3) inflammasome. Here, we investigated the potential influence of BHB on the in vitro migration of C6 glioma cells and the activation of NLRP3 inflammasome. Our results indicated that administration of BHB suppressed C6 cells migration and NLRP3 inflammasome activation, reducing the levels of activated cysteinyl aspartate-specific proteinase 1 (caspase-1) and mature Interleukin 1β (IL-1β). Fully activation of NLRP3 inflammasome was induced by lipopolysaccharide (LPS) prime plus adenosine triphosphate (ATP) stimulation in C6 cells, which promoted in vitro migration of C6 cell. BHB also counteracted the LPS/ATP-promoted cell migration by suppressing the activation of caspase-1 and the maturation of IL-1β. The enhancement of phospho-signal transducer and activator of transcription 3 (p-STAT3), degradation of nuclear factor of kappa light polypeptide gene enhancer in B-cells inhibitor, alpha (IκBα) as well as the overexpression of fibroblast growth factor 2 (FGF2) resulting from LPS/ATP treatment, and subsequent IL-1β maturation could also be compensated by BHB. Our results suggested that BHB inhibits the activation of NLRP3 inflammasome in C6 glioma cells and consequently suppressed the C6 cell migration. These findings also implicated that by inhibiting NLRP3 inflammasome, BHB reduced the inflammatory microenvironment which provided ancillary therapeutic benefits for the intervention of glioma.







Similar content being viewed by others
References
Ahmed K, Tunaru S, Offermanns S (2009) GPR109A, GPR109B and GPR81, a family of hydroxy-carboxylic acid receptors. Trends Pharmacol Sci 30:557–562. https://doi.org/10.1016/j.tips.2009.09.001
Bauernfeind FG et al (2009) Cutting edge: NF-kappaB activating pattern recognition and cytokine receptors license NLRP3 inflammasome activation by regulating NLRP3 expression. J Immunol 183:787–791. https://doi.org/10.4049/jimmunol.0901363
Bauernfeind F, Ablasser A, Bartok E, Kim S, Schmid-Burgk J, Cavlar T, Hornung V (2011) Inflammasomes: current understanding and open questions. Cell Mol Life Sci 68:765–783. https://doi.org/10.1007/s00018-010-0567-4
Caldera V, Mellai M, Annovazzi L, Valente G, Tessitore L, Schiffer D (2008) Stat3 expression and its correlation with proliferation and apoptosis/autophagy in gliomas. J Oncol 2008:219241. https://doi.org/10.1155/2008/219241
Fathima Hurmath K, Ramaswamy P, Nandakumar DN (2014) IL-1beta microenvironment promotes proliferation, migration, and invasion of human glioma cells. Cell Biol Int 38:1415–1422. https://doi.org/10.1002/cbin.10353
Fu SP et al (2014) BHBA suppresses LPS-induced inflammation in BV-2 cells by inhibiting NF-kappaB activation. Mediators Inflamm 2014:983401. https://doi.org/10.1155/2014/983401
Fu SP et al (2015) Anti-inflammatory effects of BHBA in both in vivo and in vitro Parkinson’s disease models are mediated by GPR109A-dependent mechanisms. J Neuroinflammation 12:9. https://doi.org/10.1186/s12974-014-0230-3
Goldberg EL et al (2017) beta-Hydroxybutyrate Deactivates Neutrophil NLRP3 Inflammasome to Relieve Gout Flares. Cell Rep 18:2077–2087. https://doi.org/10.1016/j.celrep.2017.02.004
Itokazu Y, Pagano RE, Schroeder AS, O’Grady SM, Limper AH, Marks DL (2014) Reduced GM1 ganglioside in CFTR-deficient human airway cells results in decreased beta1-integrin signaling and delayed wound repair. Am J Physiol Cell Physiol 306:C819–C830. https://doi.org/10.1152/ajpcell.00168.2013
Kahlenberg JM, Lundberg KC, Kertesy SB, Qu Y, Dubyak GR (2005) Potentiation of caspase-1 activation by the P2 × 7 receptor is dependent on TLR signals and requires NF-kappaB-driven protein synthesis. J Immunol 175:7611–7622. https://doi.org/10.4049/jimmunol.175.11.7611
Kolb R, Liu GH, Janowski AM, Sutterwala FS, Zhang W (2014) Inflammasomes in cancer: a double-edged sword. Protein Cell 5:12–20. https://doi.org/10.1007/s13238-013-0001-4
Lee JG, Heur M (2013) Interleukin-1beta enhances cell migration through AP-1 and NF-kappaB pathway-dependent FGF2 expression in human corneal endothelial cells. Biol Cell 105:175–189. https://doi.org/10.1111/boc.201200077
Li L, Liu Y (2015) Aging-related gene signature regulated by Nlrp3 predicts glioma progression. Am J Cancer Res 5:442–449 (can’t find DOI number)
Mrugala MM (2013) Advances and challenges in the treatment of glioblastoma: a clinician’s perspective. Discov Med 15:221–230
Newman JC, Verdin E (2014a) beta-hydroxybutyrate: much more than a metabolite. Diab Res Clin Pract 106:173–181. https://doi.org/10.1016/j.diabres.2014.08.009
Newman JC, Verdin E (2014b) Ketone bodies as signaling metabolites. Trends Endocrinol Metab 25:42–52. https://doi.org/10.1016/j.tem.2013.09.002
Okamoto M et al (2010) Constitutively active inflammasome in human melanoma cells mediating autoinflammation via caspase-1 processing and secretion of interleukin-1beta. J Biol Chem 285:6477–6488. https://doi.org/10.1074/jbc.M109.064907
Qaddoumi I, Sultan I, Gajjar A (2009) Outcome and prognostic features in pediatric gliomas: a review of 6212 cases from the Surveillance, Epidemiology, and End Results database. Cancer 115:5761–5770. https://doi.org/10.1002/cncr.24663
Rahman M et al (2014) The beta-hydroxybutyrate receptor HCA2 activates a neuroprotective subset of macrophages. Nature Commun 5:3944. https://doi.org/10.1038/ncomms4944
Rossi A, Woolf E, Brooks S, Fairres K, Scheck MC A (2015) Abstract 3346: the ketone body β-hydroxybutyrate increases radiosensitivity in glioma cell lines in vitro. Cancer Res 75:3346. https://doi.org/10.1158/1538-7445.AM2015-3346
Saxena S, Jha S (2017) Role of NOD- like Receptors in Glioma Angiogenesis: Insights into future therapeutic interventions. Cytokine Growth Factor Rev 34:15–26. https://doi.org/10.1016/j.cytogfr.2017.02.001
Schroder K, Tschopp J (2010) The inflammasomes. Cell 140:821–832. https://doi.org/10.1016/j.cell.2010.01.040
Shimazu T et al (2013) Suppression of oxidative stress by beta-hydroxybutyrate, an endogenous histone deacetylase inhibitor. Science 339:211–214. https://doi.org/10.1126/science.1227166
Shu M et al (2011) Targeting oncogenic miR-335 inhibits growth and invasion of malignant astrocytoma cells. Mol Cancer 10:59. https://doi.org/10.1186/1476-4598-10-59
Szebenyi G, Fallon JF (1999) Fibroblast growth factors as multifunctional signaling factors. Int Rev Cytol 185:45–106
Tarassishin L, Casper D, Lee SC (2014a) Aberrant expression of interleukin-1β and inflammasome activation in human malignant gliomast. PLoS ONE 9:e103432. https://doi.org/10.1371/journal.pone.0103432
Tarassishin L, Lim J, Weatherly DB, Angeletti RH, Lee SC (2014b) Interleukin-1-induced changes in the glioblastoma secretome suggest its role in tumor progression. J Proteomics 99:152–168. https://doi.org/10.1016/j.jprot.2014.01.024
Tieu K et al (2003) D-β-Hydroxybutyrate rescues mitochondrial respiration and mitigates features of Parkinson disease. J Clin Investig 112:892–901. https://doi.org/10.1172/jci200318797
Wang Y et al (2016) Activation of NLRP3 inflammasome enhances the proliferation and migration of A549 lung cancer cells. Oncol Rep 35:2053–2064. https://doi.org/10.3892/or.2016.4569
Won YJ, Lu VB, Puhl HL, Ikeda SR (2013) beta-Hydroxybutyrate modulates N-type calcium channels in rat sympathetic neurons by acting as an agonist for the G-protein-coupled receptor FFA3. J Neurosci 33:19314–19325. https://doi.org/10.1523/JNEUROSCI.3102-13.2013
Woolf EC, Syed N, Scheck AC (2016) Tumor Metabolism, the Ketogenic Diet and beta-Hydroxybutyrate: Novel Approaches to Adjuvant Brain Tumor Therapy. Front Mol Neurosci 9:122. https://doi.org/10.3389/fnmol.2016.00122
Xie J et al (2009) Anesthetic pentobarbital inhibits proliferation and migration of malignant glioma cells. Cancer Lett 282:35–42. https://doi.org/10.1016/j.canlet.2009.02.055
Yamanashi T et al (2017) Beta-hydroxybutyrate, an endogenic NLRP3 inflammasome inhibitor, attenuates stress-induced behavioral and inflammatory responses. Sci Rep 7:7677. https://doi.org/10.1038/s41598-017-08055-1
Youm YH et al (2015) The ketone metabolite beta-hydroxybutyrate blocks NLRP3 inflammasome-mediated inflammatory disease. Nat Med 21:263–269. https://doi.org/10.1038/nm.3804
Zhang Y, Kong W, Jiang J (2017) Prevention and treatment of cancer targeting chronic inflammation: research progress, potential agents, clinical studies and mechanisms. Sci China Life Sci 60:601–616. https://doi.org/10.1007/s11427-017-9047-4
Zhiyu W et al (2016) The inflammasome: an emerging therapeutic oncotarget for cancer prevention. Oncotarget 7:50766–50780. https://doi.org/10.18632/oncotarget.9391
Zhou K, Shi L, Wang Y, Chen S, Zhang J (2016) Recent Advances of the NLRP3 Inflammasome in Central Nervous System Disorders. J Immunol Res 2016:9238290. https://doi.org/10.1155/2016/9238290
Zitvogel L, Kepp O, Galluzzi L, Kroemer G (2012) Inflammasomes in carcinogenesis and anticancer immune responses. Nat Immunol 13:343–351. https://doi.org/10.1038/ni.2224
Acknowledgements
This work was supported by the National Natural Science Foundation of China (Nos. 81371288, 81172170).
Author information
Authors and Affiliations
Contributions
Conceived and designed the experiments: LX, LH, and SS. Performed the experiments: SS and WL. Analyzed the data: SS and ZY. All authors read and approved the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests. We declare that we do not have any commercial or associative interest that represents a conflict of interest in connection with the work submitted.
Electronic supplementary material
Below is the link to the electronic supplementary material.
10571_2018_617_MOESM1_ESM.tif
Supplementary Figure 1 BHB inhibits in-vitro cell migration of LN229 and U2251 cell lines. A: U251 cells; B: LN229 cells. Cells were treated with BHB for 24h, cell migration status was analyzed using Transwell chamber migration assay. The representative images were captured by phase-contrast microscope. The number of U251 and LN229 cells migrated into the down chamber were obtained from counting of five microscopic fields (up, down, left, right and middle). ∗P < 0.05, ∗∗P < 0.01, compared with the control group. (TIF 34525 KB)
10571_2018_617_MOESM2_ESM.tif
Supplementary Figure 2 BHB inhibits the activity of NLRP3 inflammasome in LN229 and U251 cells. A: U251 cells; B: LN229 cells. LN229 and U251 cells were treated with BHB for 24h, respectively. The protein levels of Pro-caspase-1, Pro-IL-1β and their mature form of caspase-1 and IL-1β were detected by Western blotting. β-actin was served as an internal control. ∗P < 0.05, ∗∗P < 0.01, compared with the control group. (TIF 88324 KB)
10571_2018_617_MOESM3_ESM.tif
Supplementary Figure 3 BHB inhibited the LPS/ATP induced cell migration in LN229 cell line. LN229 cells were pre-treated with LPS for 4 h, and then treated with BHB for 2 h, followed by 5 mM ATP simulation for 30 min. 24 h later, cell migration status was analyzed by Transwell chamber migration assay. A: The representative images were captured by phase-contrast microscope. B: The number of LN229 cells migrated into the down chamber were obtained from counting of five microscopic fields (up, down, left, right and middle). ∗P < 0.05, ∗∗P < 0.01, compared with the control group. # P <0.05, ## P < 0.01, compared with the LPS/ATP group. (TIF 23066 KB)
10571_2018_617_MOESM4_ESM.tif
Supplementary Figure 4 BHB inhibited the LPS/ATP induced NLRP3 inflammasome activation in LN229 cell line. LN229 cells were pre-treated with LPS for 4 h, and then treated with BHB for 2 h, followed by 5 mM ATP simulation for 30 min. a The protein levels of Pro-caspase-1, Pro-IL-1β and their mature form of caspase-1 and IL-1β were detected by Western blotting. β-actin was served as an internal control. b, c, d, e Densitometry of immoblots shown in a. ∗P < 0.05, ∗∗P < 0.01, compared with the control group. # P <0.05, ## P < 0.01, compared with the LPS/ATP group (TIF 41897 KB)
Rights and permissions
About this article
Cite this article
Shang, S., Wang, L., Zhang, Y. et al. The Beta-Hydroxybutyrate Suppresses the Migration of Glioma Cells by Inhibition of NLRP3 Inflammasome. Cell Mol Neurobiol 38, 1479–1489 (2018). https://doi.org/10.1007/s10571-018-0617-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-018-0617-2