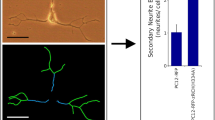
Abstract
A rapid drop of the transcription repressor REST/NRSF during precursor differentiation into nerve cells is known to release the repression of hundreds of specific genes and thus to orchestrate the acquisition of the specific phenotype. REST, however, is important not only for differentiation, but also for the maintenance of key properties in mature nerve cell. The PC12 line is uniquely favorable for studying REST because, in addition to the wild-type, low REST neurosecretory cells, it includes spontaneously defective clones lacking neurosecretion, where REST is as high as in non-nerve cells. In this article, we summarize our cell biologic studies of two nerve cell-specific processes dependent on REST, neurosecretion and neurite outgrowth. We demonstrate that, in wild-type PC12 transfected with REST constructs, expression of genes encoding proteins of dense-core and synaptic-like vesicles is decreased, though, to different extents, with chromogranins being the most and the SNAREs (except SNAP25) the least affected. Concomitantly, dense core-vesicles decrease markedly in size but can still be discharged by regulated exocytosis. When, in contrast, dominant-negative constructs of REST are transfected in high-REST PC12, and the main effector enzymes of REST, histone deacetylases, are blocked, dense-core vesicles reappear and are discharged upon stimulation. In high-REST PC12, also neurite outgrowth is inhibited by down regulation of the NGF receptor. Concomitantly, however, high REST induces the expression of proteins and of an exocytic organelle, the enlargeosome, which sustain a Rac1-dependent form of neurite outgrowth, unknown until now, operative in PC12, in neuroblastoma SH-SY5Y cells, and also in neurons.





Similar content being viewed by others
References
Abderrahmani A, Niederhauser G, Plaisance V, Haefliger JA, Regazzi R, Waeber G (2004) Neuronal traits are required for glucose-induced insulin secretion. FEBS Lett 565:133–138
Ariano P, Zamburlin P, D’Alessandro R, Meldolesi J, Lovisolo D (2010) Differential repression by the transcription factor REST/NRSF of the various Ca2+ signaling mechanisms in pheochromocytoma PC12 cells. Cell Calcium 47:360–368
Ballas N, Mandel G (2005) The many faces of REST oversee epigenetic programming of neuronal genes. Curr Opin Neurobiol 15:500–506
Bittler CM, Zhang MB, Howard BD (1986) PC12 variants deficient in catecholamine transport. J Neurochem 47:1286–1293
Borgonovo B, Cocucci E, Racchetti G, Podini P, Bachi A, Meldolesi J (2002) Regulated exocytosis: a novel, widely expressed system. Nat Cell Biol 4:955–962
Bruce AW, Donaldson IJ, Wood IC, Yerbury SA, Sadowski MI, Chapman M, Gottgens B, Buckley NJ (2004) Genome-wide analysis of repressor element-1 silencing transcription factor/neuron restrictive silencing factor (REST/NRSF) target genes. Proc Natl Acad Sci USA 101:10458–10463
Bruce AW, Krejci A, Ooi L, Deuchars J, Wood IC, Dolezal V, Buckley NJ (2006) The transcriptional repressor REST is a critical regulator of the neurosecretory phenotype. J Neurochem 98:1828–1840
Bruce AW, Lopez-Contreras AJ, Ficeck P, Dhami P, Dillon SC, Koch CM, Langford CF, Dunham I, Andrews RM, Vetrie D (2009) Functional diversity for REST defined by in vivo binding affinity hierarchies at the DNA sequence level. Genome Res 19:994–1005
Calderone A, Jover T, Noh K, Tanaka H, Yokota H, Lin Y, Grooms S, Regis R, Bennett MV, Zukin RS (2003) Ischemic insults derepress the gene silencer REST in neurons destined to die. J Neurosci 23:2112–2121
Conaco C, Otto S, Han JJ, Mandel G (2006) Reciprocal actions of REST and microRNA promote neuronal identity. Proc Natl Acad Sci USA 103:2422–2427
Corradi N, Borgonovo B, Clementi E, Bassetti M, Racchetti G, Consalez GG, Huttner WB, Meldolesi J, Rosa P (1996) Overall lack of regulated secretion in a PC12 variant cell clone. J Biol Chem 271:27116–27124
Coulson JM (2005) Transcriptional regulation: cancer, neurons and the REST. Curr Biol 17:R665–R668
Courel M, Rodemer C, Nguyen ST, Pance A, Jackson AP, O’Connor DT, Taupenot L (2006) Secretory granule biogenesis in sympathoadrenal cells: identification of a granulogenic determinant in the secretory prohormone chromogranin A. J Biol Chem 281:38038–38051
D’Alessandro R, Klaijn A, Stucchi L, Podini P, Malosio ML, Meldolesi J (2008) Expression of the neurosecretory process in PC12 cells is governed by REST. J Neurochem 105:1369–1383
Day R, Benjannet S, Matsuuchi L, Kelly RB, Marchinkiewicz M, Chrétien M, Seidah NG (1995) Maintained PC1 and PC2 expression in the AtT-20 variant cell line 6T3 lacking regulated secretion and POMC: restored POMC expression and regulated secretion after cAMP treatment. DNA Cell Biol 14:175–188
Ekici M, Hohl M, Schuit F, Martinez-Serrano A, Thiel G (2008) Transcription of genes encoding synaptic vesicle proteins in human neural stem cells: chromatin accessibility, histome methylation pattern and the essential role of REST. J Biol Chem 283:9257–9268
Formisano L, Noh KM, Miyawaki T, Mashiko T, Bennett MV, Zukin RS (2003) Ischemic insults promote epigenetic reprogramming of mu opioid receptor expression in hippocampal neurons. Proc Natl Acad Sci USA 104:4170–4175
Garriga-Canut M, Schoenike B, Qazi R, Bergendahl K, Daley TJ, Pfender RM, Morrison JF, Ockuly J, Stafstrom C, Sutula T, Roopra A (2006) 2-Deoxy-d-glucose reduces epilepsy progression by NRSF/REST-dependent metabolic regulation of chromatin structure. Nat Neurosci 9:1382–1387
Greenway JD, Street M, Jeffries A, Buckley NJ (2007) RE1 silencing transcription factor maintains a repressive chromatin environment in embryonic hippocampal neural stem cells. Stem Cells 25:354–363
Grundschober C, Malosio ML, Astolfi L, Giordano T, Nef P, Meldolesi J (2002) Neurosecretion competence. A comprehensive gene expression program identified in PC12 cells. J Biol Chem 277:36715–36724
Hohl M, Thiel G (2005) Cell type-specific regulation of RE-1 silencing transcription factor (REST) target genes. Eur J Neurosci 22:2216–2230
Johnson DS, Mortazavi A, Myers RM, Wold B (2007) Genome-wide mapping of in vivo protein–DNA interactions. Science 316:1497–1502
Kasai H, Kishimoto T, Liu T-T, Miyashita Y, Podini P, Grohovaz F, Meldolesi J (1999) Multiplicity and diversity of regulated exocytosis in wild-type and defective PC12 cells. Proc Natl Acad Sci USA 96:945–949
Klajn A, Ferrai C, Stucchi L, Prada I, Podini P, Baba T, Rocchi M, Meldolesi J, D’Alessandro R (2009) The rest repression of the neurosecretory phenotype is negatively modulated by BHC80, a protein of the BRAF/HDAC complex. J Neurosci 29:6296–6307
Kim CS, Hwang CH, Song KY, Choi HS, Kim do K, Law PY, Wei LN, Loh HH (2008) Novel function of neuron-restrictive silencing factor (NRSF) for posttranscriptional regulation. Biochim Biophys Acta 1783:1835–1846
Leoni C, Menegon A, Benfenati F, Toniolo D, Pennuto M, Valtorta F (1999) Neurite extension occurs in the absence of regulated exocytosis in PC12 subclones. Mol Biol Cell 10:2919–2931
Liu Z, Liu M, Niu G, Cheng Y, Fei J (2009) Genome-wide identification of target genes repressed by the zinc finger transcription factor REST/NRSF in the HEK 293 cell line. Acta Biochim Biophys Sin (Shanghai) 41:1008–1017
Lv H, Pan G, Zheng G, Wu X, Ren H, Liu Y, Wen J (2010) Expression and functions of the repressor element 1 (RE-1)-silencing transcription factor (REST) in breast cancer. J Cell Biochem 110:968–974
Magin A, Lietz M, Cibelli G, Thiel G (2002) RE-1 silencing transcription factor-4 (REST4) is neither a transcriptional repressor nor a de-repressor. Neurochem Int 40:195–202
Majumder S (2008) REST in good times and bad. Roles of the tumor suppressor and oncogenic activities. Cell Cycle 5:1929–1935
Martin D, Allagnat F, Chaffard G, Caille D, Fukuda M, Regazzi R, Abderrahmani A, Waeber G, Meda P, Maechler P, Haeflinger J-A (2008) Functional significance of repressor element 1 silencing transcription factor (REST) target genes in pancreatic beta cells. Diabetologia 51:1429–1439
Mortazavi A, Leeper Thompson EC, Garcia T, Myers RM, Wod B (2006) Comparative genomics modeling of the NRSF/REST repressor network: from single conserved sites to genome-wide repertoire. Genome Res 16:1208–1221
Nihihara S, Tsuda L, Ogura T (2002) The canonical Wnt pathway directly regulates NRSF/REST expression in chick spinal cord. Biochem Biophys Res Commun 311:55–63
Ooi L, Wood IC (2007) Chromatin crosstalk in development and disease: lessons from REST. Nat Rev Genet 8:544–554
Otto SJ, McCockle SR, Hover J, Conaco C, Han JJ, Impey S, Yochum GS, Dunn JJ, Goodman RH, Mandel G (2007) A new binding motif for the transcriptional repressor REST uncovers large gene networks devoted to neuronal functions. J Neurosci 27:6729–6739
Palm K, Metsis M, Timmusk T (1999) Neuron-specific splicing of zinc finger transcription factor REST/NRSF/XBR is frequent in neuroblastomas and conserved in human, mouse and rat. Brain Res Mol Brain Res 72:30–39
Pance A, Morgan K, Guest PC, Bowers K, Dean GE, Cutler DF, Jackson AP (1999) A PC12 variant lacking regulated secretory organelles: aberrant protein targeting and evidence for a factor inhibiting neuroendocrine gene expression. J Neurochem 73:21–30
Pance A, Livesey FJ, Jackson AP (2006) A role for the transcriptional repressor REST in maintaining the phenotype of neurosecretory-deficient PC12 cells. J Neurochem 99:1435–1444
Qureshi IA, Mehler MF (2009) Regulation of non-coding RNA networks in the nervous system. What’s the REST of the story? Neurosci Lett 466:73–80
Racchetti G, Lorusso A, Schulte C, Garavello D, Carabelli V, D’Alessandro R, Meldolesi J (2010) Rapid neurite outgrowth in neurosecretory cells and neurons sustained by the exocytosis of a cytoplasmic organelle, the enlargeosome. J Cell Sci 123:165–170
Schoch S, Cibelli G, Thiel G (1996) Neuron-specific expression of synapsin I. Major role of a negative regulatory mechanism. J Biol Chem 271:3317–3323
Schoenherr CJ, Paquette AJ, Anderson DJ (1996) Identification of potential target genes for the neuron-restrictive silencer factor. Proc Natl Acad Sci USA 93:9881–9886
Schulte C, Racchetti G, D’Alessandro R, Meldolesi J (2010) A new form of neurite outgrowth is sustained by the exocytosis of enlargeosomes expressed under the control of REST. Traffic 11:1304–1314
Seth KA, Majzoub JA (2001) Repressor element silencing transcription factor/neuron-restrictive silencing factor (REST/NRSF) can act as an enhancer as well as a repressor of corticotropin-releasing hormone gene transcription. J Biol Chem 276:13917–13923
Shimojo M (2006) Characterization of the nuclear targeting signal of REST/NRSF. Neurosci Lett 398:161–166
Tabuchi A, Yamada T, Sasagawa S, Naruse Y, Mori N, Tsuda M (2002) REST4-mediated modulation of REST/NRSF-silencing function during BDNF gene promoter activation. Biochem Biophys Res Commun 290:415–420
Westbrook TF, Hu G, Ang XL, Mulligan P, Pavlova NN, Liang A, Leng Y, Maehr R, Shi Y, Harper JW, Elledge SJ (2008) SCFbeta-TRCP controls oncogenic transformation and neural differentiation through REST degradation. Nature 452:370-374
Wu J, Xie X (2006) Comparative sequence analysis reveals an intricate network among REST, CREB and miRNA in mediating neuronal gene expression. Genome Biol 7:R85
Yoo AS, Staahl BT, Chen I, Crabtree GR (2009) MicroRNA-mediated switching of chromatin-remodelling complexes in neural development. Nature 460:642–646
Zheng D, Zhao K, Mehler MF (2009) Profiling RE1-REST-mediated histone modifications in the human genome. Genome Biol 10:R9
Acknowledgments
The contribution of the other members of the laboratory, in particular, of Andrijana Klajn and Gabriella Racchetti, to this study is gratefully acknowledged. The original study as summarized here was supported by grants from the Italian Ministry of Education and Research (FIRB 2003 and PRIN 2007), IIT, and Telethon 2009 (GGP09066).
Author information
Authors and Affiliations
Corresponding author
Additional information
A commentary to this article can be found at doi:10.1007/s10571-010-9610-0.
Rights and permissions
About this article
Cite this article
D’Alessandro, R., Meldolesi, J. In PC12 Cells, Expression of Neurosecretion and Neurite Outgrowth are Governed by the Transcription Repressor REST/NRSF. Cell Mol Neurobiol 30, 1295–1302 (2010). https://doi.org/10.1007/s10571-010-9602-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-010-9602-0