Abstract
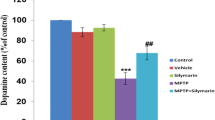
Dysfunction of the proteasome has been suggested to contribute in the degeneration of nigrostriatal dopaminergic neurons. Here, we investigated to determine whether systematic administration of proteasome inhibitor, carbobenzoxy-l-γ-t-butyl-l-glutamyl-l-alanyl-l-leucinal (PSI) protects against MPTP (1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine) neurotoxicity in mice. Three administrations of MPTP at 1-h intervals to mice reduced significantly the concentration of dopamine, DOPAC (3,4-dihydroxyphenylacetic acid) and HVA (homovanillic acid) in the striatum after 5 days. In contrast, PSI (0.3 and 1.0 mg/kg) prevented a significant decrease in dopamine, DOPAC and HVA contents of the striatum 5 days after MPTP treatment. In our Western blot analysis study, PSI at a dose of 1.0 mg/kg prevented a significant decrease in TH (tyrosine hydroxylase) protein and a significant increase in glial fibrillary acidic protein 5 days after MPTP treatment. Furthermore, our immunohistochemical study showed that PSI at a dose of 1.0 mg/kg prevented a significant loss in TH immunopositive neurons in the striatum and substantia nigra 5 days after MPTP treatment. In contrast, PSI caused a significant increase in the number of intense ubiquitin immunopositive cells in the striatum and substantia nigra 5 days after MPTP treatment. These results indicate that proteasome inhibitors can protect against MPTP neurotoxicity in mice. The neuroprotective effect of PSI against dopaminergic cell damage may be mediated by the elevation of ubiquitination. Thus, our findings provide further valuable information for the pathogenesis of Parkinson’s disease.







Similar content being viewed by others
References
Araki T, Kumagai T, Tanaka K, Matsubara M, Kato H, Itoyama Y et al (2001) Neuroprotective effect of riluzole in MPTP-treated mice. Brain Res 918:176–181. doi:10.1016/S0006-8993(01)02944-4
Bobba A, Canu N, Atlante A, Petragallo V, Calissano P (2002) Proteasome inhibitors prevent cytochrome c release during apoptosis but not in excitotoxic death of cerebellar granule neurons. FEBS Lett 515:8–12. doi:10.1016/S0014-5793(02)02231-7
Bove J, Zhou C, Jackson-Lewis V, Taylor J, Chu Y, Rideout HJ et al (2006) Proteasome inhibition and Parkinson’s disease modeling. Ann Neurol 60:260–264. doi:10.1002/ana.20937
Braak H, Del Tredici K, Rub U, de Vos RA, Jansen Steur EN, Braak E (2003) Staging of brain pathology related to sporadic Parkinson’s disease. Neurobiol Aging 24:197–211. doi:10.1016/S0197-4580(02)00065-9
Burns RS, Chiueh CC, Markey SP, Ebert MH, Jacobowitz DM, Kopin IJ (1983) A primate model of parkinsonism: selective destruction of dopaminergic neurons in the pars compacta of the substantia nigra by N-methyl-4-phenyl-1, 2, 3, 6-tetrahydropyridine. Proc Natl Acad Sci USA 80:4546–4550. doi:10.1073/pnas.80.14.4546
Canu N, Barbato C, Ciotti MT, Serafino A, Dus L, Calissano P (2000) Proteasome involvement and accumulation of ubiquitinated proteins in cerebellar granule neurons undergoing apoptosis. J Neurosci 20:589–599
Cross T (1981) Aquatic actinomycetes: a critical survey of the occurrence, growth and role of actinomycetes in aquatic habitats. J Appl Bacteriol 50:397–423
Dauer W, Przedborski S (2003) Parkinson’s disease: mechanisms and models. Neuron 39:889–909. doi:10.1016/S0896-6273(03)00568-3
Ensign JC, Normand P, Burden JP, Yallop CA (1993) Physiology of some actinomycete genera. Res Microbiol 144:657–660. doi:10.1016/0923-2508(93)90070-I
Fenteany G, Schreiber SL (1998) Latacystin, proteasome function, and cell fate. J Biol Chem 273:8545–8548. doi:10.1074/jbc.273.15.8545
Fornai F, Lenzi P, Gesi M, Ferrucci M, Lazzeri G, Busceti CL et al (2003) Fine structure and biochemical mechanisms underlying nigrostriatal inclusions and cell death after proteasome inhibition. J Neurosci 23:8955–8966
Heikkila RE, Hess A, Duvoisin RC (1984) Dopaminergic neurotoxicity of 1-methyl-4-phenyl-1, 2, 5, 6-tetrahydropyidine in mice. Science 224:1451–1453. doi:10.1126/science.6610213
Hirst SJ, Ferger B (2008) Systemic proteasomal inhibitor exposure enhances dopamine turnover and decreases dopamine levels but does not affect MPTP-induced striatal dopamine depletion in mice. Synapse 62:85–90. doi:10.1002/syn.20457
Inoue M, Zhai H, Sahazaki H, Furuyama H, Hirima M (2004) TMC-95A, a reversible proteasome inhibitor, induces neurite outgrowth in PC12 cells. Bioorg Med Chem Lett 14:663–665. doi:10.1016/j.bmcl.2003.11.043
Jana NR, Dikshit P, Goswami A, Nukina N (2004) Inhibition of poteasomal function by curcumin induces apoptosis through mitochondrial pathway. J Biol Chem 279:11680–11685. doi:10.1074/jbc.M310369200
Kadoguchi N, Kimoto H, Yano R, Kato H, Araki T (2008) Failure of acute administration with proteasome inhibitor to provide a model of Parkinson’s disease in mice. Metab Brain Dis 23:147–154. doi:10.1007/s11011-008-9082-9
Kazi A, Urbizu DA, Kuhn DJ, Acebo AL, Jackson ER, Greenfelder GP et al (2003) A natural musaceas plant extract inhibits proteasome activity and induces apotosis selectively in human tumor and transformed, but not normal and non-transformed, cells. Int J Mol Med 12:879–887
Kisselev AF, Goldberg AL (2001) Proteasome inhibitiors: from research tools to drug candidates. Chem Biol 8:739–758. doi:10.1016/S1074-5521(01)00056-4
Kitada T, Asakawa S, Hattori N, Matsumine H, Yamamura Y, Minoshima S (1998) Mutations in the parkin gene cause autosomal recessive juvenile parkinsonism. Nature 392:605–608. doi:10.1038/33416
Koguchi Y, Kohno J, Nishio M, Takahashi K, Okuda T, Ohnuki T (2000) TMC-95A, B, C, and D, novel proteasome inhibitors produced by Apiospora montagnei Sacc. Taxonomy, production, isolation, and biological activities. J Antibiot 53:105–109
Kordower JH, Kanaan NM, Chu Y, Babu RS, Stansell J, Terpstra BT et al (2006) Failure of proteasome inhibitor administration to provide a model of Parkinson’s disease in rats and monkeys. Ann Neurol 60:264–268. doi:10.1002/ana.20935
Kurosaki R, Muramatsu Y, Kato H, Araki T (2004) Biochemical, behavioral and immunohistochemical alterations in MPTP-treated mouse model of Parkinson’s disease. Pharmacol Biochem Behav 78:143–153. doi:10.1016/j.pbb.2004.03.006
Kurosaki R, Muramatsu Y, Kato H, Watanabe Y, Imai Y, Itoyama Y et al (2005) Effect of angiotensin-converting enzyme inhibitor perindopril on interneurons in MPTP-treated mice. Eur Neuropsychopharmacol 15:57–67. doi:10.1016/j.euroneuro.2004.05.007
Langston JW, Ballard P, Terud JW, Irwin I (1983) Chronic parkinsonism in humans due to a product of meperidine-analog synthesis. Science 219:979–980. doi:10.1126/science.6823561
Mathur BN, Neely MD, Dyllick-Brenzinger M, Tandon A, Deutch AY (2007) Systemic administration of a proteasome inhibitor does not cause nigrostriatal dopaminergic degeneration. Brain Res 1168:83–89. doi:10.1016/j.brainres.2007.06.076
McNaught KS, Belizaire R, Isacson O, Jenner P, Olanow CW (2003) Altered proteasomal function in sporadic Parkinson’s disease. Exp Neurol 179:38–46. doi:10.1006/exnr.2002.8050
McNaught KS, Perl DP, Brownell AL, Olanow CW (2004) Systemic exposure to proteasome inhibitors causes a progressive model of Parkinson’s disease. Ann Neurol 56:149–162. doi:10.1002/ana.20186
Miwa H, Kubo T, Suzuki A, Nishi K, Kondo T (2005) Retrograde dopaminergic neuron degeneration following intrastriatal proteasome inhibition. Neurosci Lett 380:93–98. doi:10.1016/j.neulet.2005.01.024
Momose I, Sekizawa R, Iinuma H, Takeuchi T (2002) Inhibition of proteasome activity by tyropeptin A in PC12 cells. Biosci Biotechnol Biochem 66:2256–2258. doi:10.1271/bbb.66.2256
Nam S, Smith DM, Dou QP (2001) Ester bond-containing tea polyphenols potently inhibit proteasome activity in vitro and in vivo. J Biol Chem 276:13322–13330. doi:10.1074/jbc.M004209200
Obin M, Mesco E, Gong X, Haas AL, Joseph J, Taylor A (1999) Neurite outgrowth in PC12 cells. J Biol Chem 274:11789–11795. doi:10.1074/jbc.274.17.11789
Pasquini LA, Paez PM, Besio Moreno MAN, Pasquini JM, Soto EF (2003) Inhibition of the protein by lactacystin enhances oligodendroglial cell differentiation. J Neurosci 23:4635–4644
Petrucelli L, Dawson TM (2004) Mechanism of neurodegenerative disease: role of the ubiquitin proteasome system. Ann Med 36:315–320. doi:10.1080/07853890410031948
Phillips JB, Williams AJ, Adams J, Elliott PJ, Tortella FC (2000) Proteasome inhibitor PS519 reduces infarction and attenuates leukocyte infiltration in rat model of focal cerebral ischemia. Stroke 31:1686–1693
Priyadarshi A, Khuder SA, Schaub EA, Priyadarshi SS (2001) Environmental risk factors and Parkinson’s disease: a metaanalysis. Environ Res 86:122–127. doi:10.1006/enrs.2001.4264
Sadoul R, Fernandez PA, Quiquerez AL, Martinou I, Maki M, Schroter M et al (1996) Involvement of the proteasome in the programmed cell death of NGF-deprived sympathetic neurons. EMBO J 15:3845–3852
Sawada H, Kohno R, Kihara T, Izumi Y, Sakka N, Ibi M et al (2004) Proteasome mediates dopaminergic neuronal degeneration, and its inhibition causes alpha-synuclein inclusions. J Biol Chem 279:10710–10719. doi:10.1074/jbc.M308434200
Schapira AH, Cletter MW, Muddle JR, Workman JM, Cooper JM, King RH (2006) Proteasomal inhibition causes loss of nigral tyrosine hydroxylase neurons. Ann Neurol 60:253–255. doi:10.1002/ana.20934
Sherman NY, Goldberg AL (2001) Cellular defenses against unfolded proteins: a cell biologist thinks about neurodegenerative diseases. Neuron 29:15–32. doi:10.1016/S0896-6273(01)00177-5
Sin N, Kim KB, Elofsson M, Meng L, Auth H, Kwok BH et al (1999) Total synthesis of the potent proteasome inhibitor epoxomicin: a useful tool for understanding proteasome biology. Bioorg Med Chem Lett 9:2283–2288. doi:10.1016/S0960-894X(99)00376-5
Song EJ, Hong HM, Yoo YS (2009) Proteasome inhibition induces neurite outgrowth trough posttranslational modification of TrkA receptor. Int J Biochem Cell Biol 41:539–545. doi:10.1016/j.biocel.2008.04.022
Takagi S, Hayakawa N, Kimoto H, Kato H, Araki T (2007) Damage to oligodendrocytes in the striatum after MPTP neurotoxicity in mice. J Neural Transm 114:1553–1557. doi:10.1007/s00702-007-0790-9
Tanaka A, Watanabe Y, Kato H, Araki T (2007) Immunohistochemical changes related to ageing in the mouse hippocampus and subventricular zone. Mech Ageing Dev 128:303–310. doi:10.1016/j.mad.2007.01.002
van Leyen K, Siddiq A, Ratan RR, Lo EH (2005) Proteasome inhibition protects HT22 neuronal cells from oxidative glutamate toxicity. J Neurochem 92:824–830. doi:10.1111/j.1471-4159.2004.02915.x
Watanabe Y, Kato H, Araki T (2008) Protective action of neuronal nitric oxide synthase inhibitor in the MPTP mouse model of Parkinson’s disease. Metab Brain Dis 23:51–69. doi:10.1007/s11011-007-9080-3
Yamamoto N, Sawada H, Izumi Y, Kume T, Katsuki H, Shimohama S et al (2007) Proteasome inhibition induces glutathione synthesis and protects cells from oxidative stress. J Biol Chem 282:4364–4372. doi:10.1074/jbc.M603712200
Yokoyama H, Takagi S, Watanabe Y, Kato H, Araki T (2008) Role of reactive nitrogen and reactive oxygen species against MPTP neurotoxicity in mice. J Neural Transm 115:831–842. doi:10.1007/s00702-008-0019-6
Zhang Y, Gao J, Chung KK, Huang H, Dawson VL, Dawson TM (2000) Parkin functions as an E2-dependent ubiquitin-protein ligase and promotes the degradation of the synaptic vesicle-associated protein, CDCrel-1. Proc Natl Acad Sci USA 97:13354–13359. doi:10.1073/pnas.240347797
Acknowledgments
We thank Sho Takagi and Eriko Aoki for technical assistance.
Author information
Authors and Affiliations
Corresponding author
Additional information
Takuya Oshikawa and Hayato Kuroiwa contributed equally to this work.
Rights and permissions
About this article
Cite this article
Oshikawa, T., Kuroiwa, H., Yano, R. et al. Systemic Administration of Proteasome Inhibitor Protects Against MPTP Neurotoxicity in Mice. Cell Mol Neurobiol 29, 769–777 (2009). https://doi.org/10.1007/s10571-009-9402-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-009-9402-6