Abstract
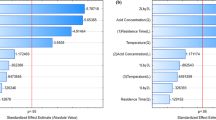
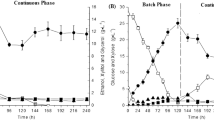
Currently, sugarcane bagasse (SB) is the most abundant agricultural residue generated in Brazil. Given that hemicelluloses can reach up to 30% of SB, bioconversion of this fraction into second generation ethanol (2G) is essential for the success of biorefinery based operations. For 2G ethanol production, techniques such as immobilization process could be an interesting strategy to improve process productivity and must receive special investigation. Cell immobilization of the native Brazilian pentose wild converting yeast, Scheffersomyces shehatae UFMG-HM 52.2, was proposed for the production of 2G ethanol from SB hemicellulosic hydrolysate. A 23 full experimental design was constructed with varying concentrations of sodium alginate (1.0, 1.5 and 2.0% w/v), and calcium chloride (0.1 M, 0.15 M and 0.2 M) and conditioning time (12, 18 and 24 h). Through statistical analysis, it was determined that highest ethanol yield (YP/S of 0.32 g/g) and productivity (QP of 0.146 g/L.h) were obtained with immobilization conditions of 1% sodium alginate, 0.2 M calcium chloride and 12 h of conditioning time. Repeated batches were conducted employing these defined conditions, showing the feasibility to use the system for 5 consecutive cycles. Results highlighted the use of proposed approach for ethanol production, promoting its inclusion in biorefinery operation portfolios.
Graphical abstract
Hemicellulosic hydrolysate from sugarcane bagasse: a large available carbon source; S. shehatae UFMG-HM 52.2 is a Brazilian wild native pentose fermenting yeast; Immobilization conditions for this yeast were defined for ethanol production; Stability in ethanol production was demonstrated in 5 repeated consecutive batches; Our approach was demonstrated feasible for industrial 2G ethanol process development.







Similar content being viewed by others
References
Akin C (1987) Biocatalysis with immobilized cells. Biotechnol Genetic Eng Rev 5:319–367
Alves LA, Felipe MGA, Silva JBA, Silva SS, Prata AMR (1998) Pretreatment of sugarcane bagasse hemicellulose hydrolysate for xylitol production by Candida guilliermondii. Appl Biochem Biotechnol 70:89–98
Alvira P, Tomás-pejó E, Ballesteros M, Negro MJ (2010) Pretreatment technologies for an efficient bioethanol production process based on enzymatic hydrolysis: a review. Bioresour Technol 101:4851–4861
Antunes FAF, Chandel AK, Santos JC; et al (2018) Bioethanol: An overview of production possibilities. In: Brienzo, M.. (Ed) Bioethanol and Beyond ¬ Andvances in Production Process and Future Directions. 1ed.NY - USA: Nova Science Publishers
Bangrak P, Savitree L, Phisalaphong M (2011) Continuous ethanol production using immobilized yeast cells entrapped in loofa-reinforced alginate carriers. Braz J Microbiol 42:676–684
Behera S, Kar S, Mohanty RC, Ray RC (2010) Comparative study of bio-ethanol production from (mahula (Madhula latifolia L.) flowers by Saccharomyces cerevisiae cells immobilized in agar and Ca alginate matrices. Appl Energy 87:96–100
Behera S, Mohanty RC, Ray RC (2012) Ethanol fermentation of sugarcane molasses by Zymomonas mobilis MTCC 92 immobilized in Luffa cylindrical L. sponge discs and Ca- alginate matrices. Braz J Microbiol 43:1499–1507
Brennan L, Owende P (2010) Biofuels from microalgae—a review of technologies for production, processing, and extractions of biofuels and co-products. Renew Sustain Energy Rev 14:557–577
Canilha L, Chandel AK, Milessi TSS, Antunes FAF, Freitas WLC, Felipe MGA, da Silva SS (2012) Review article : Bioconversion of sugarcane biomass into ethanol: an overview about composition, pretreatment methods, detoxification of hydrolysates, enzymatic saccharification, and ethanol fermentation. J Biomed Biotechnol 2012:1–15
Carvalho W, Silva SS, Vitolo M, Felipe MGA, Mancilha IM (2002) Improvement in xylitol production from sugarcane bagasse hydrolysate achieved by the use of a repeated-batch immobilized cell system. Z Naturforsch C 57:109–112
Carvalho W, Silva SS, Santos JC, Converti A (2003) Xylitol production by Ca-alginate entrapped cells: comparison of different fermentation systems. Enzyme Microb Technol 32:553–559
Cerqueira-Leite RC, Leal MRLV, Cortez LAB, Griffin WM, Scandiffio MIG (2009) Can Brazil replace 5% of the 2025 gasoline world demand with ethanol? Energy 34:655–661
Chandel AK, Antunes FAF, Anjos V, Bell MJB, Rodrigues LN, Singh OV, Rosa CA, Pagnocca FC, da Silva SS (2013) Ultra-structural mapping of sugarcane bagasse after oxalic acid fiber expansion (OAFEX) and ethanol production by Candida shehatae and Saccharomyces cerevisiae. Biotechnol Biofuels 6(4):1–15
Chandel AK, Antunes FAF, Anjos V et al (2014) Multi-scale structural and chemical analysis of sugarcane bagasse in the process of sequential acidbasepretreatment and ethanol production by Scheffersomyces shehatae and Saccharomyces cerevisiae. Biotechn Biofuels 7:63
Chandel AK, Garlapatib VK, Singhc AK, Antunes FAF, da Silva SS (2018a) The path forward for lignocellulose biorefineries: bottlenecks, solutions, and perspective on commercialization. Biores Technol 26:370–381
Chandel AK, Garlapati VK, Singh AK, Antunes FAF, Silva SS (2018b) The path forward for lignocellulose biorefineries: Bottlenecks, solutions, and perspective on commercialization. Biores Technol. https://doi.org/10.1016/j.biortech.2018.06.004
Chen KC, Huang CT (1998) Effects of the growth of T. cutaneum in calcium alginate gel beads upon bead structure and oxygen transfer characteristics. Enzyme Microb Technol 10:284–292
Cherubini F (2010) The biorefinery concept: using biomass instead of oil for producing energy and chemicals. Energy Convers Manag 51:1412–1421
CONAB (2018) National Supply Company. Monitoring of the Brazilian harvest, Brazilian Supply Ministry (Segundo Levantamento Safra 2018/2019). https://www.conab.gov.br/info-agro/safras/cana/boletim-da-safra-de-cana-de-acucar. Accessed Oct 2018
Freeman A, Lilly MD (1998) Effect of processing parameters on the feasibility and operational stability of immobilized viable microbial cells. Enzyme Microb Technol 23:335–345
Gírio FM, Fonseca C, Carvalheiro F, Duarte LC, Marques S, Bogel-Lukasik R (2010) Hemicelluloses for fuel ethanol: a review. Bioresour Technol 101:4775–4800
Ha SJ, Galazkac JM, Kima SR, Choia JH, Yang X, Seoe JHN, Glassf L, Catec JHD, Jina YS (2011) Engineered Saccharomyces cerevisiae capable of simultaneous cellobiose and xylose fermentation. Proc Natl Acad Sci (PNAS) 108:504–509
Hahn-hägerdal B, Jeppsson H, Skoog K, Prior B (1994) Biochemistry and physiology of xylose fermentation by yeasts. Enzyme Microbiol Technol 16:933–942
Harmsen PFH, Huijgen WJJ, Bermúdez Lopez LM, Bakke RC (2012) Literature review of physical and chemical pretreatment processes for lignocellulosic biomass. Food and Biomass Res Wageningen UR 1:1–49
Holtzapple T (2003) Hemicelluloses. Encycl Food Sci Nutr. https://doi.org/10.1016/B0-12-227055-X/00589-7
Ivanova V, Petrova P, Hristov J (2011) Application in the ethanol fermentation of immobilized yeast cells in matrix of alginate/magnetic nanoparticles, on chitosan-magnetite microparticles and cellulose-coated magnetic nanoparticles. Int Rev Chem Eng 3:289–299
Jeffries TW (1983) Utilization of xylose by bacteria, yeasts and fungi. Adv Biochem Eng Biotechnol 27:1–32
Knauf M, Moniruzzaman M (2004) Lignocellulosic biomass processing: a perspective. Int Sugar J 1263:147–150
Ksungur YG, Zorlu N (2001) Production of ethanol from beet molasses by Ca-alginate immobilized yeast cells in a packed-bed bioreactor. Turkish J Biol 25:265–275
Meena K, Raja TK (2006) Immobilization of Saccharomyces cerevisiae cells by gel entrapment using various metal alginates. World J Microbiol Biotechnol 22:651–653
Nelder JA (1998) The selection of terms in response surface models—how strong is the weak heredity principle. Am Stat 52:16
Nigam JN (2000) Continuous ethanol production from pineapple cannery waste using immobilized yeast cell. J Biotechnol 80:189–193
Ogbonna JC, Amano Y, Nakamura KJ (1989) Elucidation of optimum conditions for immobilization of viable cells by using calcium alginate. J Ferment Bioeng 67:92–96
Omar SH (1993) Oxygen diffusion through gels employed for immobilization. 2—In the presence of microorganisms. Appli Microbiol Biotechnol 40:173–181
Parajó JC, Dominguez H, Dominguez JM (1998) Biotechnological production of xylitol. Part 1: interest of xylitol and fundamentals of its biosynthesis. Bioresour Technol 65:191–201
Parekh SR, Yu S, Wayman M (1986) Adaptation of Candida shehatae and Pichia stipitis to wood hydrolysates for increased ethanol production. Appl Microbiol Biotechnol 25:300–304
Peart PC, Chen ARM, Reynolds WR, Reese PB (2012) Entrapment of mycelial fragments in calcium alginate: a general technique for the use of immobilized filamentous fungi in biocatalysis. Steroids 77:85–90
Perez-Bibbins B, Torrado-Agrasar A, Salgado JM, Mussato SI, Dominguez JM (2015) Xylitol production in immobilized cultures: a recent review. Crit Rev Biotechnol 10:1–14
Rowell RM, Pettersen R, Tshabalala MA (2005) Cell wall chemistry. In: Rowell RM (ed) Handbook of wood chemistry and wood composites. CRC Press, Boca Raton, pp 35–74
Sanda T, Hasunuma T, Matsuda F, Kondo A (2011) Repeated-batch fermentation of lignocellulosic hydrolysate to ethanol using a hybrid Saccharomyces cerevisiae strain metabolically engineered for tolerance to acetic and formic acids. Biores Technol 102:7917–7924
Santos JC, Silva SS, Mussatto SI, Carvalho W, Cunha MA (2005) Immobilized cells cultivated in semi-continuous mode in a fluidized bed reactor for xylitol production from sugarcane bagasse. World J Microbiol Biotechnol 21:531–535
Sarrouh BF, Silva SS (2013) Repeated batch cell-immobilized system for the biotechnological production of xylitol as a renewable green sweetener. Appl Biochem Biotechnol 169:3–12
Sarrouh BF, Tresinari DS, Silva SS (2007) Biotechnological production of xylitol in a three-phase fluidized bed bioreactor with immobilized yeast cells in Ca-alginate beads. Biotechnol J 2:759–763
Singh A, Sharma P, Saran AK, Singh N, Bishnoi NR (2013) Comparative study on ethanol production from pretreated sugarcane bagasse using immobilized Saccharomyces cerevisiae on various matrices. Renewable Energy 50:488–493
Wang Y, Tan L, Wang T, Sun Z, Tang Y, Kida K (2017) Production of ethanol from kitchen waste by using flocculating Saccharomyces cerevisiae KF-7. Environ Technol 38:316–325
Winkelhausen E, Kuzmanova S (1998) Microbial conversion of d-xylose to xylitol. J Ferment Bioeng 86:1–14
Zhao J, Xia L (2010) Ethanol production from corn stover hemicellulosic hydrolysate using immobilized recombinant yeast cells. Biochem Eng J 49:28–32
Acknowledgments
Authors would like to thank Conselho Nacional de Desenvolvimento Científico e Tecnológico—Processo CNPq [154193/2018-6], FAPESP (Fundação de Amparo à Pesquisa do Estado de São Paulo) (2014/27055-2), CAPES (Coordenação de Aperfeiçoamento de Pessoal de Nível Superior), and FAPESP-BIOEN. AKC is thankful to USP-CAPES visiting researcher program. Authors also thank Prof. Carlos A. Rosa, who kindly provided the yeast S. shehatae UFMG-HM 52.2 from the culture collection of Federal University of Minas Gerais- MG- BRAZIL.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Antunes, F.A.F., Santos, J.C., Chandel, A.K. et al. Repeated batches as a feasible industrial process for hemicellulosic ethanol production from sugarcane bagasse by using immobilized yeast cells. Cellulose 26, 3787–3800 (2019). https://doi.org/10.1007/s10570-019-02341-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-019-02341-z