Abstract
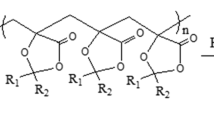
A novel synthetic method to link acetylated cellulose derivatives with methylated cellulose derivatives via Huisgen 1,3-dipolar cycloaddition was developed to produce 1,2,3-triazole-linked diblock copolymers consisting of hydrophilic cellobiose or low-molecular-weight cellulose and a hydrophobic 2,3,6-tri-O-methyl-cellulose. Huisgen 1,3-dipolar cycloaddition had the advantage over glycosylation reaction of being able to connect a hydrophilic block having higher molecular weight than cellobiose with a hydrophobic 2,3,6-tri-O-methyl-cellulose block. As a consequence, 2.0 wt% aqueous solutions of the 1,2,3-triazole-linked diblock methylcellulose analogues exhibited the thermoreversible gelation in water at around 25 °C as same as that of β-(1 → 4)-linked diblock methylcellulose. Differential scanning calorimetry measurements of 2.0 wt% aqueous solutions of the diblock copolymers revealed that an important structural factor for its thermoreversible gelation was not a β-(1 → 4)-glycosidic linkage between hydrophilic and hydrophobic blocks of diblock methylcellulose, but a sequence of anhydro 2,3,6-tri-O-methyl-glucopyranosyl units and that of unmodified glucopyranosyl ones.










Similar content being viewed by others
References
Adden R, Melander C, Brinkmalm G, Gorton L, Mischnick P (2006) New approaches to the analysis of enzymatically hydrolyzed methyl cellulose. Part 1. Investigation of the influence of structural parameters on the extent of degradation. Biomacromolecules 7(5):1399–1409
Bodvik R, Dedinaite A, Karlson L, Bergström M, Bäverbäck P, Pedersen JS, Edwards K, Karlsson G, Varga I, Claesson PM (2010) Aggregation and network formation of aqueous methylcellulose and hydroxypropylmethylcellulose solutions. Colloids Surf A Physicochem Eng Aspects 354(1–3):162–171
Chittaboina S, Xie F, Wang Q (2005) One-pot synthesis of triazole-linked glycoconjugates. Tetrahedron Lett 46(13):2331–2336
Elchinger P-H, Faugeras P-A, Boëns B, Brouillette F, Montplaisir D, Zerrouki R, Lucas R (2011) Polysaccharides: the “Click” chemistry impact. Polymers 3(4):1607–1651
Enomoto-Rogers Y, Kamitakahara H, Takano T, Nakatsubo F (2009) Cellulosic graft copolymer: poly(methyl methacrylate) with cellulose side chains. Biomacromolecules 10(8):2110–2117
Enomoto-Rogers Y, Kamitakahara H, Yoshinaga A, Takano T (2012) Comb-shaped graft copolymers with cellulose side-chains prepared via click chemistry. Carbohydr Polym 87(3):2237–2245
Fenn D, Pohl M, Heinze T (2009) Novel 3-O-propargyl cellulose as a precursor for regioselective functionalization of cellulose. React Funct Polym 69(6):347–352
Györgydeák Z, Szilágyi L, Paulsen H (1993) Synthesis, structure and reactions of glycosyl azides. J Carbohydr Chem 12(2):139–163
Hasegawa T, Numata M, Sakurai K, Shinkai S (2006) “Click chemistry” on polysaccharides: a convenient, general, and monitorable approach to develop β-1,3-glucans with various functional appendages. Carbohydr Res 341:35–40
Hirrien M, Chevillard C, Desbrières J, Axelos MAV, Rinaudo M (1998) Thermogelation of methylcelluloses: new evidence for understanding the gelation mechanism. Polymer 39(25):6251–6259
Hotha S, Kashyap S (2005) “Click Chemistry” inspired synthesis of pseudo-oligosaccharides and amino acid glycoconjugates. J Org Chem 71(1):364–367
Kamitakahara H, Nakatsubo F (2005) Synthesis of diblock copolymers with cellulose derivatives. 1. Model study with azidoalkyl carboxylic acid and cellobiosylamine derivative. Cellulose 12(2):209–219
Kamitakahara H, Nakatsubo F (2010) ABA- and BAB-triblock cooligomers of tri-O-methylated and unmodified cello-oligosaccharides: syntheses and structure–solubility relationship. Cellulose 17(1):173–186
Kamitakahara H, Enomoto Y, Hasegawa C, Nakatsubo F (2005) Synthesis of diblock copolymers with cellulose derivatives. 2. Characterization and thermal properties of cellulose triacetate-block-oligoamide-15. Cellulose 12(5):527–541
Kamitakahara H, Nakatsubo F, Klemm D (2006) Block co-oligomers of tri-O-methylated and unmodified cello-oligosaccharides as model compounds for methylcellulose and its dissolution/gelation behavior. Cellulose 13(4):375–392
Kamitakahara H, Nakatsubo F, Klemm D (2007) New class of carbohydrate-based nonionic surfactants: diblock co-oligomers of tri-O-methylated and unmodified cello-oligosaccharides. Cellulose 14(5):513–528
Kamitakahara H, Yoshinaga A, Aono H, Nakatsubo F, Klemm D, Burchard W (2008) New approach to unravel the structure–property relationship of methylcellulose. Cellulose 15(6):797–801
Kamitakahara H, Murata-Hirai K, Tanaka Y (2012) Synthesis of blockwise alkylated tetrasaccharide-organic quantum dot complexes and their utilization for live cell labeling with low cytotoxicity. Cellulose 19(1):171–187
Kato T, Yokoyama M, Takahashi A (1978) Melting temperatures of thermally reversible gels IV. Methyl cellulose–water gels. Colloid Polym Sci 256(1):15–21
Koschella A, Hartlieb M, Heinze T (2011) A “click-chemistry” approach to cellulose-based hydrogels. Carbohydr Polym 86(1):154–161
Liebert T, Hänsch C, Heinze T (2006) Click chemistry with polysaccharides. Macromol Rapid Commun 27(3):208–213
Marmuse L, Nepogodiev SA, Field RA (2005) “Click chemistry” en route to pseudo-starch. Org Biomol Chem 3(12):2225–2227
Miller GL, Dean J, Blum R (1960) A study of methods for preparing oligosaccharides from cellulose. Arch Biochem Biophys 91(1):21–26
Nakagawa A, Fenn D, Koschella A, Heinze T, Kamitakahara H (2011a) Physical properties of diblock methylcellulose derivatives with regioselective functionalization patterns: first direct evidence that a sequence of 2,3,6-tri-O-methyl-glucopyranosyl units causes thermoreversible gelation of methylcellulose. J Polym Sci, Part B: Polym Phys 49(21):1539–1546
Nakagawa A, Fenn D, Koschella A, Heinze T, Kamitakahara H (2011b) Synthesis of diblock methylcellulose derivatives with regioselective functionalization patterns. J Polym Sci Part A: Polym Chem 49(23):4964–4976
Nakagawa A, Kamitakahara H, Takano T (2011c) Synthesis of blockwise alkylated (1 → 4) linked trisaccharides as surfactants: influence of configuration of anomeric position on their surface activities. Carbohydr Res 346(13):1671–1683
Negishi K, Mashiko Y, Yamashita E, Otsuka A, Hasegawa T (2011) Cellulose chemistry meets click chemistry: syntheses and properties of cellulose-based glycoclusters with high structural homogeneity. Polymers 3(1):489–508
Neto V, Granet R, Krausz P (2010) Novel class of non-ionic monocatenary and bolaform alkylglycoside surfactants. Synthesis by microwave-assisted glycosylation and olefin cross-metathesis or by ‘click-chemistry’: physicochemical studies. Tetrahedron 66(25):4633–4646
Pohl M, Schaller J, Meister F, Heinze T (2008) Selectively dendronized cellulose: synthesis and characterization. Macromol Rapid Commun 29(2):142–148
Pohl M, Michaelis N, Meister F, Heinze T (2009) Biofunctional surfaces based on dendronized cellulose. Biomacromolecules 10(2):382–389
Sarkar N, Walker LC (1995) Hydration–dehydration properties of methylcellulose and hydroxypropylmethylcellulose. Carbohydr Polym 27(3):177–185
Schatz C, Lecommandoux S (2010) Polysaccharide-containing block copolymers: synthesis, properties and applications of an emerging family of glycoconjugates. Macromol Rapid Commun 31:1664–1684
Wilkinson BL, Bornaghi LF, Poulsen S-A, Houston TA (2006) Synthetic utility of glycosyl triazoles in carbohydrate chemistry. Tetrahedron 62(34):8115–8125
Zhang F, Bernet B, Bonnet V, Dangles O, Sarabia F, Vasella A (2008) 2-Azido-2-deoxycellulose: synthesis and 1,3-dipolar cycloaddition. Helv Chim Acta 91(4):608–617
Zhou J, Xu Y, Wang X, Qin Y, Zhang L (2008) Microstructure and aggregation behavior of methylcelluloses prepared in NaOH/urea aqueous solutions. Carbohydr Polym 74(4):901–906
Acknowledgments
This investigation was supported in part by a Grant-in-Aid for Scientific Research from the Ministry of Education, Science, and Culture of Japan (Nos. 18680009 and 21580205).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nakagawa, A., Kamitakahara, H. & Takano, T. Synthesis and thermoreversible gelation of diblock methylcellulose analogues via Huisgen 1,3-dipolar cycloaddition. Cellulose 19, 1315–1326 (2012). https://doi.org/10.1007/s10570-012-9703-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-012-9703-7