Abstract
Purpose
The 26S proteasome plays important roles in many intracellular processes and is therefore a critical intracellular cellular target for anticancer treatments. The primary aim of the current study was to identify critical proteins that may play roles in opposing the antisurvival effect of the proteasome inhibitor bortezomib together with the calcium-chelator BAPTA-AM in cancer cells using label-free LC-MS/MS. In addition, based on the results of the proteomic technique, a novel and more effective inhibitor combination involving bortezomib as well as OTSSP167 was developed for breast cancer cells.
Methods and results
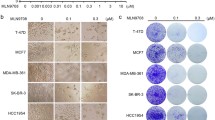
Using label-free LC-MS/MS, it was found that expressions of 1266 proteins were significantly changed between the experimental groups. Among these proteins were cell division cycle 5-like (Cdc5L) and drebrin-like (DBNL). We then hypothesized that inhibition of the activities of these two proteins may lead to more effective anticancer inhibitor combinations in the presence of proteasomal inhibition. In fact, as presented in the current study, Cdc5L phosphorylation inhibitor CVT-313 and DBNL phosphorylation inhibitor OTSSP167 were highly cytotoxic in 4T1 breast cancer cells and their IC50 values were 20.1 and 43 nM, respectively. Under the same experimental conditions, the IC50 value of BAPTA-AM was found 19.9 μM. Using WST 1 cytotoxicity assay, it was determined that 10 nM bortezomib + 10 nM CVT-313 was more effective than the control, the single treatments, or than 5 nM bortezomib + 5 nM CVT-313. Similarly, 10 nM bortezomib + 10 nM OTSSP167 was more cytotoxic than the control, the monotherapies, 5 nM bortezomib + 5 nM OTSSP167, or than 5 nM bortezomib + 10 nM OTSSP167, indicating that bortezomib + OTSSP167 was also more effective than bortezomib + CVT-313 in a dose-dependent manner. Furthermore, the 3D spheroid model proved that bortezomib + OTSSP167 was more effective than the monotherapies as well as bortezomib + CVT-313 and bortezomib + BAPTA-AM combinations. Finally, the effect of bortezomib + OTSSP167 combination was tested on MDA-MB-231 breast cancer cells, and it similarly determined that 20 nM bortezomib +40 nM OTSSP167 combination completely blocked the formation of 3D spheroids.
Conclusions
Altogether, the results presented here indicate that bortezomib + OTSSP167 is a novel and effective combination and may be tested further for cancer treatment in vivo and in clinical settings.








Similar content being viewed by others
References
Adams J. Development of the proteasome inhibitor PS-341. Oncologist. 2002;7:9–16.
Aras B, Yerlikaya A. Bortezomib and etoposide combinations exert synergistic effects on the human prostate cancer cell line PC-3. Oncol Lett. 2016;11:3179–84.
Bayram D, Çetin ES, Kara M, Özgöçmen M, Candan IA. The apoptotic effects of silibinin on MDA-MB-231 and MCF-7 human breast carcinoma cells. Hum Exp Toxicol. 2017;36:573–86.
Bradford MM. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976;72:248–54.
Chan EY, Sutton JN, Jacobs JM, Bondarenko A, Smith RD, Katze MG. Dynamic host energetics and cytoskeletal proteomes in human immunodeficiency virus type 1-infected human primary CD4 cells: analysis by multiplexed label-free mass spectrometry. J Virol. 2009;83:9283–95.
Chao A, Wang TH. Molecular mechanisms for synergistic effect of proteasome inhibitors with platinum-based therapy in solid tumors. Taiwan J Obstet Gynecol. 2016;55:3–8.
Chen L, Madura K. Increased proteasome activity, ubiquitin-conjugating enzymes, and eEF1A translation factor detected in breast cancer tissue. Cancer Res. 2005;65:5599–606.
Chen LY, Chiang AS, Hung JJ, Hung HI, Lai YK. Thapsigargin-induced grp78 expression is mediated by the increase of cytosolic free calcium in 91 rat brain tumor cells. J Cell Biochem. 2000;78:404–16.
Chung CH, Aulino J, Muldowney NJ, Hatakeyama H, Baumann J, Burkey B, et al. Nuclear factor-kappa B pathway and response in a phase II trial of bortezomib and docetaxel in patients with recurrent and/or metastatic head and neck squamous cell carcinoma. Ann Oncol. 2010;21:864–70.
Chung S, Suzuki H, Miyamoto T, Takamatsu N, Tatsuguchi A, Ueda K, et al. Development of an orally-administrative MELK-targeting inhibitor that suppresses the growth of various types of human cancer. Oncotarget. 2012;3:1629–40.
Ding X, Matsuo K, Xu L, Yang J, Zheng L. Optimized combinations of bortezomib, camptothecin, and doxorubicin show increased efficacy and reduced toxicity in treating oral cancer. Anti-Cancer Drugs. 2015;26:547–54.
Frankland-Searby S, Bhaumik SR. The 26S proteasome complex: an attractive target for cancer therapy. Biochim Biophys Acta. 2012;1825:64–76.
Freshney RI. Cytotoxicity, in culture culture of animal cells: a manual of basic technique. 5th ed. Hoboken: Wiley; 2005. p. 359.
Gräub R, Lancero H, Pedersen A, Chu M, Padmanabhan K, Xu X-Q, et al. Cell cycle-dependent phosphorylation of human CDC5 regulates RNA processing. Cell Cycle. 2008;7:1795–803.
Guarguaglini G, Duncan PI, Stierhof YD, Holmström T, Duensing S, Nigg EA. The forkhead-associated domain protein Cep170 interacts with polo-like kinase 1 and serves as a marker for mature centrioles. Mol Biol Cell. 2005;16:1095–107.
Hambley B, Caimi PF, William BM. Bortezomib for the treatment of mantle cell lymphoma: an update. Ther Adv Hematol. 2016;7:196–208.
Hendershot LM. The ER function BiP is a master regulator of ER function. Mt Sinai J Med. 2004;71:289–97.
Jakobi R, McCarthy CC, Koeppel MA, Stringer DK. Caspase-activated PAK-2 is regulated by subcellular targeting and proteasomal degradation. J Biol Chem. 2003;278:38675–85.
Kane RC, Bross PF, Farrell AT, Pazdur R. Velcade®: U.S. FDA approval for the treatment of multiple myeloma progressing on prior therapy. Oncologist. 2003;8:508–13.
Kane RC, Dagher R, Farrell A, Ko CW, Sridhara R, Justice R, et al. Bortezomib for the treatment of mantle cell lymphoma. Clin Cancer Res. 2007;13:5291–4.
Kozuch PS, Rocha-Lima CM, Dragovich T, Hochster H, O'Neil BH, Atiq OT, et al. Bortezomib with or without irinotecan in relapsed or refractory colorectal cancer: results from a randomized phase II study. J Clin Oncol. 2008;26:2320–6.
Li B, Cheng XL, Yang YP, Li ZQ. GRP78 mediates radiation resistance of a stem cell-like subpopulation within the MCF-7 breast cancer cell line. Oncol Rep. 2013;30:2119–26.
Li Z, Park HR, Shi Z, Li Z, Pham CD, Du Y, et al. Pro-oncogenic function of HIP-55/Drebrin-like (DBNL) through Ser269/Thr291-phospho-sensor motifs. Oncotarget. 2014;5:3197–209.
Löffek S, Wöll S, Höhfeld J, Leube RE, Has C, Bruckner-Tuderman L, et al. The ubiquitin ligase CHIP/STUB1 targets mutant keratins for degradation. Hum Mutat. 2010;31:466–76.
Lundberg KC, Fritz Y, Johnston A, Foster AM, Baliwag J, Gudjonsson JE, et al. Proteomics of skin proteins in psoriasis: from discovery and verification in a mouse model to confirmation in humans. Mol Cell Proteomics. 2015;14:109–19.
Neubert H, Bonnert TP, Rumpel K, Hunt BT, Henle ES, James IT. Label-free detection of differential protein expression by LC/MALDI mass spectrometry. J Proteome Res. 2008;7:2270–9.
Orlowski RZ, Kuhn DJ. Proteasome inhibitors in cancer therapy: lessons from the first decade. Clin Cancer Res. 2008;14:1649–57.
Qiu H, Zhang X, Ni W, Shi W, Fan H, Xu J, et al. Expression and clinical role of Cdc5L as a novel cell cycle protein in hepatocellular carcinoma. Dig Dis Sci. 2016a;61:795–805.
Qiu H, Zhang X, Ni W, Shi W, Fan H, Xu J, et al. Expression and clinical role of Cdc5L as a novel cell cycle protein in hepatocellular carcinoma. Dig Dis Sci. 2016b;61:795–805.
Schlatzer DM, Sugalski J, Dazard JE, Chance MR, Anthony DD. A quantitative proteomic approach for detecting protein profiles of activated human myeloid dendritic cells. J Immunol Methods. 2012;375:39–45.
Shimada Y, Nishimura E, Hoshina H, Kobayashi H, Higuchi T, Eto Y, et al. Proteasome inhibitor bortezomib enhances the activity of multiple mutant forms of lysosomal α-glucosidase in pompe disease. JIMD Rep. 2015;18:33–9.
Sirenko O, Mitlo T, Hesley J, Luke S, Owens W, Cromwell EF. High-content assays for characterizing the viability and morphology of 3D cancer spheroid cultures. Assay Drug Dev Technol. 2015;13:402–14.
Tomechko SE, Liu G, Tao M, Schlatzer D, Powell CT, Gupta S, et al. Tissue specific dysregulated protein subnetworks in type 2 diabetic bladder urothelium and detrusor muscle. Mol Cell Proteomics. 2015;14:635–45.
Wang H, Song X, Logsdon C, Zhou G, Evans DB, Abbruzzese JL, et al. Proteasome-mediated degradation and functions of hematopoietic progenitor kinase 1 in pancreatic cancer. Cancer Res. 2009;69:1063–70.
Wiśniewski JR, Zougman A, Nagaraj N, Mann M. Universal sample preparation method for proteome analysis. Nat Methods. 2009;6:359–62.
Yerlikaya A, Erin N. Differential sensitivity of breast cancer and melanoma cells to proteasome inhibitor velcade. Int J Mol Med. 2008;22:817–23.
Yerlikaya A, Yöntem M. The significance of ubiquitin proteasome pathway in cancer development. Recent Pat Anticancer Drug Discov. 2013;8:298–309.
Yerlikaya A, Erdoğan E, Okur E, Yerlikaya Ş, Savran B. A novel combination treatment for breast cancer cells involving BAPTA-AM and proteasome inhibitor bortezomib. Oncol Lett. 2016;12:323–30.
Zhang Y, Tseng CC, Tsai YL, Fu X, Schiff R, Lee AS. Cancer cells resistant to therapy promote cell surface relocalization of GRP78 which complexes with PI3K and enhances PI(3,4,5)P3 production. PLoS One. 2013;8:e80071.
Acknowledgments
This study was supported in part by Scientific and Research Council of Turkey (Grant No. 113S400).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
All authors declare that they have no conflicts of interest.
Electronic supplementary material
Supplemental Figure 1
Graphical presentation of selected proteins significantly upregulated in response to bortezomib + BAPTA-AM. The expression level of were identified by label-free LC-MS/MS. Con, control; Bor, 10 nM bortezomib; BAPTA, 5 μM BAPTA-AM. (DOCX 242 kb)
Supplemental Table 1
Significantly upregulated or downregulated proteins in response to bortezomib + BAPTA-AM. The analyzes were carried out by label-free LC-MS/MS. (XLSX 227 kb)
Rights and permissions
About this article
Cite this article
Okur, E., Yerlikaya, A. A novel and effective inhibitor combination involving bortezomib and OTSSP167 for breast cancer cells in light of label-free proteomic analysis. Cell Biol Toxicol 35, 33–47 (2019). https://doi.org/10.1007/s10565-018-9435-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10565-018-9435-z