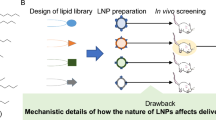
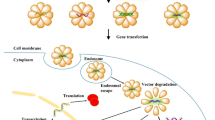
Abstract
DNA formulated into aggregates with polycationic reagents are referred to by a variety of terms including non-viral vectors, synthetic vectors, lipoplexes, polyplexes and more recently nanoparticles. The capacity for delivery of multiple genes, genomic-sized constructs and siRNA delivery, with a diversity of possible formulations, as well as the possibilities of improved efficiency of in vivo gene deliveries, means that nanoparticles, or nanocomplexes to reflect self-assembling systems, will be investigated with increasing vigour in the coming years. This review briefly outlines the applications and challenges for nanoparticle technologies in the field of gene therapy then focuses on the development of a specific kind of formulation, receptor-targeted nanocomplex (RTN), that we have found to be particularly useful in our gene therapy research. An overriding guiding concept that has emerged in the development of synthetic nanodelivery systems is the idea to develop formulations and structures that mimic viruses, whilst retaining the safety elements of synthetic, non-viral systems. RTNs have been optimised and developed for airway epithelial transfection, leading towards gene therapy for cystic fibrosis and for vascular transfection in vein grafts used in bypass surgery. The modular design of the RTN platform further allows for the testing of specific hypotheses relating to the structure and functional role of components in the formation of stable particles and in the transfection pathway, leading to their ultimate disassembly in the nucleus.


Similar content being viewed by others
Abbreviations
- RTN:
-
Receptor-targeted nanocomplex
- siRNA:
-
Short interfering RNA
- RNAi:
-
RNA interference
- SCID X1:
-
Severe combined immunodeficiency disease type X1
- bp:
-
Base pairs
- kb:
-
Kilobase pairs
- nt:
-
Nucleotides
- DOTMA:
-
1,2-Di-((Z)-octadec-9-enyloxy)-N,N,N-trimethylammonium propane chloride
- DOPE:
-
Dioleoyl phosphatidylethanolamine
References
Alton EW, Stern M, Farley R, Jaffe A, Chadwick SL, Phillips J, et al. Cationic lipid-mediated CFTR gene transfer to the lungs and nose of patients with cystic fibrosis: a double-blind placebo-controlled trial. Lancet. 1999;353:947–54.
Ambegia E, Ansell S, Cullis P, Heyes J, Palmer L, Maclachlan I. Stabilized plasmid-lipid particles containing PEG-diacylglycerols exhibit extended circulation lifetimes and tumor selective gene expression. Biochim Biophys Acta. 2005;1669:155–63.
Bai M, Harfe B, Freimuth P. Mutations that alter an Arg-Gly-Asp (RGD) sequence in the adenovirus type 2 penton base protein abolish its cell-rounding activity and delay virus reproduction in flat cells. J Virol. 1993;67:5198–205.
Bainbridge JW, Smith AJ, Barker SS, Robbie S, Henderson R, Balaggan K, et al. Effect of gene therapy on visual function in Leber's congenital amaurosis. N Engl J Med. 2008;358:2231–9.
Barker SE, Grosse SM, Siapati EK, Kritz A, Kinnon C, Thrasher AJ, et al. Immunotherapy for neuroblastoma using syngeneic fibroblasts transfected with IL-2 and IL-12. Br J Cancer. 2007;97:210–7.
Bourassa MG. Fate of venous grafts: the past, the present and the future. J Am Coll Cardiol. 1991;17:1081–3.
Caplen NJ, Alton EWFW, Middleton PG, Dorin JR, Stevenson BJ, Gao X, et al. Liposome-mediated CFTR gene transfer to the nasal epithelium of patients with cystic fibrosis. Nat Med. 1995;1:39–46.
Carmona S, Jorgensen MR, Kolli S, Crowther C, Salazar FH, Marion PL, et al. Controlling HBV replication in vivo by intravenous administration of triggered PEGylated siRNA-nanoparticles. Mol Pharm. 2009;6:706–17.
Cavazzana-Calvo M, Hacein-Bey S, De Saint Basile G, Gross F, Yvon E, Nusbaum P, et al. Gene therapy of human severe combined immunodeficiency (SCID)-X1 disease. Science. 2000;288:669–72.
Cideciyan AV, Aleman TS, Boye SL, Schwartz SB, Kaushal S, Roman AJ, et al. Human gene therapy for RPE65 isomerase deficiency activates the retinoid cycle of vision but with slow rod kinetics. Proc Natl Acad Sci U S A. 2008;105:15112–7.
Compton SH, Mecklenbeck S, Mejia JE, Hart SL, Rice M, Cervini R, et al. Stable integration of large (>100 kb) PAC constructs in HaCaT keratinocyted using an integrin-targeting peptide delivery system. Gene Ther. 2000;7:1600–5.
Cullis PR, Chonn A, Semple SC. Interactions of liposomes and lipid-based carrier systems with blood proteins: relation to clearance behaviour in vivo. Adv Drug Deliv Rev. 1998;32:3–17.
Cunningham S, Meng QH, Klein N, Mcanulty RJ, Hart SL. Evaluation of a porcine model for pulmonary gene transfer using a novel synthetic vector. J Gene Med. 2002;4:438–46.
Dean DA, Byrd JNJ, Dean BS. Nuclear targeting of plasmid DNA in human corneal cells. Curr Eye Res. 1999;19:66–75.
Driessen WH, Fujii N, Tamamura H, Sullivan SM. Development of peptide-targeted lipoplexes to CXCR4-expressing rat glioma cells and rat proliferating endothelial cells. Mol Ther. 2008;16:516–24.
Eagle KA, Guyton RA, Davidoff R, Ewy GA, Fonger J, Gardner TJ, et al. ACC/AHA guidelines for coronary artery bypass graft surgery: executive summary and recommendations: a report of the American college of cardiology/American heart association task force on practice guidelines (Committee to Revise the 1991 Guidelines for Coronary Artery Bypass Graft Surgery). Circulation. 1999;100:1464–80.
Felgner PL, Gadek TR, Holm M, Roman R, Chan HW, Wenz M, et al. Lipofection: a highly efficient, lipid-mediated DNA-transfection procedure. PNAS. 1987;84:7413–7.
Fenske DB, Maclachlan I, Cullis PR. Long-circulating vectors for the systemic delivery of genes. Curr Opin Mol Ther. 2001;3:153–8.
Galán JE, Ginocchio C, Costeas P. Molecular and functional characterization of the Salmonella invasion gene invA: homology of invA to members of a new protein family. J Bacteriol. 1992;174:4338–49.
Gary DJ, Puri N, Won YY. Polymer-based siRNA delivery: perspectives on the fundamental and phenomenological distinctions from polymer-based DNA delivery. J Control Release. 2007;121:64–73.
Gaspar HB, Parsley KL, Howe S, King D, Gilmour KC, Sinclair J, et al. Gene therapy of X-linked severe combined immunodeficiency by use of a pseudotyped gammaretroviral vector. Lancet. 2004;364:2181–7.
Gill DR, Southern KW, Mofford KA, Seddon T, Huang L, Sorgi F, et al. A placebo-controlled study of liposome-mediated gene transfer to the nasal epithelium of patients with cystic fibrosis. Gene Ther. 1997;4:199–209.
Grayson AC, Doody AM, Putnam D. Biophysical and structural characterization of polyethylenimine-mediated siRNA delivery in vitro. Pharm Res. 2006;23:1868–76.
Greve JM, Davis G, Meyer AM, Forte CP, Yost SC, Marlor CW, et al. The major human rhinovirus receptor is ICAM-1. Cell. 1989;56:839–47.
Hacein-Bey-Abina S, Von Kalle C, Schmidt M, Le Deist F, Wulffraat N, Mcintyre E, et al. A serious adverse event after successful gene therapy for X-linked severe combined immunodeficiency. N Engl J Med. 2003a;348:255–6.
Hacein-Bey-Abina S, Von Kalle C, Schmidt M, Mccormack MP, Wulffraat N, Leboulch P, et al. LMO2-associated clonal T cell proliferation in two patients after gene therapy for SCID-X1. Science. 2003b;302:415–9.
Harbottle RP, Cooper RG, Hart SL, Ladhoff A, Mckay T, Knight AM, et al. An RGD-oligolysine peptide: a prototype construct for integrin-mediated gene delivery [see comments]. Hum Gene Ther. 1998;9:1037–47.
Hart S. Use of adhesion molecules for gene delivery. Exp Nephrol. 1999;7:193–9.
Hart SL, Knight AM, Harbottle RP, Mistry A, Hunger HD, Cutler DF, et al. Cell binding and internalization by filamentous phage displaying a cyclic Arg-Gly-Asp-containing peptide. J Biol Chem. 1994;269:12468–74.
Hart SL, Harbottle RP, Cooper R, Miller A, Williamson R, Coutelle C. Gene delivery and expression mediated by an integrin-binding peptide. Gene Ther. 1995;2:552–4. published erratum appears in Gene Ther 1996;3(11):1032–3.
Hart SL, Collins L, Gustafsson K, Fabre JW. Integrin-mediated transfection with peptides containing arginine–glycine–aspartic acid domains. Gene Ther. 1997;4:1225–30.
Hart SL, Arancibia-Carcamo CV, Wolfert MA, Mailhos C, O'reilly NJ, Ali RR, et al. Lipid-mediated enhancement of transfection by a nonviral integrin- targeting vector. Hum Gene Ther. 1998;9:575–85.
Heyes J, Palmer L, Chan K, Giesbrecht C, Jeffs L, Maclachlan I. Lipid encapsulation enables the effective systemic delivery of polyplex plasmid DNA. Mol Ther. 2007;15:713–20.
Hillis GS, Mlynski RA, Simpson JG, Macleod AM. The expression of beta 1 integrins in human coronary artery. Basic Res Cardiol. 1998;93:295–302.
Howe SJ, Mansour MR, Schwarzwaelder K, Bartholomae C, Hubank M, Kempski H, et al. Insertional mutagenesis combined with acquired somatic mutations causes leukemogenesis following gene therapy of SCID-X1 patients. J Clin Invest. 2008;118:3143–50.
Huang L, Hung M-C, Wagner E. Nonviral vectors for gene therapy, part 1. Amsterdam: Elsevier; 2005.
Hurley CA, Wong JB, Ho J, Writer M, Irvine SA, Lawrence MJ, et al. Mono- and dicationic short PEG and methylene dioxyalkylglycerols for use in synthetic gene delivery systems. Org Biomol Chem. 2008;6:2554–9.
Irvine SA, Meng QH, Afzal F, Ho J, Wong JB, Hailes HC, et al. Receptor-targeted nanocomplexes optimized for gene transfer to primary vascular cells and explant cultures of rabbit aorta. Mol Ther. 2008;16:508–15.
Isberg RR, Leong JM. Multiple beta 1 chain integrins are receptors for invasin, a protein that promotes bacterial penetration into mammalian cells. Cell. 1990;60:861–71.
Izzat MB, West RR, Bryan AJ, Angelini GD. Coronary artery bypass surgery: current practice in the United Kingdom. Br Heart J. 1994;71:382–5.
Jenkins RG, Herrick SE, Meng QH, Kinnon C, Laurent GJ, Mcanulty RJ, et al. An integrin-targeted non-viral vector for pulmonary gene therapy. Gene Ther. 2000;7:393–400.
Jenkins RG, Meng QH, Hodges RJ, Lee LK, Bottoms SE, Laurent GJ, et al. Formation of LID vector complexes in water alters physicochemical properties and enhances pulmonary gene expression in vivo. Gene Ther. 2003;10:1026–34.
Kamaly N, Kalber T, Thanou M, Bell JD, Miller AD. Folate receptor targeted bimodal liposomes for tumor magnetic resonance imaging. Bioconjug Chem. 2009;20:648–55.
Katas H, Alpar HO. Development and characterisation of chitosan nanoparticles for siRNA delivery. J Control Release. 2006;115:216–25.
Koivunen E, Gay DA, Ruoslahti E. Selection of peptides binding to the a5b1 integrin from phage display library. J Biol Chem. 1993;268:20205–10.
Koivunen E, Wang B, Ruoslahti E. Isolation of a highly specific ligand for the alpha 5 beta 1 integrin from a phage display library. J Cell Biol. 1994;124:373–80.
Koltover I, Salditt T, Radler JO, Safinya CR. An inverted hexagonal phase of cationic liposome–DNA complexes related to DNA release and delivery. Science. 1998;281:78–81.
Kostarelos K, Miller AD. Synthetic, self-assembly ABCD nanoparticles; a structural paradigm for viable synthetic non-viral vectors. Chem Soc Rev. 2005;34:970–94.
Kumar P, Wu H, Mcbride JL, Jung KE, Kim MH, Davidson BL, et al. Transvascular delivery of small interfering RNA to the central nervous system. Nature. 2007;448:39–43.
Laitinen M, Pakkanen T, Donetti E, Baetta R, Luoma J, Lehtolainen P, et al. Gene transfer into the carotid artery using an adventitial collar: comparison of the effectiveness of the plasmid–liposome complexes, retroviruses, pseudotyped retroviruses, and adenoviruses. Hum Gene Ther. 1997;8:1645–50.
Leng Q, Scaria P, Zhu J, Ambulos N, Campbell P, Mixson AJ. Highly branched HK peptides are effective carriers of siRNA. J Gene Med. 2005;7:977–86.
Li SD, Huang L. Surface-modified LPD nanoparticles for tumor targeting. Ann N Y Acad Sci. 2006;1082:1–8.
Liu Y, Huang R, Han L, Ke W, Shao K, Ye L, et al. Brain-targeting gene delivery and cellular internalization mechanisms for modified rabies virus glycoprotein RVG29 nanoparticles. Biomaterials. 2009;30:4195–202.
Maclachlan I, Cullis P. Diffusible-PEG-lipid stabilized plasmid lipid particles. Adv Genet. 2005;53:157–88.
Maguire AM, Simonelli F, Pierce EA, Pugh EN Jr, Mingozzi F, Bennicelli J, et al. Safety and efficacy of gene transfer for Leber's congenital amaurosis. N Engl J Med. 2008;358:2240–8.
Mann MJ. Gene therapy for vein grafts. Curr Cardiol Rep. 2000;2:29–33.
Manno CS, Pierce GF, Arruda VR, Glader B, Ragni M, Rasko JJ, et al. Successful transduction of liver in hemophilia by AAV-Factor IX and limitations imposed by the host immune response. Nat Med. 2006;12:342–7.
Martin ME, Rice KG. Peptide-guided gene delivery. Aaps J. 2007;9:E18–29.
Mason PW, Rieder E, Baxt B. RGD sequence of foot-and-mouth disease virus is essential for infecting cells via the natural receptor but can be bypassed by an antibody-dependent enhancement pathway. PNAS. 1994;91:1932–6.
Meng QH, Robinson D, Jenkins RG, Mcanulty RJ, Hart SL. Efficient transfection of non-proliferating human airway epithelial cells with a synthetic vector system. J Gene Med. 2004;6:210–21.
Meng QH, Jamal W, Hart SL, Mcewan JR. Application to vascular adventitia of a nonviral vector for TIMP-1 gene therapy to prevent intimal hyperplasia. Hum Gene Ther. 2006;17:717–27.
Molas M, Gomez-Valades AG, Vidal-Alabro A, Miguel-Turu M, Bermudez J, Bartrons R, et al. Receptor-mediated gene transfer vectors: progress towards genetic pharmaceuticals. Curr Gene Ther. 2003;3:468–85.
Motwani JG, Topol EJ. Aortocoronary saphenous vein graft disease: pathogenesis, predisposition, and prevention. Circulation. 1998;97:916–31.
Mustapa MF, Bell PC, Hurley CA, Nicol A, Guenin E, Sarkar S, et al. Biophysical characterization of an integrin-targeted lipopolyplex gene delivery vector. Biochemistry. 2007;46:12930–44.
Mustapa MF, Grosse SM, Kudsiova L, Elbs M, Raiber EA, Wong JB, et al. Stabilized integrin-targeting ternary LPD (Lipopolyplex) vectors for gene delivery designed to disassemble within the target cell. Bioconjug Chem. 2009;20:518–32.
Ng QK, Sutton MK, Soonsawad P, Xing L, Cheng H, Segura T. Engineering clustered ligand binding into nonviral vectors: alphavbeta3 targeting as an example. Mol Ther. 2009;17:828–36.
Nicklin SA, White SJ, Watkins SJ, Hawkins RE, Baker AH. Selective targeting of gene transfer to vascular endothelial cells by use of peptides isolated by phage display. Circulation. 2000;102:231–7.
Noone PG, Hohneker KW, Zhou Z, Johnson LG, Foy C, Gipson C, et al. Safety and biological efficacy of a lipid-CFTR complex for gene transfer in the nasal epithelium of adult patients with cystic fibrosis. Mol Ther. 2000;1:105–14.
Oba M, Fukushima S, Kanayama N, Aoyagi K, Nishiyama N, Koyama H, et al. Cyclic RGD peptide-conjugated polyplex micelles as a targetable gene delivery system directed to cells possessing alphavbeta3 and alphavbeta5 integrins. Bioconjug Chem. 2007;18:1415–23.
Parkes R, Meng QH, Elena Siapati K, Mcewan JR, Hart SL. High efficiency transfection of porcine vascular cells in vitro with a synthetic vector system. J Gene Med. 2002;4:292–9.
Patil ML, Zhang M, Betigeri S, Taratula O, He H, Minko T. Surface-modified and internally cationic polyamidoamine dendrimers for efficient siRNA delivery. Bioconjug Chem. 2008;19:1396–403.
Porteous DJ, Dorin JR, Mclachlan G, Davidson-Smith H, Davidson H, Stevenson BJ, et al. Evidence for safety and efficacy of DOTAP cationic liposome mediated CFTR gene transfer to the nasal epithelium of patients with cystic fibrosis. Gene Ther. 1997;4:210–8.
Raper SE, Chirmule N, Lee FS, Wivel NA, Bagg A, Gao GP, et al. Fatal systemic inflammatory response syndrome in a ornithine transcarbamylase deficient patient following adenoviral gene transfer. Mol Genet Metab. 2003;80:148–58.
Siapati KE, Barker S, Kinnon C, Michalski A, Anderson R, Brickell P, et al. Improved antitumour immunity in murine neuroblastoma using a combination of IL-2 and IL-12. Br J Cancer. 2003;88:1641–8.
Sun YX, Zeng X, Meng QF, Zhang XZ, Cheng SX, Zhuo RX. The influence of RGD addition on the gene transfer characteristics of disulfide-containing polyethyleneimine/DNA complexes. Biomaterials. 2008;29:4356–65.
Tagalakis AD, Mcanulty RJ, Devaney J, Bottoms SE, Wong JB, Elbs M, et al. A receptor-targeted nanocomplex vector system optimized for respiratory gene transfer. Mol Ther. 2008;16:907–15.
Uduehi A, Mailhos C, Truman H, Thrasher AJ, Kinnon C, Hart SL. Enhancement of integrin-mediated transfection of haematopoietic cells with a synthetic vector system. Biotechnol Appl Biochem. 2003;38:201–9.
Varga CM, Wickham TJ, Lauffenburger DA. Receptor-mediated targeting of gene delivery vectors: insights from molecular mechanisms for improved vehicle design. Biotechnol Bioeng. 2000;70:593–605.
Varga CM, Hong K, Lauffenburger DA. Quantitative analysis of synthetic gene delivery vector design properties. Mol Ther. 2001;4:438–46.
Wagner E. Application of membrane-active peptides for nonviral gene delivery. Adv Drug Deliv Rev. 1999;38:279–89.
Wagner E. Strategies to improve DNA polyplexes for in vivo gene transfer: will “artificial viruses” be the answer? Pharm Res. 2004;21:8–14.
Wagner E, Culmsee C, Boeckle S. Targeting of polyplexes: toward synthetic virus vector systems. Adv Genet. 2005;53:333–54.
Wang M, Lowik DW, Miller AD, Thanou M. Targeting the urokinase plasminogen activator receptor with synthetic self-assembly nanoparticles. Bioconjug Chem. 2009;20:32–40.
Wells DJ. Gene therapy progress and prospects: electroporation and other physical methods. Gene Ther. 2004;11:1363–9.
Wheeler JJ, Palmer L, Ossanlou M, Maclachlan I, Graham RW, Zhang YP, et al. Stabilized plasmid-lipid particles: construction and characterization. Gene Ther. 1999;6:271–81.
White RE, Wade-Martins R, Hart SL, Frampton J, Huey B, Desai-Mehta A, et al. Functional delivery of large genomic DNA to human cells with a peptide-lipid vector. J Gene Med. 2003;5:883–92.
Writer MJ, Marshall B, Pilkington-Miksa MA, Barker SE, Jacobsen M, Kritz A, et al. Targeted gene delivery to human airway epithelial cells with synthetic vectors incorporating novel targeting peptides selected by phage display. J Drug Target. 2004;12:185–93.
Writer M, Hurley CA, Sarkar S, Copeman DM, Wong JB, Odlyha M, et al. Analysis and optimization of the cationic lipid component of a lipid/peptide vector formulation for enhanced transfection in vitro and in vivo. J Liposome Res. 2006;16:373–89.
Yla-Herttuala S, Martin JF. Cardiovascular gene therapy. Lancet. 2000;355:213–22.
Yu J, Hu K, Smuga-Otto K, Tian S, Stewart R, Slukvin Ii, et al. Human induced pluripotent stem cells free of vector and transgene sequences. Science. 2009;324:797–801.
Yusa K, Rad R, Takeda J, Bradley A. Generation of transgene-free induced pluripotent mouse stem cells by the piggyBac transposon. Nat Methods. 2009;6:363–9.
Zabner J, Cheng SH, Meeker D, Launspach J, Balfour R, Perricone MA, et al. Comparison of DNA–lipid complexes and DNA alone for gene transfer to cystic fibrosis airway epithelia in vivo. J Clin Invest. 1997;100:1529–37.
Acknowledgements
I would like to thank my colleagues and collaborators over the years who contributed to the work on RTNs. In particular, Charles Coutelle and Bob Williamson from the former St. Mary’s Hospital Medical School, London (now Imperial College), Helen Hailes and Alethea Tabor from UCL Department of Chemistry, Jean McEwan from UCL Centre for Cardiovascular Biology and Medicine, Robin McAnulty from UCL Centre for Respiratory Research and Dr. Adam Jaffe from Great Ormond Street Hospital and UCL Institute of Child Health. Also, my Ph.D. students who have contributed to the work including Susie Barker, Elena Siapati, Shahla Salehi, Albert Kwok, Angelika Kritz and Gisli Jenkins and postdocs at ICH including Qing Hai Meng, Aris Tagalakis, Maria Manunta, Michele Writer, Aima Uduehi, Carolina Mailhos and Richard Parkes and Richard Harbottle from Imperial College. Thanks to the grant-funding bodies for supporting this work: Wellcome Trust, BBSRC, EPSRC, Cystic Fibrosis Trust, Sparks and North Bristol NHS Trust. My thanks also go to staff at Genex Biosystems Ltd. for their work on the translational development of RTN formulations.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hart, S.L. Multifunctional nanocomplexes for gene transfer and gene therapy. Cell Biol Toxicol 26, 69–81 (2010). https://doi.org/10.1007/s10565-009-9141-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10565-009-9141-y