Abstract
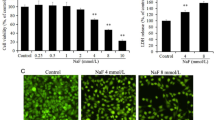
It has been documented that medical prosthetic alloys release metal ions into surrounding tissues and cause cytotoxicity, but the mechanisms remain undefined. In that regard the cellular oxidative stress may be a common pathway in cellular responses to metal ions. The objective of this study was to approach the hypothesis that oxidative stress mediates chromium-induced cytotoxicity in rat calvarial osteoblasts. Osteoblasts were exposed to different concentrations of Cr6+ or Cr3+ (5–20 μM) in the presence or absence of the antioxidant N-acetyl-cysteine (NAC; 1–5 mM). Cellular viability, differentiation, and intracellular ultrastructural alterations were evaluated by MTT assay, alkaline phosphatase (ALP) activity assay, and transmission electron microscopy. Cellular oxidative stress was evaluated by intracellular reactive oxygen species (ROS) production. ROS production was monitored by the oxidation-sensitive fluorescent probe 2′7′-dichlorofluorescin diacetate (DCFH-DA). A time- and concentration- dependent increased cytotoxicity, time-dependent increased intracellular ROS production were indicated on exposure to Cr6+. Pretreatment of osteoblasts with 1–5 mM NAC afforded dose-dependent cytoprotective effects against Cr6+-induced cytotoxicity in osteoblasts. NAC decreased the level of intracellular ROS induced by Cr6+, too. While Cr3+ and NAC did not have any significant effects on osteoblasts (5–20 μM). These results suggest that oxidative stress is involved in Cr6+-induced cytotoxicity in osteoblasts, and NAC can provide protection for osteoblasts against Cr6+-induced oxidative stress. Cr3+ (5–20 μM) have no significant cytotoxicity in osteoblasts based on the results of this study.




Similar content being viewed by others
References
Bagchi D, Stohs SJ, Downs BW, et al. Cytotoxicity and oxidative mechanisms of different forms of chromium. Toxicology 2002;180(1):5–22.
Barreiro E, Galdiz JB, Marinan M, et al. Respiratory loading intensity and diaphragm oxidative stress: N-acetyl-cysteine effects. J Appl Physiol 2006;100(2):555–63.
Bradford MM. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 1976;72:248–54.
Camera E, Picardo M. A nalytical methods to investigate glutathione and related compounds in biological and pathological processes. J Chromatogr B Analyt Technol Biomed Life Sci 2002;781(1–2):181–206.
Chapple ILC. Role of free radicals and antioxidants in the pathogenesis of the inflammatory periodontal diseases. J Clin Pathol: Mol Pathol 1996;49:247–55.
Chapple ILC. Reactive oxygen species and antioxidants in inflammatory diseases. J Clin Periodontol 1997;24:287–96.
Cobb AG, Schmalzreid TP. The clinical significance of metal ion release from cobalt–chromium metal-on-metal hip joint arthroplasty. Proc Inst Mech Eng H 2006;220(2):385–98.
Cuypers A, Vangronsveld J, Clijsters H. The chemical behavior of heavy metals play a prominent role in the induction of oxidative stress. Free Radic Res 1999;31:39–43.
Davies KJA, Pryor WA. The evolution of Free Radical Biology & Medicine: A 20-year history. Free Radic Biol Med 2005;39(10):1263–4.
Dayeh VR, Lynn DH, Bols NC. Cytotoxicity of metals common in mining effluent to rainbow trout cell lines and to the ciliated protozoan, Tetrahymena thermophila. Toxicol in Vitro 2005;19(3):399–410.
Dee KC, Rueger DC, Andersen TT, et al. Conditions which promote mineralization at the bone-implant interface: a model in vitro study. Biomaterials 1996;17(2):209–15.
Elbekai RH, El-Kadi AOS. The role of oxidative stress in the modulation of aryl hydrocarbon receptor-regulated genes by As3+, Cd2+, and Cr6+. Free Radic Biol Med 2005;39(11):1499–511.
Ercal N, Gurer-Orhan H, Aykin-Burns N. Toxic metals and oxidative stress part i: mechanisms involved in metal-induced oxidative damage. Curr Top Med Chem 2001;1(6):529–39.
Garvey BT, Bizios R. A transmission electron microscopy examination of the interface between osteoblasts and metal biomaterials. J Biomed Mater Res 1995;29(8):987–92.
Geissler U, Hempel U, Wolf C, et al. Collagen type I-coating of Ti6Al4V promotes adhesion of osteoblasts. J Biomed Mater Res 2000;51(4):752–60.
Granchi D, Ciapetti G, Savarino L, et al. Assessment of metal extract toxicity on human lymphocytes cultured in vitro. J Biomed Mater Res 1996;31(2):183–91.
Grinberg L, Fibach E, Amer J, et al. N-acetylcysteine amide, a novel cell-permeating thiol, restores cellular glutathione and protects human red blood cells from oxidative stress. Free Radic Biol Med 2005;38(1):136–45.
Halliwell B. Reactive oxygen species in living systems: source, biochemistry, and role in human disease. Am J Med 1991;91(3C):14–22.
Halliwell B, Gutteridge JM. The antioxidants of human extracellular fluids. Arch Biochem Biophys 1990;280(1):1–8.
Hinoi E, Fujimori S, Takemori A, et al. Demonstration of expression of mRNA for particular AMPA and kainate receptor subunits in immature and mature cultured rat calvarial osteoblasts. Brain Res 2002;943(1):112–6.
Holdiness MR. Clinical pharmacokinetics of N-acetylcysteine. Clin Pharmacokinet 1991;20(2):123–34.
Hsu CC, Huang CN, Hung YC, et al. Five cysteine-containing compounds have antioxidative activity in Balb/cA mice. J Nutr 2004;134(1):149–52.
Hultberg B, Andersson A, Isaksson A. Interaction of metals and thiols in cell damage and glutathione distribution: potentiation of mercury toxicity by dithiothreitol. Toxicology 2001;156(2–3):93–100.
Jones AL, Jarvie DR, Simpson D, et al. Pharmacokinetics of N-acetylcysteine are altered in patients with chronic liver disease. Aliment Pharmacol Ther 1997;11(4):787–91.
Kampen AH, Tollersrud T, Lund A. Flow cytometric measurement of neutrophil respiratory burst in whole bovine blood using live Staphylococcus aureus. J Immunol Methods 2004;294(1–2):211.
Kirsch T, Nah HD, Shapiro IM, et al. Regulated production of mineralization-competent matrix vesicles in hypertrophic chondrocytes. J Cell Biol 1997;137(5):1149–60.
Kubo E, Miyazawa T, Fatma N, et al. Development- and age-associated expression pattern of peroxiredoxin 6, and its regulation in murine ocular lens. Mech Ageing Dev 2006;127(3):249–56.
Lee DH, Lim BS, Lee YK, et al. Effects of hydrogen peroxide (H2O2) on alkaline phosphatase activity and matrix mineralization of odontoblast and osteoblast and osteoblast cell lines. Cell Biol Toxicol 2006;22:39–46.
Lin HY, Bumgardner JD. In vitro biocorrosion of Co–Cr–Mo implant alloy by macrophage cells. J Orthop Res 2004;22(6):1231–6.
Lopez-Alias JF, Martinez-Gomis J, Anglada JM, et al. Ion release from dental casting alloys as assessed by a continuous flow system: Nutritional and toxicological implications. Dent Mater 2006;22(9):832–7.
MacKenzie GG, Zago MP, Erlejman AG, et al. Alpha-lipoic acid and N-acetyl cysteine prevent zinc deficiency-induced activation of NF-kappa B and AP-1 transcription factors in human neuroblastoma IMR-32 cells. Free Radic Res 2006;40(1):75–84.
Mantell LL. Introduction to serial reviews on redox signaling in immune function and cellular responses in lung injury and diseases. Free Radic Biol Med 2006;41(1):1–3.
Messer J, Reynolds M, Stoddard L, et al. Causes of DNA single-strand breaks during reduction of chromate by glutathione in vitro and in cells. Free Radic Biol Med 2006;40:1981–1992.
Messer RLW, Bishop S, Lucas LC. Effects of metallic ion toxicity on human gingival fibroblasts morphology. Biomaterials 1999;20(18):1647–57.
Messer RLW, Doeller JE, Kraus DW, et al. An investigation of fibroblast mitochondria enzyme activity and respiration in response to metal ions released from dental alloys. J Biomed Mater Res 2000;50(4):598–604.
Mittoo S. Combinatorial library synthesis of antioxidant compounds. Comb Chem High Throughput Screen 2006;9(6):421–3.
Mohamadin AM, Abdel-Naim AB. Chloroacetonitrile-induced toxicity and oxidative stress in rat gastric epithelial cells. Pharmacol Res 1999;40(4):377–83.
Okazaki Y, Gotoh E. Comparison of metal release from various metallic biomaterials in vitro. Biomaterials 2005;26(1):11–21.
Okazaki Y, Nishimura E. Corrosion resistance of dental alloys in pseudo-oral environment. Mater Trans JIM 2001;42 (2):350–5.
Park YH, Han DW, Suh H, et al. Protective effects of green tea polyphenol against reactive oxygen species induced oxidative stress in cultured rat calvarial osteoblast. Cell Biol Toxicol 2003;19(5):325–37.
Patierno SR, Banh D, Landolph JR. Transformation of C3H/10T1/2 mouse embryo cells to focus formation and anchorage independence by insoluble lead chromate but not soluble calcium chromate: relationship to mutagenesis and internalization of lead chromate particles. Cancer Res 1988;48(18):5280–8
Quievryn G, Peterson E, Messer J, et al. Genotoxicity and mutagenicity of chromium(VI)/ascorbate-generated DNA adducts in human and bacterial cells. Biochemistry 2003;42(4):1062–70.
Ramudo L, Manso MA, Vicente S, et al. Pro-and anti-inflammatory response of acinar cells during acute pancreatitis. Effect of N-acetyl cysteine. Cytokine 2005;32(3–4):125–31.
Reclaru L, Eschler PY, Lerf R, et al. Electrochemical corrosion and metal ion release from Co–Cr–Mo prosthesis with titanium plasma spray coating. Biomaterials 2005;26(23):4747–56.
Rudolf E, Cervinka M, Cerman J, et al. Hexavalent chromium disrupts the actin cytoskeleton and induces mitochondria-dependent apoptosis in human dermal fibroblasts. Toxicol in Vitro 2005;19(6):713–23.
Rudolf E, Cervinka M. The role of intracellular zinc in chromium(VI)-induced oxidative stress, DNA damage and apoptosis. Chem-Biol Interact 2006;162(3):212–27.
Schmalz G, Garhammer P. Biological interactions of dental cast alloys with oral tissues. Dent Mater 2002;18(5):396–406.
Shi H, Hudson LG, Liu KJ. Serial Review: Redox-active Metal Ions, Reactive Oxygen Species, and Apoptosis. Free Radic Biol Med 2004;37(5):582–93.
Spagnuolo G, Annunzizta M, Rengo S. Cytotoxicity and oxidative stress caused by dental adhesive systems cured with halogen and LED lights. Clin Oral Investig 2004;8:81–5.
Stohs SJ, Bagchi D. Oxidative mechanisms in the toxicity of metal ions. Free Radic Biol Med 1993;18:321–36.
Utting JC, Robins SP, Brandao-Burch A, et al. Hypoxia inhibits the growth, differentiation and bone-forming capacity of rat osteoblasts. Exp Cell Res 2006;312(10):1693–702.
Vandana S, Ram S, Ilavazhagan M, et al. Comparative cytoprotective activity of vitamin C, E and beta-carotene against chromium induced oxidative stress in murine macrophages. Biomed Pharmacother 2006;60(2):71–6.
Wang ML, Hauschka PV, Tuan RS, et al. Exposure to particles stimulates superoxide production by human THP-1 macrophages and avian HD-11EM osteoclasts activated by tumor necrosis factor-αand PMA. J Arthroplast 2002;17(3):335–46.
Wataha JC, Lewis JB, Lockwood PE, et al. Effect of dental metal ion on glutathione levels in THP-1 human monocytes. J Oral Rehabil 2000;27(6):508–16.
Wataha JC, Lockwood PE. Release of elements from dental casting alloys into cell-culture medium over 10 months. Dent Mater 1998;14(2):158–63.
Yang HC, Pon LA. Toxicity of Metal Ions Used in Dental Alloys: A Study in the Yeast Saccharmyces cerevisiae. Drug Chem Toxicol 2003;26(2):75–85.
Yang YZ, Glover R, Ong JL. Fibronectin adsorption on titanium surfaces and its effect on osteoblast precursor cell attachment. Colloids Surf B Biointerfaces 2003;30(4):291–7.
Zhitkovich A, Song Y, Quievryn G, et al. Non-oxidative mechanisms are responsible for the induction of mutagenesis by reduction of Cr(VI) with cysteine: Role of ternary DNA adducts in Cr(III)-dependent mutagenesis. Biochemistry 2001;40(2):549–60.
Acknowledgements
This study was supported by the Chinese National Science Foundation (Fund number 30271428) and the Distinguished Young Teacher Plan Fund (Education Bureau grant number 2002-40).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fu, J., Liang, X., Chen, Y. et al. Oxidative stress as a component of chromium-induced cytotoxicity in rat calvarial osteoblasts. Cell Biol Toxicol 24, 201–212 (2008). https://doi.org/10.1007/s10565-007-9029-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10565-007-9029-7