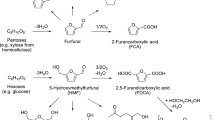
Abstract
Catalytic conversion processes of furfural to 1,5- and 1,2-pentanediols are reviewed. There are two reaction paths. One is the combination of total hydrogenation of furfural to tetrahydrofurfuryl alcohol (THFA) and selective hydrogenolysis of THFA to 1,5-pentanediol. The former reaction is catalyzed by noble metal catalysts such as Pd-based ones at low temperature (≤313 K), and the latter reaction is catalyzed by M–M′Ox catalysts (M = Rh or Ir; M′ = V, Mo, W or Re) at higher temperature (≥373 K). Another path has the ring-opening of furfuryl alcohol as a key step, and noble metal catalysts with basic supports such as hydrotalcite are effective. 1,2-Pentanediol can be produced via this path.








Similar content being viewed by others
References
Bozell JJ, Petersen GR (2010) Green Chem 12:539
Lange JP, van der Heide E, van Buijtenen J, Price R (2012) ChemSusChem 5:150
Nakagawa Y, Liu S, Tamura M, Tomishige K (2015) ChemSusChem 8:1114
Nakagawa Y, Tamura M, Tomishige K (2013) ACS Catal 3:2655
Nakagawa Y, Tomishige K (2011) Catal Surv Asia 15:111
Nakagawa Y, Tomishige K (2012) Catal Today 195:136
Koso S, Furikado I, Shimao A, Miyazawa T, Kunimori K, Tomishige K (2009) Chem Commun 2035
Koso S, Ueda N, Shinmi Y, Okumura K, Kizuka T, Tomishige K (2009) J Catal 267:89
Chen K, Koso S, Kubota T, Nakagawa Y, Tomishige K (2010) ChemCatChem 2:547
Chia M, Pagán-Torres YJ, Hibbitts D, Tan Q, Pham HN, Datye AK, Neurock M, Davis RJ, Dumesic JA (2011) J Am Chem Soc 133:12675
Chen K, Mori K, Watanabe H, Nakagawa Y, Tomishige K (2012) J Catal 294:171
Pholjaroen B, Li N, Huang Y, Li L, Wang A, Zhang T (2015) Catal Today 245:93
Wang Z, Pholjaroen B, Li M, Dong W, Li N, Wang A, Wang X, Cong Y, Zhang T (2014) J Energy Chem 23:427
Guan J, Peng G, Cao Q, Mu X (2014) J Phys Chem C 118:25555
Buntara T, Noel S, Phua PH, Melián-Cabrera I, de Vries JG, Heeres HJ (2012) Top Catal 55:612
Buntara T, Melián-Cabrera I, Tan Q, Fierro JLG, Neurock M, de Vries JG, Heeres HJ (2013) Catal Today 210:106
Koso S, Watanabe H, Okumura K, Nakagawa Y, Tomishige K (2012) Appl Catal B 111–112:27
Amada Y, Shinmi Y, Koso S, Kubota T, Nakagawa Y, Tomishige K (2011) Appl Catal B 105:117
Koso S, Watanabe H, Okumura K, Nakagawa Y, Tomishige K (2012) J Phys Chem C 116:3079
Amada Y, Watanabe H, Tamura M, Nakagawa Y, Okumura K, Tomishige K (2012) J Phys Chem C 116:23503
Nakagawa Y, Ning X, Amada Y, Tomishige K (2012) Appl Catal A 433–434:128
Nakagawa Y, Mori K, Chen K, Amada Y, Tamura M, Tomishige K (2013) Appl Catal A 468:418
Tomishige K, Tamura M, Nakagawa Y (2014) Chem Rec 14:1041
Chatterjee M, Kawanami H, Ishizaka T, Sato M, Suzuki T, Suzuki A (2011) Catal Sci Technol 1:1466
Rodiansono, Khairi S, Hara T, Ichikuni N, Shimazu S (2012) Catal Sci Technol 2:2139
Nakagawa Y, Tomishige K (2010) Catal Commun 12:154
Nakagawa Y, Nakazawa H, Watanabe H, Tomishige K (2012) ChemCatChem 4:1791
Chen B, Li F, Huang Z, Yuan G (2015) Appl Catal A 500:23
Merat N, Godawa C, Gaset A (1990) J Chem Technol Biotechnol 48:145
Biradar NS, Hengne AM, Birajdar SN, Niphadkar PS, Joshi PN, Rode CV (2014) ACS Sustain Chem Eng 2:272
Alamillo R, Tucker M, Chia M, Pagán-Torres Y, Dumesic J (2012) Green Chem 14:1413
Nakagawa Y, Takada K, Tamura M, Tomishige K (2014) ACS Catal 4:2718
Tamura M, Tokonami K, Nakagawa Y, Tomishige K (2013) Chem Commun 49:7034
Liu S, Amada Y, Tamura M, Nakagawa Y, Tomishige K (2014) Green Chem 16:617
Liu S, Amada Y, Tamura M, Nakagawa Y, Tomishige K (2014) Catal Sci Technol 4:2535
Adkins H, Connor R (1932) J Am Chem Soc 53:1091
Xu W, Wang H, Liu X, Ren J, Wang Y, Lu G (2011) Chem Commun 47:3924
Mizugaki T, Yamakawa T, Nagatsu Y, Maeno Z, Mitsudome T, Jitsukawa K, Kaneda K (2014) ACS Sustain Chem Eng 2:2243
Koch O, Köckritz A, Kant M, Martin A, Schöning, A, Armbruster U, Bartoszek M, Evert S, Lange B, Bienert R (2014) US Patent 8,921,617
Wei W, Zhu H, Zhao C, Huang M, Jiang Y (2004) React Funct Polym 59:33
Bhogeswararao S, Srinivas D (2015) J Catal 327:65
Taylor MJ, Durndell LJ, Isaacs MA, Parlett CMA, Wilson K, Lee AF, Kyriakou G (2016) Appl Catal B 180:580
Zhang B, Zhu Y, Ding G, Zheng H, Li Y (2012) Green Chem 14:3402
Omeis M, Neumann M, Brehme V, Theis C, Wolf D, Claus P, Lucas M, Eckert R (2014) US Patent Application 2014/0243562
Hronec M, Fulajtarová K (2012) Catal Commun 24:100
Hronec M, Fulajtarová K, Liptaj T (2012) Appl Catal A 437–438:104
Guo J, Xu G, Han Z, Zhang Y, Fu Y, Guo Q (2014) ACS Sustain Chem Eng 2:2259
Zhou M, Zhu H, Niu L, Xiao G, Xiao R (2014) Catal Lett 144:235
Yang Y, Du Z, Huang Y, Lu F, Wang F, Gao J, Xu J (2013) Green Chem 15:1932
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Nakagawa, Y., Tamura, M. & Tomishige, K. Catalytic Conversions of Furfural to Pentanediols. Catal Surv Asia 19, 249–256 (2015). https://doi.org/10.1007/s10563-015-9194-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10563-015-9194-2