Abstract
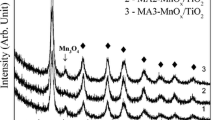
Manganese oxide catalysts having pure hausmanite (Mn3O4) phase but different morphologies were synthesized using three different facile methods namely precipitation method (MPM), reduction method (MRM) and solution combustion method (MSM). In spite of having the same phase, their catalytic activities showed strong dependence on their morphologies. Mn3O4 synthesized using the reduction method had a 3D framework of interconnected hexagonal crystallites. Consequently, it showed the best activity among the synthesized and commercial Mn3O4 catalysts both for carbon monoxide and toluene oxidation. This superior activity of MRM was correlated to its uniform pores and ability to desorb oxygen adspecies easily due to its morphology. The catalytic oxidation activity of this catalyst was not significantly affected at high space velocities. Thermal stability of the best performing catalyst was demonstrated through its stable cyclic performance, both for carbon monoxide and toluene oxidation.
Graphic Abstract















Similar content being viewed by others
References
Ojala S, Pitkäaho S, Laitinen T, Niskala Koivikko N, Brahmi R, Gaálová J, Matejova L, Kucherov A, Päivärinta S, Hirschmann C, Nevanperä T, Riihimäki M, Pirilä M, Keiski RL (2011) Catalysis in VOC Abatement. Top Catal 54:1224–1256. https://doi.org/10.1007/s11244-011-9747-1
Everaert K, Baeyens J (2004) Catalytic combustion of volatile organic compounds. J Hazard Mater 109:113–139. https://doi.org/10.1016/j.jhazmat.2004.03.019
Huang H, Xu Y, Feng Q, Leung DYC (2015) Low temperature catalytic oxidation of volatile organic compounds: a review. Catal Sci Technol 5:2649–2669. https://doi.org/10.1039/c4cy01733a
Kamal MS, Razzak SA, Hossain MM (2016) Catalytic oxidation of volatile organic compounds (VOCs): a review. Atmos Environ 140:117–134. https://doi.org/10.1016/j.atmosenv.2016.05.031
Liotta LF (2010) Catalytic oxidation of volatile organic compounds on supported noble metals. Appl Catal B 100:403–412. https://doi.org/10.1016/j.apcatb.2010.08.023
Brock SL, Duan N, Tian ZR, Giraldo O, Zhou H, Suib SL (1998) A review of porous manganese oxide materials. Chem Mater 10:2619–2628. https://doi.org/10.1021/cm980227h
Solsona B, García T, Sanchis R, Soriano MD, Moreno M, Rodríguez-Castellón E, Agouram S, Dejoz A, López Nieto JM (2016) Total oxidation of VOCs on mesoporous iron oxide catalysts: soft chemistry route versus hard template method. Chem Eng J 290:273–281. https://doi.org/10.1016/j.cej.2015.12.109
Li WB, Wang JX, Gong H (2009) Catalytic combustion of VOCs on non-noble metal catalysts. Catal Today 148:81–87. https://doi.org/10.1016/j.cattod.2009.03.007
Einaga H, Hyodo S, Teraoka Y (2010) Complete oxidation of benzene over perovskite-type oxide catalysts. Top Catal 53:629–634. https://doi.org/10.1007/s11244-010-9497-5
Mo S, Li S, Li W, Li J, Chen J, Chen Y (2016) Excellent low temperature performance for total benzene oxidation over mesoporous CoMnAl composited oxides from hydrotalcites. J Mater Chem A 4:8113–8122. https://doi.org/10.1039/c6ta02593e
Alifanti M, Florea M, Pârvulescu VI (2007) Ceria-based oxides as supports for LaCoO3 perovskite; catalysts for total oxidation of VOC. Appl Catal B 70:400–405. https://doi.org/10.1016/j.apcatb.2005.10.037
Xia Y, Dai H, Jiang H, Zhang L (2010) Three-dimensional ordered mesoporous cobalt oxides: highly active catalysts for the oxidation of toluene and methanol. Catal Commun 11:1171–1175. https://doi.org/10.1016/j.catcom.2010.07.005
Yan Q, Li X, Zhao Q, Chen G (2012) Shape-controlled fabrication of the porous Co3O4 nanoflower clusters for efficient catalytic oxidation of gaseous toluene. J Hazard Mater 209–210:385–391. https://doi.org/10.1016/j.jhazmat.2012.01.039
Xia Y, Dai H, Jiang H, Deng J, He H, Au CT (2009) Mesoporous chromia with ordered three-dimensional structures for the complete oxidation of toluene and ethyl acetate. Environ Sci Technol 43:8355–8360. https://doi.org/10.1021/es901908k
Wu G, Gao Y, Ma F, Zheng B, Liu L, Sun H, Wu W (2015) Catalytic oxidation of benzyl alcohol over manganese oxide supported on MCM-41 zeolite. Chem Eng J 271:14–22. https://doi.org/10.1016/j.cej.2015.01.119
Zhang J, Li Y, Wang L, Zhang C, He H (2015) Catalytic oxidation of formaldehyde over manganese oxides with different crystal structures. Catal Sci Technol 5:2305–2313. https://doi.org/10.1039/c4cy01461h
Lahousse C, Bernier A, Grange P, Delmon B, Papaefthimiou P, Ioannides T, Verykios X (1998) Evaluation of γ-MnO2 as a VOC removal catalyst: comparison with a noble metal catalyst. J Catal 178:214–225. https://doi.org/10.1006/jcat.1998.2148
Aguero FN, Scian A, Barbero BP, Cadús LE (2008) Influence of the support treatment on the behavior of MnOx/Al2O3 catalysts used in VOC combustion. Catal Lett 128:268–280. https://doi.org/10.1007/s10562-008-9695-y
Santos VP, Pereira MFR, Órfão JJM, Figueiredo JL (2009) Synthesis and characterization of manganese oxide catalysts for the total oxidation of ethyl acetate. Top Catal 52:470–481. https://doi.org/10.1007/s11244-009-9187-3
Garcia T, Sellick D, Varela F, Vázquez I, Dejoz A, Agouram S, Taylor SH, Solsona B (2013) Total oxidation of naphthalene using bulk manganese oxide catalysts. Appl Catal A 450:169–177. https://doi.org/10.1016/j.apcata.2012.10.029
Delimaris D, Ioannides T (2008) VOC oxidation over MnOx–CeO2 catalysts prepared by a combustion method. Appl Catal B 84:303–312. https://doi.org/10.1016/j.apcatb.2008.04.006
Averlant R, Royer S, Giraudon J-M, Bellat J-P, Bezverkhyy I, Weber G, Lamonier J-F (2013) Mesoporous silica-confined manganese oxide nanoparticles as highly efficient catalysts for the low-temperature elimination of formaldehyde. ChemCatChem 6:152–161. https://doi.org/10.1002/cctc.201300544
Kang M, Park ED, Kim JM, Yie JE (2007) Manganese oxide catalysts for NOx reduction with NH3 at low temperatures. Appl Catal A 327:261–269. https://doi.org/10.1016/j.apcata.2007.05.024
Kim SC, Shim WG (2010) Catalytic combustion of VOCs over a series of manganese oxide catalysts. Appl Catal B 98:180–185. https://doi.org/10.1016/j.apcatb.2010.05.027
Kapteijn F, Singoredjo L, Andreini A, Moulijn JA (1994) Activity and selectivity of pure manganese oxides in the selective catalytic reduction of nitric oxide with ammonia. Appl Catal B 3:173–189. https://doi.org/10.1016/0926-3373(93)e0034-9
Hu Z, Chen D, Dong J, Li Q, Chen Z, Yin D, Zhao B, Wu CML, Shek C-H (2016) Retracted article: facile synthesis of hierarchical mn3o4 superstructures and efficient catalytic performance. Phys Chem Chem Phys 18:26602–26608. https://doi.org/10.1039/c6cp05004b
Bastos SST, Órfão JJM, Freitas MMA, Pereira MFR, Figueiredo JL (2009) Manganese oxide catalysts synthesized by exotemplating for the total oxidation of ethanol. Appl Catal B 93:30–37. https://doi.org/10.1016/j.apcatb.2009.09.009
Skliri E, Papadogiorgakis S, Lykakis IN, Armatas GS (2016) Mesoporous assembled Mn3O4 nanoparticle networks as efficient catalysts for selective oxidation of alkenes and aryl alkanes. ChemPlusChem 82:136–143. https://doi.org/10.1002/cplu.201600460
Li J, Li L, Cheng W, Wu F, Lu X, Li Z (2014) Controlled synthesis of diverse manganese oxide-based catalysts for complete oxidation of toluene and carbon monoxide. Chem Eng J 244:59–67. https://doi.org/10.1016/j.cej.2014.01.041
Liang S, Teng F, Bulgan G, Zong R, Zhu Y (2008) Effect of phase structure of MnO2 nanorod catalyst on the activity for CO oxidation. J Phys Chem C 112:5307–5315. https://doi.org/10.1021/jp0774995
Xu J, Deng Y-Q, Zhang X-M, Luo Y, Mao W, Yang X-J, Ouyang L, Tian P, Han Y-F (2014) Preparation, characterization, and kinetic study of a core-shell Mn3O4@SiO2 nanostructure catalyst for CO oxidation. ACS Catal 4:4106–4115. https://doi.org/10.1021/cs5011376
Chen H, He J, Zhang C, He H (2007) Self-assembly of novel mesoporous manganese oxide nanostructures and their application in oxidative decomposition of formaldehyde. J Phys Chem C 111:18033–18038. https://doi.org/10.1021/jp076113n
Fei Z-Y, Sun B, Zhao L, Ji W-J, Au C-T (2013) Strong morphological effect of Mn3O4 nanocrystallites on the catalytic activity of Mn3O4 and Au/Mn3O4 in benzene combustion. Chem Eur J 19:6480–6487. https://doi.org/10.1002/chem.201204112
Wang F, Dai H, Deng J, Bai G, Ji K, Liu Y (2012) Manganese Oxides with Rod-, Wire-, Tube-, and Flower-Like Morphologies: Highly Effective Catalysts for the Removal of Toluene. Environ Sci Technol 46:4034–4041. https://doi.org/10.1021/es204038j
Piumetti M, Fino D, Russo N (2015) Mesoporous manganese oxides prepared by solution combustion synthesis as catalysts for the total oxidation of VOCs. Appl Catal B 163:277–287. https://doi.org/10.1016/j.apcatb.2014.08.012
Blanco J, Petre AL, Yates M, Martin MP, Martin JA, Martin-Luengo MA (2007) Tailor-made high porosity VOC oxidation catalysts prepared by a single-step procedure. Appl Catal B 73:128–134. https://doi.org/10.1016/j.apcatb.2006.06.017
Chen C, Jian H, Mai K, Ren Z, Wang J, Fu X, Fan C, Sun C, Qian G, Wang Z (2014) Shape- and size-controlled synthesis of Mn3O4 nanocrystals at room temperature. Eur J Inorg Chem 2014:3023–3029. https://doi.org/10.1002/ejic.201400013
Indra A, Menezes PW, Driess M (2015) Uncovering structure-activity relationships in manganese-oxide-based heterogeneous catalysts for efficient water oxidation. Chemsuschem 8:776–785. https://doi.org/10.1002/cssc.201402812
Song W, Poyraz AS, Meng Y, Ren Z, Chen S-Y, Suib SL (2014) Mesoporous Co3O4 with Controlled Porosity: Inverse Micelle Synthesis and High-Performance Catalytic CO Oxidation at −60 °C. Chem Mater 26:4629–4639. https://doi.org/10.1021/cm502106v
Genuino HC, Dharmarathna S, Njagi EC, Mei MC, Suib SL (2012) Gas-phase total oxidation of benzene, toluene, ethylbenzene, and xylenes using shape-selective manganese oxide and copper manganese oxide catalysts. J Phys Chem C 116:12066–12078. https://doi.org/10.1021/jp301342f
Haneda M, Taguchi R, Hattori M (2015) Influence of particle morphology on catalytic performance of CeO2/ZrO2 for soot oxidation. J Ceram Soc Jpn 123:414–418. https://doi.org/10.2109/jcersj2.123.414
Chen J, Chen X, Chen X, Xu W, Xu Z, Jia H, Chen J (2018) Homogeneous introduction of CeOy into MnOx-based catalyst for oxidation of aromatic VOCs. Appl Catal B 224:825–835. https://doi.org/10.1016/j.apcatb.2017.11.036
Tholkappiyan R, Nirmalesh Naveen A, Vishista K, Hamed F (2018) Investigation on the electrochemical performance of hausmannite Mn3O4 nanoparticles by ultrasonic irradiation assisted co-precipitation method for supercapacitor electrodes. J Taibah Univ Sci 12(5):669–677. https://doi.org/10.1080/16583655.2018.1497440
Dhanalaxmi K, Singuru R, Kundu SK, Reddy BM, Bhaumik A, Mondal J (2016) Strongly coupled Mn3O4–porous organic polymer hybrid: a robust, durable and potential nanocatalyst for alcohol oxidation reactions. RSC Adv 6(43):36728–36735. https://doi.org/10.1039/C6RA07200C
Raj AM, Victoria SG, Jothy VB, Ravidhas C, Wollschläger J, Suendorf M, Neumann M, Jayachandran M, Sanjeeviraja C (2010) XRD and XPS characterization of mixed valence Mn3O4 hausmannite thin films prepared by chemical spray pyrolysis technique. Appl Surf Sci 256(9):2920–2926. https://doi.org/10.1016/j.apsusc.2009.11.051
Acknowledgements
Financial support from DST, India under the Nano-Mission Programme (DST no: SR/NM/NS-1176/2013(G)) is gratefully acknowledged. Dr. Satyanarayana Chilikuri and his team from CSIR-NCL, India are being thanked for carrying out TPD experiments. A part of this work was performed at King Saud University, Saudi Arabia. The authors extend their appreciation to the International Scientific Partnership Program ISPP at King Saud University for funding this research work through ISPP# 0057. NEERI KRC No.: CSIR-NEERI/KRC/2019/NOV/ERMD/1.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Pulleri, J.K., Singh, S.K., Yearwar, D. et al. Morphology Dependent Catalytic Activity of Mn3O4 for Complete Oxidation of Toluene and Carbon Monoxide. Catal Lett 151, 172–183 (2021). https://doi.org/10.1007/s10562-020-03278-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10562-020-03278-w