Abstract
The oxygen reduction reaction (ORR) and hydrogen evolution reaction (HER) are two of the core reactions that occur in fuel cells and water electrolysis devices. Heteroatom-doped carbon materials are promising metal-free electrocatalysts to improve the conversion efficiency of these devices. To optimize the nanostructures of such carbon-based catalysts, herein, we reported an effective template method to synthesize N doped carbon nanotubes by using polydopamine as a precursor. The use of the ZnO nanowire not only serves as a self-sacrificial template to direct the formation of the nanotube, but also greatly extends the porosity of the carbon nanotube. Moreover, the polydopamine precursor also leads to effective N doping. An optimized sample, NCNT-900, shows high ORR performance comparable with that of Pt/C as well as good HER performance in both alkaline and acid media, making it one of the most effective carbon-based HER catalysts. This strategy offers an opportunity to synthesize catalysts with higher activity by rational design of a carbon precursor with higher N content or multi-heteroatom co-doping.
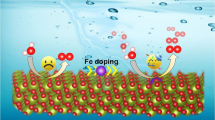
Graphical Abstract

Nitrogen doped carbon nanotube with high performance for both ORR and HER was synthesized using ZnO nanowires as template. The obtained materials show effective N doping that provides abundant active sites, high surface area and unique textural parameters that can effectively enhance mass transfer. When used for electrocatalysts, NCNT-900 shows high ORR performance comparable with that of Pt/C and good HER performance in both alkaline and acid media.





Similar content being viewed by others
References
Cook TR, Dogutan DK, Reece SY, Surendranath Y, Teets TS, Nocera DG (2010) Chem Rev 110:6474
Turner JA (2004) Science 305:972
Lewis NS, Nocera DG (2006) Proc Natl Acad Sci USA 103:15729
Duan J, Chen S, Jaroniec M, Qiao S (2015) ACS Catal 5:5207
Wang J, Xu F, Jin H, Chen Y, Wang Y (2017) Adv Mater 29:1605838
Huang Z, Wang J, Peng Y, Jung C, Fisher A, Wang X (2017) Adv Energy Mater 7:1700544
Li J, Hou P, Liu C (2017) Small 7:1702002
Debe MK (2012) Nature 486:43
Cheng N, Banis MN, Liu J, Riese A, Mu S, Li R, Sham TK, Sun X (2015) Energy Environ Sci 8:1450
Wu G, Zelenay P (2013) Acc Chem Res 46:1878
Fan X, Zhou H, Guo X (2015) ACS Nano 9:5125
Jin Y, Wang H, Li J, Yue X, Han Y, Shen PK, Cui Y (2016) Adv Mater 28:3785
Li Y, Xu H, Huang H, Gao L, Zhao Y, Ma T (2017) Electrochim Acta 254:148
Li JC, Hou PX, Zhang L, Liu C, Cheng HM (2014) Nanoscale 6:12065
Liu X, Zhou W, Yang L, Li L, Zhang Z, Ke Y, Chen S (2015) J Mater Chem A 3:8840
Gong K, Du F, Xia Z, Durstock M, Dai L (2009) Science 323:760
Zhao Y, Yang L, Chen S, Wang X, Ma Y, Wu Q, Jiang Y, Qian W, Hu Z (2013) J Am Chem Soc 135:1201
Guo D, Shibuya R, Akiba C, Saji S, Kondo T, Nakamura J (2016) Science 351:361
Davodi F, Tavakkoli M, Lahtinen J, Kallio T (2017) J Catal 353:19
Wang D, Su D (2014) Energy Environ Sci 7:576
Paraknowitsch JP, Thomas A (2013) Energy Environ Sci 6:2839
Gong YJ, Fei HL, Zou XL, Zhou W, Yang SB, Ye GL, Liu Z, Peng ZW, Lou J, Vajtai R, Yakobson BI, Tour JM, Ajayan PM (2015) Chem Mater 27:1181
Qu K, Zheng Y, Zhang X, Davey K, Dai S, Qiao S (2017) ACS Nano 11:7293
Song Z, Liu W, Cheng N, Banis MN, Li X, Sun Q, Xiao B, Liu Y, Lushington A, Li R, Liu L, Sun X (2017) Mater Horiz 4:900
Chen Y, Wang C, Wu Z, Xiong Y, Xu Q, Yu S, Jiang H (2015) Adv Mater 27:5010
Ferrero GA, Preuss K, Fuertes AB, Sevilla M, Titirici MM (2016) J Mater Chem A 4:2581
Xing T, Zheng Y, Li L, Cowie BCC, Gunzelmann D, Qiao S, Huang S, Chen Y (2014) ACS Nano 8:6856
Liu R, Wu D, Feng X, Mullen K (2010) Angew Chem 122:2619
Ding W, Wei Z, Chen S, Qi X, Yang T, Hu J, Wang D, Wan L, Alvi S, Li L (2013) Angew Chem Int Ed 52:11755
Shuai XM, Shen WZ WZ (2011) J Phys Chem C 2011, 115:6415
Zhang Z, Yi Z, Wang J, Tian X, Xu P, Shi G, Wang S (2017) J Mater Chem A 5:17064
Chen B, Li F, Mei Q, Yang Y, Liu H, Yuan G, Han B (2017) Chem Commun 53:13019
Li Y, Xu H, Huang H, Gao L, Ma T (2018) J Electrochem Soc 165:F158
Cheon JY, Kim JH, Kim J, Goddeti JH, Park KC, Joo JY SH (2014) J Am Chem Soc 136:8875
Ding W, Li L, Xiong K, Wang Y, Li W, Nie Y, Chen S, Qi X, Wei Z (2015) J Am Chem Soc 137:5414
Fechler N, Zussblatt NP, Rothe R, Schlogl R, Willinger MG, Chmelka BF, Antonietti M (2016) Adv Mater 28:1287
Li S, Cheng C, Liang H, Feng X, Thomas A (2017) Adv Mater 29:1700707
Wei W, Liang H, Parvez K, Zhuang X, Feng X, Mullen K (2014) Angew Chem Int Ed 53:1570
Muthukrishnan A, Nabae Y, Chang C, Okajimaa T, Ohsaka T (2015) Catal Sci Technol 5:1764
Singh D, Jenjeti R, Sampath S, Eswaramoorthy M (2017) J Mater Chem A 5:6025
Skulason E, Tripkovic V, Bjorketun ME, Gudmundsdottir S, Karlberg G, Rossmeisl J, Bligaard T, Jonsson H, Norskov JK (2010) J Phys Chem C 114:18182
Conway BE, Tilak BV (2002) Electrochim Acta 47:3571
Zheng Y, Jiao Y, Zhu Y, Li H, Han L, Chen Y, Du Y, Jaroniec A, Qiao M S (2014) Nat Commun 5:3783
Lai J, Li S, Wu F, Saqi M, Luque R, Xu G (2016) Energy Environ Sci 9:1210
Yue X, Huang S, Cai J, Jin Y, Shen P (2017) J Mater Chem A 5:7784
Zheng Y, Jiao Y, Li L, Xing T, Chen Y, Jaroniec M, Qiao S (2014) ACS Nano 8:5290
Ito Y, Cong W, Fujita T, Tang Z, Chen M (2015) Angew Chem Int Ed 54:2131
Yan X, Jia Y, Odedairo T, Zhao X, Jin Z, Zhu Z, Yao X (2016) Chem Commun 52:8156
Zhang B, Wen Z, Ci S, Chen J, He Z (2014) RSC Adv 4:49161
Zhao X, Li S, Cheng H, Schmidt J, Thomas A (2018) ACS Appl Mater Interfaces 10:3912
Acknowledgements
This work is supported financially by the National Natural Science Foundation of China (Grant No. 51772039), the Fundamental Research Funds for the Central University (DUT18LK13) and the Research Center for Solar Light Energy Conversion, Kyushu Institute of Technology, Japan.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Li, Y., Huang, H., Chen, S. et al. Killing Two Birds with One Stone: A Highly Active Tubular Carbon Catalyst with Effective N Doping for Oxygen Reduction and Hydrogen Evolution Reactions. Catal Lett 149, 486–495 (2019). https://doi.org/10.1007/s10562-018-2636-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10562-018-2636-5