Abstract
The photocatalytic reduction of CO2 to organic molecules is one promising approache for both decreasing CO2 concentration in the atmosphere and storing energies. However, most of the photocatalytic reduction of CO2 cannot avoid the utilization of sacrificial agents, which are not atomic economy and restricts the practical application of the photocatalytic reduction of CO2. In this contribution, an atomic economy photocatalytic reduction of CO2 to CH3OH coupling with the oxidation of amine to imine by Cu/TiO2 was reported, which can avoid using the sacrificial agents in the reaction systems. CO2 was reduced to CH3OH by photo-induced electrons; meanwhile benzylamine was selected as the reductant to react with photo-generated holes and converted into imine with high selectivity. The results showed that the maximum conversion of benzylamine to imine is 88.7% with selectivity of 98%, and the highest yield of CH3OH is 961.4 µmol g−1 using ca.0.5 wt% Cu/TiO2 as the catalyst. And also, the reaction mechanism was also investigated by DFT calculation, which would give a detailed explanation for the reaction process.
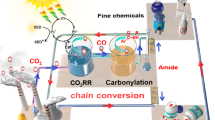
Graphical Abstract
Schematic illustration is that maximum conversion of benzylamine to imine is 88.7% with selectivity of 98%, and the highest yield of CH3OH is 961.4 μmol g−1 using ca.0.5 wt% Cu/TiO2 as the catalyst







Similar content being viewed by others
References
Fujishima A, Honda K (1972) Nature 238:37–38
Pal B, Sharon M (2002) Mater Chem Phys 76:82–87
Park H-A, Choi JH, Choi KM et al (2012) J Mater Chem 22:5304–5307
Bessekhouad Y, Mohammedi M, Trari M (2002) Sol Energy Mater Sol Cells 73:339–350
Moomaw WR, Unruh GC (1997) Environ Dev Econ 2:451–463
Takeda H, Cometto C, Ishitani O et al (2016) ACS Catal 7:70–88
Li Y, Lu G, Li S (2001) Appl Catal A 214:179–185
Nguyen T-V, Kim KJ, Yang OB (2005) J Photochem Photobiol A 173:56–63
Tamaki Y, Ishitani O (2017) ACS Catal 7:3394–3409
Wang L, Zhang X, Yang L et al (2015) Catal Sci Technol 5:4800–4805
Fujishima A, Nagahara LA, Yoshiki H et al (1994) Electrochim Acta 39:1229–1236
Lin L, Ou H, Zhang Y et al (2016) ACS Catal 6:3921–3931
Wang D, Liu Z, Yang W (2017) ACS Catal 7:2744–2752
Schneider TW, Ertem MZ, Muckerman JT et al (2016) ACS Catal 6:5473–5481
Habisreutinger SN, Schmidt-Mende L, Stolarczyk JK (2013) Angew Chem Int Ed 52:7372–7408
Zhou Y, Tian Z, Zhao Z et al (2011) ACS Appl Mater Interfaces 3:3594–3601
Xing M, Shen F, Qiu B et al (2014) Sci Rep 4:6341
Yang Y, Zhang T, Le L et al (2014) Sci Rep 4:6341
Pan J, Wu X, Wang L et al (2011) Chem Commun 47:8361–8363
Tan L, Chai S, Mohamed AR (2012) ChemSusChem 5:1868–1882
Liu L, Zhao C, Pitts D et al (2014) Catal Sci Technol 4:1539–1546
Li H, Zhang X, MacFarlane DR (2015) Adv Energy Mater 5:1401077
Liang Y, Vijayan BK, Gray KA et al (2011) Nano Lett 11:2865–2870
Inoue T, Fujishima A, Konishi S et al (1979) Nature 277:637–638
Fu Y, Sun D, Chen Y et al (2012) Angew Chem Int Ed 124:3420–3423
Simon T, Bouchonville N, Berr MJ et al (2014) Nat Mater 13:1013–1018
Huang D, Yin L, Niu J (2016) Environ Sci Technol 50:5857–5863
Sun Y, Cheng H, Gao S et al (2012) Angew Chem Int Ed 51:8727–8731
Guan M, Xiao C, Zhang J et al (2013) J Am Chem Soc 135:10411–10417
Spadavecchia F, Ceotto M, Presti L et al (2014) Chin J Chem 32:1195–1213
Yamada N, Suzumura M, Koiwa F et al (2013) Water Res 47:2770–2776
De Angelis F, Di Valentin C, Fantacci S et al (2014) Chem Rev 114:9708–9753
Zheng Z, Zhao J, Yuan Y et al (2013) Chem Eur J 19:5731–5741
Jiao W, Wang L, Liu G et al (2012) ACS Catal 2:1854–1859
Zeng G, Qiu J, Li Z et al (2014) ACS Catal 4:3512–3516
Ramesha GK, Brennecke JF, Kamat PV (2014) ACS Catal 4:3249–3254
Liu S, Liu J, Li X et al (2015) Plasma Processes Polym 12:422–430
Marschall R, Wang L (2014) Catal Today 225:111–135
Li Y, Xu D, Oh J II et al (2012) ACS Catal 2:391–398
Li M, Zhang S, Peng Y et al (2015) RSC Adv 5:7363–7369
Liu L, Chen X (2014) Chem Rev 114:9890–9918
Zuo F, Bozhilov K, Dillon RJ et al (2012) Angew Chem Int Ed 124:6327–6330
Xing M, Li X, Zhang J (2014) Sci Rep 4:5493
De Angelis F, Fantacci S, Selloni A et al (2007) Nano Lett 7:3189–3195
Zanella R, Rodríguez-González V, Arzola Y ACS Catal 2:1–11
Borlaf M, Colomer MT, de Andrés A et al (2014) Eur J Inorg Chem 30:5152–5159
Liyanage AD, Perera SD, Tan K et al (2014) ACS Catal 4:577–584
Song K, Han X, Shao G (2013) Alloys Compd 551:118–124
Yu X, Hou T, Sun X et al (2012) Chem Phys Chem 13:1514–1521
Priebe JB, Radnik J, Lennox AJJ et al (2015) ACS Catal 5:2137–2148
Chen P, Khetan A, Yang F et al (2017) ACS Catal 7:1197–1206
Du Q, Wu J, Yang H (2013) ACS Catal 4:144–151
Ola O, Maroto-Valer MM (2015) J Photochem Photobiol C 24:16–42
Blöchl PE (1994) Phys Rev B 50:17953
Kresse G, Joubert D (1999) Phys Rev B 59:1758–1775
Frisch MG, Trucks W, Schlegel HB et al (2009) Gaussian, Inc., Wallingford CT
Poulston S, Parlett PM, Stone P et al (1996) Surf Interface Anal 24:811–820
Ghodselahi T, Vesaghi MA, Shafiekhani A et al (2008) Appl Surf Sci 255:2730–2734
Acknowledgements
We are grateful to the support of the National Natural Science Foundation of China (21363014 and 21103082). This work was supported by the Natural Science Foundation of the Jiangxi Province of China (Grant Nos. 20142BCB23003 and 20143ACB21021).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yang, T., Yu, Q. & Wang, H. Photocatalytic Reduction of CO2 to CH3OH Coupling with the Oxidation of Amine to Imine. Catal Lett 148, 2382–2390 (2018). https://doi.org/10.1007/s10562-018-2412-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10562-018-2412-6