Abstract
Molecular sieves, complex organic bases and radical oxidants are commonly used in alcohols oxidation reactions. In this work, we have evaluated the beneficial effects of addition of K2CO3 to Pd(II)-catalyzed oxidation alcohols, which resulted in a remarkable increase in the oxidation reaction rates without selectivity losses. Herein, in a metallic reoxidant-free system, terpenic alcohols (β-citronellol, nerol and geraniol) were selectively converted into respective aldehydes from Pd(II)-catalyzed oxidation reactions in presence of dioxygen. High conversions and selectivities (greater than 90%) were achieved in the presence of the Pd(OAc)2/K2CO3 catalyst and pyridine excess. The exogenous role of others auxiliary anionic and nitrogen compounds was appraised.
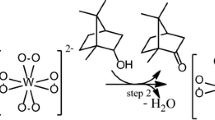
Graphical Abstract
Reaction conditions: β-citronellol (2.75 mmol); Pd(OAc)2 (0.05 mmol); pyridine (5.0 mmol); K2CO3 (2.5 mmol); toluene (10 mL); MS3A (0.5 g); O2 (0.10 MPa); 60 °C.

Comparison of systems used in Pd(OAc)2-catayzed oxidation of β-citronellol by dioxygen










Similar content being viewed by others
References
Muzart J (2003) Tetrahedron 59:5789
Gallezot P (2007) Catal Today 121:76
Pybus DH, Sell CS (2001) The chemistry of fragrances. RSC Paperbacks, Cambridge
Lenardão EJ, Botteselle GV, Azambuja F, Perin G, Jacob RG (2007) Tetrahedron 63:6671
Yoji H, Takeshi I, Yoshiki O (2002) European Patent EP122516, 2002
Brennfhrer A, Neumann H, Beller M (2009) ChemCatChem 1:28
da Silva MJ, Teixeira RR, Carari DM (2009) J Organomet Chem 694:3254
da Silva MJ, de Oliveira AA, da Silva ML (2009) Catal Lett 130:424
da Silva MJ, Gusevskaya EV (2001) J Mol Catal A 176:23
Bäckvall JE, Gogoll AJ (1987) J Chem Soc Chem Commun 1236
Peterson KP, Larock RC (1998) J Org Chem 63:3185
Bagdanoff JT, Ferreira EM, Stoltz BM (2003) Org Lett 5:835
Stoltz BM (2004) Chem Lett 33:362
Schultz MJ, Park CC, Sigman MS (2002) Chem Commun 3034
Nishimura T, Onoue T, Ohe K, Uemura S (1998) Tetrahedron Lett 39:6011
Nishimura T, Kakiuchi N, Onoue T, Ohe K, Uemura S (2000) J Chem Soc. Perkin Trans 1:1915
Nishimura T, Onoue T, Ohe K, Uemura S (1999) J Org Chem 64:6750
Nishimura T, Maeda Y, Kakiuchi N, Uemura S (2000) J Chem Soc. Perkin Trans 1:4301
Kumpulainen ETT, Koskinen AMP (2009) Chem Eur J 15:10901
Beccalli EM, Broggini G, Martinelli M, Sottocornola S (2007) Chem Rev 5318
Milstein D (2010) Top Catal 53:915
Mandal SK, Jensen DR, Pugsley JS, Sigman MS (2003) J Org Chem 68:4600
Steinhoff BA, Guzei IA, Stahl SS (2004) J Am Chem Soc 126:11268
Gligorich KM, Sigman MS (2009) Chem Commun 3854
Cornell CN, Sigman MS (2006) Org Lett 8:4117
Neumann D, Krauss G, Hieke M, Groger D (1983) Plant Med 48:20
Lide DR (ed) (1999) CRC handbook of chemistry and physics, 80th edn. CRC Press, Boca Raton
Muzart J (2009) J Mol Catal A 308:15
Ringsdoff H, Schlarb B, Venzmer J (1988) Angew Chem Int Ed 27:113
Grushin VV, Alper H (1993) Organometallics 12:1890
Schultz MJ, Hamilton SS, Jensen DR, Sigman MS (2005) J Org Chem 70:3343
Steinhoff BA, King AE, Stahl SS (2006) J Org Chem 71:1861
Nishimura T, Uemura S (2000) Catal Surv Jpn 4:31
Ferreira EM, Stoltz BM (2001) J Am Chem Soc 123:7725
Popp BV, Stahl SS (2007) J Am Chem Soc 129:4410
Popp BV, Stahl SS (2009) Chem Eur J 15:2915
Acknowledgements
The authors are grateful for the financial support from CAPES, CNPq, FAPEMIG and FUNARBE (Brazil). They also wish to thank Prof. Luis Claudio Barbosa for the GC–MS analyses.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Carari, D.M., da Silva, M.J. A Highly Selective Pd(OAc)2/Pyridine/K2CO3 System for Oxidation of Terpenic Alcohols by Dioxygen. Catal Lett 142, 251–258 (2012). https://doi.org/10.1007/s10562-011-0754-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10562-011-0754-4