Abstract
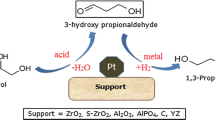
The evaluation of supported precious metal catalysts, in combination with and without solid acids, for the hydrogenolysis of highly concentrated glycerol solutions, with the aim to produce lower alcohols, in particular 1-propanol was studied. GCMS analysis of the product mixtures obtained has shed some light on the possible mechanism leading to unwanted condensation products, as well as the composition of these compounds.
Graphical Abstract





Similar content being viewed by others
References
Miyazawa T, Kusunoki Y, Kunimori K, Tomishige K (2006) J Catal 240:213
Dasari MA, Kiatsimkul PP, Sutterlin WR, Suppes GJ (2005) Appl Catal A 281:225
Tsukuda E, Sato S, Takahashi R, Sodesawa T (2007) Catal Comm 8:1349
Hirai T, Ikenaga N, Miyake T, Suzuki T (2005) Energy Fuels 19:1761
Furikado I, Miyazawa T, Koso S, Shimao A, Kunimori K, Tomishige K (2007) Green Chem 9:582
Tanikella MSSR (1983) E.I. Du Pont de Nemours and Company. US Patent 4,404,411
Miyazawa T, Koso S, Kunimori K, Tomishige K (2007) Appl Catal A 329:30
Miyazawa T, Kusunoki Y, Kunimori K, Tomishige K (2005) Catal Comm 6:645
Balaraju M, Rekha V, Sai Prasad PS, Prabhavathi Devi BLA, Prasad RBN, Lingaiah N (2009) Appl Catal A 354:82
Chaminand J, Djakovitch L, Gallezot P, Marion P, Pinel C, Rosier C (2004) Green Chem 6:359
Ma L, He D, Li Z (2008) Catal Comm 9:2489
Yuan Z, Wu P, Gao J, Lu X, Hou Z, Zheng X (2009) Catal Lett 130:261
Feng J, Fu H, Wang J, Li R, Chen H, Li X (2008) Catal Comm 9:1458
Shinmi Y, Koso S, Kubato T, Nakagawa Y, Tomishige K (2010) Appl Catal B 94:318
Pavlechko PD (2009) Higher alcohols from syngas. SRI Consulting. PEP Report no. 268
Amada Y, Koso S, Nakagawa Y, Tomishige K (2010) ChemSusChem 3:728
Lahr DG, Shanks BH (2005) J Catal 232:386
Montassier C, Menezo JC, Hoang LC, Renaud C, Barbier J (1991) J Mol Catal 70:99
Akiyama M, Sato S, Takahashi R, Inui K, Yokota M (2009) Appl Catal A 371:60
Busca G (2007) Chem Rev 107:5366
Santacesaria E, Grasso D (1990) Appl Catal 64:83
Gandarias I, Arias PL, Requies J, Guemez MB, Fierro JLG (2010) Appl Catal B 97:248
Deutsch J, Martin A, Lieske H (2007) J Catal 245:428
Ruiz VR, Velty A, Santos LL, Leyva-Perez A, Sabater MJ, Iborra S, Corma A (2010) J Catal 271:351
Patel SM, Chudasama UV, Ganeshpure PA (2003) J Mol Catal A 194:267
Acknowledgments
Thanks are due to Johnson & Matthey UK for the preparation of the precious metal catalysts and Sasol Technology for the support to perform this study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
van Ryneveld, E., Mahomed, A.S., van Heerden, P.S. et al. Direct Hydrogenolysis of Highly Concentrated Glycerol Solutions Over Supported Ru, Pd and Pt Catalyst Systems. Catal Lett 141, 958–967 (2011). https://doi.org/10.1007/s10562-011-0617-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10562-011-0617-z