Abstract
Background
Coronary effluent from an isolated perfused heart undergoing ischemic preconditioning can be transferred to precondition another naïve isolated heart. We investigated the effects of this effluent on mitochondrial integrity and function following a global infarct model of ischemia/reperfusion and the role of adenosine in this model of remote preconditioning.
Methods and Results
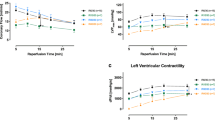
Coronary effluent from isolated perfused rabbit hearts was collected prior to (control effluent) and during three cycles of 5-min ischemia and 10-min reperfusion (IPC effluent). Adenosine concentration was significantly increased in IPC effluent (2.6 ± 1.1 μM) versus control effluent (0.21 ± 0.06 μM, P < 0.01). Infarct size (% necrotic LV mass) after 30-min global ischemia and 90-min reperfusion was significantly reduced in hearts preconditioned with IPC effluent (IPCeff, 23 ± 7 %) and control effluent supplemented with 2.5 μM exogenous adenosine (Ceff + 2.5 μM ADO, 25 ± 10 %) when compared to control effluent perfused hearts (Ceff, 41 ± 8 %, P < 0.05). Compared to Ceff mitochondria, IPCeff mitochondria had preserved complex I/State3 and complex IV/State 3 respiration and outer membrane integrity, and reduced cytochrome c release. In contrast, Ceff + 2.5 μM ADO mitochondria had improved state 2 respiration and coupling to oxidative phosphorylation, reduced reactive oxygen species production and preserved outer membrane integrity. Administration of adenosine receptor blocker 8-(p-sulfophenyl)theophylline abolished the infarct limiting effect (46 ± 7 %) and the mitochondrial integrity and function preservation of IPC effluent.
Conclusion
Remote cardioprotection by IPC effluent preserves mitochondrial integrity and function in an adenosine receptor dependent mechanism, and although infarct size reduction can be mimicked by adenosine, IPC effluent contains additional factor(s) contributing to modulation of the mitochondrial response to ischemia/reperfusion injury.





Similar content being viewed by others
References
Murry CE, Jennings RB, Reimer KA. Preconditioning with ischemia: a delay of lethal cell injury in ischemic myocardium. Circulation. 1986;74(5):1124–36.
Oxman T, Arad M, Klein R, Avazov N, Rabinowitz B. Limb ischemia preconditions the heart against reperfusion tachyarrhythmia. Am J Physiol. 1997;273(4 Pt 2):H1707–12.
Pell TJ, Baxter GF, Yellon DM, Drew GM. Renal ischemia preconditions myocardium: role of adenosine receptors and ATP-sensitive potassium channels. Am J Physiol. 1998;275(5 Pt 2):H1542–7.
Konstantinov IE, Li J, Cheung MM, Shimizu M, Stokoe J, Kharbanda RK, et al. Remote ischemic preconditioning of the recipient reduces myocardial ischemia-reperfusion injury of the denervated donor heart via a Katp channel-dependent mechanism. Transplantation. 2005;79(12):1691–5.
Yang X, Cohen MV, Downey JM. Mechanism of cardioprotection by early ischemic preconditioning. Cardiovasc Drugs Ther. 2010;24(3):225–34.
Shimizu M, Tropak M, Diaz RJ, Suto F, Surendra H, Kuzmin E, et al. Transient limb ischaemia remotely preconditions through a humoral mechanism acting directly on the myocardium: evidence suggesting cross-species protection. Clin Sci (Lond). 2009;117(5):191–200.
Wang L, Oka N, Tropak M, Callahan J, Lee J, Wilson G, et al. Remote ischemic preconditioning elaborates a transferable blood-borne effector that protects mitochondrial structure and function and preserves myocardial performance after neonatal cardioplegic arrest. J Thorac Cardiovasc Surg. 2008;136(2):335–42.
Dickson EW, Lorbar M, Porcaro WA, Fenton RA, Reinhardt CP, Gysembergh A, et al. Rabbit heart can be “preconditioned” via transfer of coronary effluent. Am J Physiol. 1999;277(6 Pt 2):H2451–7.
Serejo FC, Rodrigues Jr LF, da Silva Tavares KC, de Carvalho AC, Nascimento JH. Cardioprotective properties of humoral factors released from rat hearts subject to ischemic preconditioning. J Cardiovasc Pharmacol. 2007;49(4):214–20.
Breivik L, Helgeland E, Aarnes EK, Mrdalj J, Jonassen AK. Remote postconditioning by humoral factors in effluent from ischemic preconditioned rat hearts is mediated via PI3K/Akt-dependent cell-survival signaling at reperfusion. Basic Res Cardiol. 2010;106(1):135–45.
Chen Q, Moghaddas S, Hoppel CL, Lesnefsky EJ. Ischemic defects in the electron transport chain increase the production of reactive oxygen species from isolated rat heart mitochondria. Am J Physiol Cell Physiol. 2008;294(2):C460–6.
Chen Q, Vazquez EJ, Moghaddas S, Hoppel CL, Lesnefsky EJ. Production of reactive oxygen species by mitochondria: central role of complex III. J Biol Chem. 2003;278(38):36027–31.
Borutaite V, Jekabsone A, Morkuniene R, Brown GC. Inhibition of mitochondrial permeability transition prevents mitochondrial dysfunction, cytochrome c release and apoptosis induced by heart ischemia. J Mol Cell Cardiol. 2003;35(4):357–66.
Lesnefsky EJ, Tandler B, Ye J, Slabe TJ, Turkaly J, Hoppel CL. Myocardial ischemia decreases oxidative phosphorylation through cytochrome oxidase in subsarcolemmal mitochondria. Am J Physiol. 1997;273(3 Pt 2):H1544–54.
Jiang X, Wang X. Cytochrome C-mediated apoptosis. Annu Rev Biochem. 2004;73:87–106.
Crestanello JA, Lingle DM, Kamelgard J, Millili J, Whitman GJ. Ischemic preconditioning decreases oxidative stress during reperfusion: a chemiluminescence study. J Surg Res. 1996;65(1):53–8.
Javadov SA, Clarke S, Das M, Griffiths EJ, Lim KH, Halestrap AP. Ischaemic preconditioning inhibits opening of mitochondrial permeability transition pores in the reperfused rat heart. J Physiol. 2003;549(Pt 2):513–24.
Lundberg KC, Szweda LI. Preconditioning prevents loss in mitochondrial function and release of cytochrome c during prolonged cardiac ischemia/reperfusion. Arch Biochem Biophys. 2006;453(1):130–4.
Liu GS, Thornton J, Van Winkle DM, Stanley AW, Olsson RA, Downey JM. Protection against infarction afforded by preconditioning is mediated by A1 adenosine receptors in rabbit heart. Circulation. 1991;84(1):350–6.
Takaoka A, Nakae I, Mitsunami K, Yabe T, Morikawa S, Inubushi T, et al. Renal ischemia/reperfusion remotely improves myocardial energy metabolism during myocardial ischemia via adenosine receptors in rabbits: effects of “remote preconditioning”. J Am Coll Cardiol. 1999;33(2):556–64.
Goto M, Cohen MV, van Wylen DG, Downey JM. Attenuated purine production during subsequent ischemia in preconditioned rabbit myocardium is unrelated to the mechanism of protection. J Mol Cell Cardiol. 1996;28(3):447–54.
Chen Q, Camara AK, Stowe DF, Hoppel CL, Lesnefsky EJ. Modulation of electron transport protects cardiac mitochondria and decreases myocardial injury during ischemia and reperfusion. Am J Physiol Cell Physiol. 2007;292(1):C137–47.
Palmer JW, Tandler B, Hoppel CL. Biochemical properties of subsarcolemmal and interfibrillar mitochondria isolated from rat cardiac muscle. J Biol Chem. 1977;252(23):8731–9.
Ricci JE, Gottlieb RA, Green DR. Caspase-mediated loss of mitochondrial function and generation of reactive oxygen species during apoptosis. J Cell Biol. 2003;160(1):65–75.
Lee AC, Zizi M, Colombini M. Beta-NADH decreases the permeability of the mitochondrial outer membrane to ADP by a factor of 6. J Biol Chem. 1994;269(49):30974–80.
Mootha VK, Wei MC, Buttle KF, Scorrano L, Panoutsakopoulou V, Mannella CA, et al. A reversible component of mitochondrial respiratory dysfunction in apoptosis can be rescued by exogenous cytochrome c. EMBO J. 2001;20(4):661–71.
William JN. A method for the simultaneous quantitative estimation of cytochrome a, b, c1 and and c in mitochondrial. Arch Biochem Biophys. 1964;107:537–43.
Leung CH, Wang L, Fu YY, Yuen W, Caldarone CA. Transient mitochondrial permeability transition pore opening after neonatal cardioplegic arrest. J Thorac Cardiovasc Surg. 2011;141(4):975–82.
Dickson EW, Blehar DJ, Carraway RE, Heard SO, Steinberg G, Przyklenk K. Naloxone blocks transferred preconditioning in isolated rabbit hearts. J Mol Cell Cardiol. 2001;33(9):1751–6.
Schrader J, Haddy FJ, Gerlach E. Release of adenosine, inosine and hypoxanthine from the isolated guinea pig heart during hypoxia, flow-autoregulation and reactive hyperemia. Pflugers Arch. 1977;369(1):1–6.
Peart JN, Gross GJ. Adenosine and opioid receptor-mediated cardioprotection in the rat: evidence for cross-talk between receptors. Am J Physiol Heart Circ Physiol. 2003;285(1):H81–9.
Surendra H, Diaz RJ, Harvey K, Tropak M, Callahan J, Hinek A, et al. Interaction of delta and kappa opioid receptors with adenosine A1 receptors mediates cardioprotection by remote ischemic preconditioning. J Mol Cell Cardiol. 2013;60:142–50.
Halestrap AP, Clarke SJ, Javadov SA. Mitochondrial permeability transition pore opening during myocardial reperfusion–a target for cardioprotection. Cardiovasc Res. 2004;61(3):372–85.
Brand MD, Pakay JL, Ocloo A, Kokoszka J, Wallace DC, Brookes PS, et al. The basal proton conductance of mitochondria depends on adenine nucleotide translocase content. Biochem J. 2005;392(Pt 2):353–62.
Yang Z, Sun W, Hu K. Molecular mechanism underlying adenosine receptor-mediated mitochondrial targeting of protein kinase C. Biochim Biophys Acta. 2012;1823(4):950–8.
Baines CP, Song CX, Zheng YT, Wang GW, Zhang J, Wang OL, et al. Protein kinase Cepsilon interacts with and inhibits the permeability transition pore in cardiac mitochondria. Circ Res. 2003;92(8):873–80.
Scorrano L, Petronilli V, Bernardi P. On the voltage dependence of the mitochondrial permeability transition pore. A critical appraisal. J Biol Chem. 1997;272(19):12295–9.
Dana A, Jonassen AK, Yamashita N, Yellon DM. Adenosine A(1) receptor activation induces delayed preconditioning in rats mediated by manganese superoxide dismutase. Circulation. 2000;101(24):2841–8.
Clarke SJ, Khaliulin I, Das M, Parker JE, Heesom KJ, Halestrap AP. Inhibition of mitochondrial permeability transition pore opening by ischemic preconditioning is probably mediated by reduction of oxidative stress rather than mitochondrial protein phosphorylation. Circ Res. 2008;102(9):1082–90.
Kerr PM, Suleiman MS, Halestrap AP. Reversal of permeability transition during recovery of hearts from ischemia and its enhancement by pyruvate. Am J Physiol. 1999;276(2 Pt 2):H496–502.
Kristiansen SB, Henning O, Kharbanda RK, Nielsen-Kudsk JE, Schmidt MR, Redington AN, et al. Remote preconditioning reduces ischemic injury in the explanted heart by a KATP channel-dependent mechanism. Am J Physiol Heart Circ Physiol. 2005;288(3):H1252–6.
Wang L, Cherednichenko G, Hernandez L, Halow J, Camacho SA, Figueredo V, et al. Preconditioning limits mitochondrial Ca(2+) during ischemia in rat hearts: role of K(ATP) channels. Am J Physiol Heart Circ Physiol. 2001;280(5):H2321–8.
Murata M, Akao M, O’Rourke B, Marban E. Mitochondrial ATP-sensitive potassium channels attenuate matrix Ca(2+) overload during simulated ischemia and reperfusion: possible mechanism of cardioprotection. Circ Res. 2001;89(10):891–8.
Fryer RM, Eells JT, Hsu AK, Henry MM, Gross GJ. Ischemic preconditioning in rats: role of mitochondrial K(ATP) channel in preservation of mitochondrial function. Am J Physiol Heart Circ Physiol. 2000;278(1):H305–12.
Koomen JM, Wilson CR, Guthrie P, Androlewicz MJ, Kobayashi R, Taegtmeyer H. Proteome analysis of isolated perfused organ effluent as a novel model for protein biomarker discovery. J Proteome Res. 2006;5(1):177–82.
Naydenova Z, Rose JB, Coe IR. Inosine and equilibrative nucleoside transporter 2 contribute to hypoxic preconditioning in the murine cardiomyocyte HL-1 cell line. Am J Physiol Heart Circ Physiol. 2008;294(6):H2687–92.
Jin X, Shepherd RK, Duling BR, Linden J. Inosine binds to A3 adenosine receptors and stimulates mast cell degranulation. J Clin Invest. 1997;100(11):2849–57.
Peart J, Matherne GP, Cerniway RJ, Headrick JP. Cardioprotection with adenosine metabolism inhibitors in ischemic-reperfused mouse heart. Cardiovasc Res. 2001;52(1):120–9.
Steensrud T, Li J, Dai X, Manlhiot C, Kharbanda RK, Tropak M, et al. Pretreatment with the nitric oxide donor SNAP or nerve transection blocks humoral preconditioning by remote limb ischemia or intra-arterial adenosine. Am J Physiol Heart Circ Physiol. 2010;299(5):H1598–603.
Grants
This study was supported by The Leducq Foundation (grant number 06/CVD) and the Heart and Stroke Foundation of Ontario.
Conflicts of Interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Supplementary Figure 1
Ions detected in the effluent by reverse phase liquid chromatography mass spectrometry. Intensity of select ions in IPC effluent (n = 2) relative to Control effluent (n = 2) is shown on the y-axis. Mass to charge (m/z) ratio for detected ions are shown on the x-axis. This ratio is a reflection of the molecular weight of the individual ions detected by the mass spectrometer (LTQ Orbitrap). (JPEG 12 kb)
Rights and permissions
About this article
Cite this article
Leung, C.H., Wang, L., Nielsen, J.M. et al. Remote Cardioprotection by Transfer of Coronary Effluent from Ischemic Preconditioned Rabbit Heart Preserves Mitochondrial Integrity and Function via Adenosine Receptor Activation. Cardiovasc Drugs Ther 28, 7–17 (2014). https://doi.org/10.1007/s10557-013-6489-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10557-013-6489-2