Abstract
Introduction
One year is the standard duration of adjuvant trastuzumab for human epidermal receptor-2 (HER2) positive (HER2+) breast cancer (BC). Indeed, a shorter duration of trastuzumab can reduce cardiotoxicity and the costs involved and could provide the same benefit as a one-year treatment. We evaluated the available evidence from randomised controlled trials (RCTs) by comparing 1 year versus a shorter duration of adjuvant trastuzumab for HER2+ BC.
Patients and methods
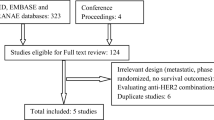
A systematic search of PubMed, EMBASE, The Cochrane Library and conference proceedings was carried out in order to identify the RCTs that investigated a standard versus a shorter duration of adjuvant trastuzumab in HER2+ BC patients. Using the fixed and random effects models, the pooled hazard ratios (HRs) and risk ratio (RR) with 95% confidence intervals (CI) were calculated for overall survival (OS), disease-free survival (DFS) and cardiac events.
Results
Five RCTs with a total of 11,381 patients were included. Overall, one year of adjuvant trastuzumab improved OS (HR 1.22, 95% CI 1.07–1.39; P = 0.003) and DFS (HR 1.19, 95% CI 1.08–1.3; P < 0.001) compared with a shorter duration (6 months and 9 weeks). In the subgroup analysis, there was a trend towards better DFS with the 1-year duration for patients with high-risk features, and also for concomitant administration of chemotherapy and trastuzumab. Cardiac events were significantly lower with a shorter duration (RR 0.4, 95% CI 0.32–0.49; P < 0.001).
Conclusions
One-year adjuvant trastuzumab is associated with better DFS and OS compared with shorter durations and should still be considered the standard duration. However, selected patients with low-risk HER2+ BC can most likely be spared from an excess of cardiac toxicity with a shorter course.





Similar content being viewed by others
References
Slamon DJ, Clark GM, Wong SG, Levin WJ, Ullrich A, McGuire WL (1987) Human breast cancer: correlation of relapse and survival with amplification of the HER-2lneu Oncogene. Science. https://doi.org/10.1126/science.3798106
Piccart-Gebhart MJ, Procter M, Leyland-Jones B et al (2005) Trastuzumab after adjuvant chemotherapy in HER2-positive breast cancer. N Engl J Med 353(16):1659–1672. https://doi.org/10.1056/NEJMoa052306
Romond EH, Perez EA, Bryant J et al (2005) Trastuzumab plus adjuvant chemotherapy for operable HER2-positive breast cancer. N Engl J Med 353(16):1673–1684. https://doi.org/10.1056/NEJMoa052122
Slamon D, Eiermann W, Robert N et al (2011) Adjuvant trastuzumab in HER2-positive breast cancer. N Engl J Med 365(14):1273–1283. https://doi.org/10.1056/NEJMoa0910383
Perez EA, Romond EH (2014) Suman V. J. et al. Trastuzumab plus adjuvant chemotherapy for human epidermal growth factor receptor 2–positive breast cancer: planned joint analysis of overall survival from NSABP B-31 and NCCTG N9831. J Clin Oncol 32(33):3744–3752. https://doi.org/10.1200/JCO.2014.55.5730
Smith I, Procter M, Gelber RD et al (2007) 2-year follow-up of trastuzumab after adjuvant chemotherapy in HER2-positive breast cancer: a randomised controlled trial. Lancet. https://doi.org/10.1016/S0140-6736(07)60028-2
Gianni L, Dafni U, Gelber RD et al (2011) Treatment with trastuzumab for 1 year after adjuvant chemotherapy in patients with HER2-positive early breast cancer: a 4-year follow-up of a randomised controlled trial. Lancet Oncol. https://doi.org/10.1016/S1470-2045(11)70033-X
Cameron D, Piccart-Gebhart MJ, Gelber RD et al (2017) 11 years’ follow-up of trastuzumab after adjuvant chemotherapy in HER2-positive early breast cancer: final analysis of the Herceptin adjuvant (HERA) trial. Lancet. https://doi.org/10.1016/S0140-6736(16)32616-2
Goldhirsch A, Gelber RD, Piccart-Gebhart MJ et al (2013) 2 years versus 1 year of adjuvant trastuzumab for HER2-positive breast cancer (HERA): an open-label, randomised controlled trial. Lancet 382(9897):1021–1028. https://doi.org/10.1016/S0140-6736(13)61094-6
Joensuu H, Kellokumpu-Lehtinen PL, Bono P et al (2006) Adjuvant cocetaxel or vinorelbine with or without trastuzumab for breast cancer. N Engl J Med 354:809–820. https://doi.org/10.1056/NEJMoa053028
Joensuu H, Bono P, Kataja V et al (2009) Fluorouracil, epirubicin, and cyclophosphamide with either docetaxel or vinorelbine, with or without trastuzumab, as adjuvant treatments of breast cancer: final results of the FinHer trial. J Clin Oncol 27(34):5685–5692. https://doi.org/10.1200/JCO.2008.21.4577
Moher D, Liberati A, Tetzlaff J, Altman DG (2009) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. BMJ 339(1):b2535–b2535. https://doi.org/10.1136/bmj.b2535
Jadad AR, Moore RA, Carroll D et al (1996) Assessing the quality of reports of randomized clinical trials: is blinding necessary? Control Clin Trials 17(1):1–12. https://doi.org/10.1016/0197-2456(95)00134-4
Wells GA, Shea B, O’Connell D et al (2014) The Newcastle-Ottawa Scale (NOS) for assessing the quality of non-randomised studies in meta-analyses. Ottawa Hosp Res Inst. http://www.ohri.ca/programs/clinical_epidemiology/oxford.asp. Accessed 13 Aug 2014
Higgins JPT, Thompson SG, Deeks JJ, Altman DG (2003) Measuring inconsistency in meta-analyses. BMJ Br Med J 327(7414):557–560. https://doi.org/10.1136/bmj.327.7414.557
C P (2017) P. C, G. B et al. Final analysis of the phase III multicentric Italian study Short-HER: 9 weeks vs 1-year adjuvant trastuzumab for HER21 early breast cancer. Ann Oncol. https://doi.org/10.1093/annonc/mdx421
Joensuu H, Fraser J, Wildiers H et al (2018) Effect of adjuvant trastuzumab for a duration of 9 weeks vs 1 year with concomitant chemotherapy for early human epidermal growth factor receptor 2-positive breast cancer: the SOLD randomized clinical trial. JAMA Oncol. https://doi.org/10.1001/jamaoncol.2018.1380
Mavroudis D, Saloustros E, Malamos N, Kakolyris S, Boukovinas I (2015) Six versus twelve months of adjuvant trastuzumab in combination with dose-dense chemotherapy for women with HER2-positive breast cancer: a multicenter randomized study by the Hellenic Oncology Research Group (HORG). Ann Oncol 26(7):1333–1340
Pivot X, Romieu G, Debled M et al (2013) 6 months versus 12 months of adjuvant trastuzumab for patients with HER2-positive early breast cancer (PHARE): a randomised phase 3 trial. Lancet Oncol 14(8):741–748. https://doi.org/10.1016/S1470-2045(13)70225-0
Dahabreh IJ, Linardou H, Siannis F, Fountzilas G, Murray S (2008) Trastuzumab in the adjuvant treatment of early-stage breast cancer: a systematic review and meta-analysis of randomized controlled trials. Oncologist 13(6):620–630. https://doi.org/10.1634/theoncologist.2008-0001
Moher D, Liberati A, Tetzlaff J, Altman DG, The PRISMA Group (2009) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med 6(6): e1000097. https://doi.org/10.1371/journal.pmed1000097. http://www.prisma-statement.org
Denduluri N, Chavez-MacGregor M, Telli ML et al. Selection of optimal adjuvant chemotherapy and targeted therapy for early breast cancer: ASCO Clinical Practice Guideline Focused Update. J Clin Oncol. 2018:JCO2018788604. https://doi.org/10.1200/JCO.2018.78.8604
Gyawali B, Niraula S (2017) Duration of adjuvant trastuzumab in HER2 positive breast cancer: overall and disease-free survival results from meta-analyses of randomized controlled trials. Cancer Treat Rev 60:18–23. https://doi.org/10.1016/j.ctrv.2017.08.001
Murray GD (2001) Switching between superiority and non-inferiority. Br J Clin Pharmacol 52(3):219. https://doi.org/10.1046/j.0306-5251.2001.01397.x
von Minckwitz G, Procter M, de Azambuja E et al (2017) Adjuvant pertuzumab and trastuzumab in early HER2-positive breast cancer. N Engl J Med 377(2):122–131. https://doi.org/10.1056/NEJMoa1703643
Martin M, Holmes FA, Ejlertsen B et al (2017) Neratinib after trastuzumab-based adjuvant therapy in HER2-positive breast cancer (ExteNET): 5-year analysis of a randomised, double-blind, placebo-controlled, phase-3 trial. Lancet Oncol 18(12):1688–1700. https://doi.org/10.1016/S1470-2045(17)30717-9
Funding
None received.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Research involving human and animal rights
This article does not contain any studies with human participants or animals performed by any of the authors.
Rights and permissions
About this article
Cite this article
Inno, A., Barni, S., Ghidini, A. et al. One year versus a shorter duration of adjuvant trastuzumab for HER2-positive early breast cancer: a systematic review and meta-analysis. Breast Cancer Res Treat 173, 247–254 (2019). https://doi.org/10.1007/s10549-018-5001-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-018-5001-x