Abstract
Purpose
Although axillary lymph node status has traditionally been a key factor in informing adjuvant breast cancer therapy recommendations, this information may be less relevant as our focus shifts more towards tumor biology, particularly in older patients where comorbidity influences treatment decisions and nodal staging and/or surgery may not improve outcomes. We examined patterns of axillary surgery and associations between axillary surgery and receipt of adjuvant treatment in older breast cancer patients.
Methods
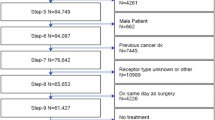
Women aged ≥ 65 years with clinically node-negative, stage I–II breast cancer treated between 2012 and 2013 were identified using the National Cancer Data Base. Using multivariable logistic regression, we examined associations between axillary surgery and age, adjusting for patient, clinical, and facility factors. We also examined receipt of adjuvant treatment by nodal surgery.
Results
Among 68,205 women, 40.1% were aged 65–70, 24.5% were 71–75, 17.4% were 76–80, and 18.0% were > 80. Overall, 91.2% had axillary surgery (67.8% sentinel lymph node biopsy, 11.7% axillary lymph node dissection, 11.7% unspecified/unknown axillary surgery); 88.0% of those aged ≥ 70 with lower risk, hormone receptor-positive tumors underwent axillary surgery. In adjusted analyses, compared to patients aged 65–70, increasing age was associated with lower odds of any axillary surgery (ages 71–75: OR 0.64, 95% CI 0.57–0.71; ages 76–80: OR 0.33, 95% CI 0.30–0.37; age > 80: OR 0.08, 95% CI 0.07–0.08). Axillary surgery was associated with higher odds of receipt of radiation after breast conservation and receipt of chemotherapy in human epidermal growth factor 2-positive disease.
Conclusions
In a large nationwide dataset, the vast majority of older women with clinically node-negative breast cancer underwent axillary staging despite uncertainty about its impact on survival, particularly for those with lower-risk disease. Further study on how to tailor node assessment in older patients is warranted.

Similar content being viewed by others
References
Giuliano AE, Hunt KK, Ballman KV, Beitsch PD, Whitworth PW, Blumencranz PW, Leitch AM, Saha S, McCall LM, Morrow M (2011) Axillary dissection vs no axillary dissection in women with invasive breast cancer and sentinel node metastasis: a randomized clinical trial. JAMA 305(6):569–575. doi:10.1001/jama.2011.90
Giuliano AE, Ballman K, McCall L, Beitsch P, Whitworth PW, Blumencranz P, Leitch AM, Saha S, Morrow M, Hunt KK (2016) Locoregional recurrence after sentinel lymph node dissection with or without axillary dissection in patients with sentinel lymph node metastases: long-term follow-up from the American College of Surgeons Oncology Group (Alliance) ACOSOG Z0011 randomized trial. Ann Surg 264(3):413–420. doi:10.1097/SLA.0000000000001863
Galimberti V, Cole BF, Zurrida S, Viale G, Luini A, Veronesi P, Baratella P, Chifu C, Sargenti M, Intra M, Gentilini O, Mastropasqua MG, Mazzarol G, Massarut S, Garbay JR, Zgajnar J, Galatius H, Recalcati A, Littlejohn D, Bamert M, Colleoni M, Price KN, Regan MM, Goldhirsch A, Coates AS, Gelber RD, Veronesi U, International Breast Cancer Study Group Trial 23-01 investigators (2013) Axillary dissection versus no axillary dissection in patients with sentinel-node micrometastases (IBCSG 23-01): a phase 3 randomised controlled trial. Lancet Oncol 14(4):297–305. doi:10.1016/S1470-2045(13)70035-4
Martelli G, Miceli R, Costa A, Coradini D, Zurrida S, Piromalli D, Vetrella G, Greco M (2008) Elderly breast cancer patients treated by conservative surgery alone plus adjuvant tamoxifen: fifteen-year results of a prospective study. Cancer 112(3):481–488. doi:10.1002/cncr.23213
Martelli G, Boracchi P, Ardoino I, Lozza L, Bohm S, Vetrella G, Agresti R (2012) Axillary dissection versus no axillary dissection in older patients with T1N0 breast cancer: 15-year results of a randomized controlled trial. Ann Surg 256(6):920–924. doi:10.1097/SLA.0b013e31827660a8
Martelli G, Boracchi P, De Palo M, Pilotti S, Oriana S, Zucali R, Daidone MG, De Palo G (2005) A randomized trial comparing axillary dissection to no axillary dissection in older patients with T1N0 breast cancer: results after 5 years of follow-up. Ann Surg 242(1):1–6 (discussion 7–9)
Elkin EB, Hurria A, Mitra N, Schrag D, Panageas KS (2006) Adjuvant chemotherapy and survival in older women with hormone receptor-negative breast cancer: assessing outcome in a population-based, observational cohort. J Clin Oncol 24(18):2757–2764. doi:10.1200/JCO.2005.03.6053
Howlader N, Altekruse SF, Li CI, Chen VW, Clarke CA, Ries LA, Cronin KA (2014) US incidence of breast cancer subtypes defined by joint hormone receptor and HER2 status. J Natl Cancer Inst. doi:10.1093/jnci/dju055
Schonberg MA, Marcantonio ER, Ngo L, Li D, Silliman RA, McCarthy EP (2011) Causes of death and relative survival of older women after a breast cancer diagnosis. J Clin Oncol 29(12):1570–1577. doi:10.1200/JCO.2010.33.0472
Satariano WA, Ragland DR (1994) The effect of comorbidity on 3-year survival of women with primary breast cancer. Ann Intern Med 120(2):104–110
Hughes KS, Schnaper LA, Berry D, Cirrincione C, McCormick B, Shank B, Wheeler J, Champion LA, Smith TJ, Smith BL, Shapiro C, Muss HB, Winer E, Hudis C, Wood W, Sugarbaker D, Henderson IC, Norton L, Cancer and Leukemia Group B, Radiation Therapy Oncology Group, Eastern Cooperative Oncology Group (2004) Lumpectomy plus tamoxifen with or without irradiation in women 70 years of age or older with early breast cancer. N Engl J Med 351(10):971–977. doi:10.1056/NEJMoa040587
Hughes KS, Schnaper LA, Bellon JR, Cirrincione CT, Berry DA, McCormick B, Muss HB, Smith BL, Hudis CA, Winer EP, Wood WC (2013) Lumpectomy plus tamoxifen with or without irradiation in women age 70 years or older with early breast cancer: long-term follow-up of CALGB 9343. J Clin Oncol 31(19):2382–2387. doi:10.1200/JCO.2012.45.2615
Pesce C, Czechura T, Winchester DJ, Huo D, Winchester DP, Yao K (2013) Axillary surgery among estrogen receptor positive women 70 years of age or older with clinical stage I breast cancer, 2004-2010: a report from the National Cancer Data Base. Ann Surg Oncol 20(10):3259–3265. doi:10.1245/s10434-013-3153-9
International Breast Cancer Study Group, Rudenstam CM, Zahrieh D, Forbes JF, Crivellari D, Holmberg SB, Rey P, Dent D, Campbell I, Bernhard J, Price KN, Castiglione-Gertsch M, Goldhirsch A, Gelber RD, Coates AS (2006) Randomized trial comparing axillary clearance versus no axillary clearance in older patients with breast cancer: first results of International Breast Cancer Study Group Trial 10-93. J Clin Oncol 24(3):337–344. doi:10.1200/JCO.2005.01.5784
Rutter CE, Lester-Coll NH, Mancini BR, Corso CD, Park HS, Yeboa DN, Gross CP, Evans SB (2015) The evolving role of adjuvant radiotherapy for elderly women with early-stage breast cancer. Cancer 121(14):2331–2340. doi:10.1002/cncr.29377
Lerro CC, Robbins AS, Phillips JL, Stewart AK (2013) Comparison of cases captured in the national cancer data base with those in population-based central cancer registries. Ann Surg Oncol 20(6):1759–1765. doi:10.1245/s10434-013-2901-1
About the National Cancer Database. American College of Surgeons web site. https://www.facs.org/quality-programs/cancer/ncdb/about. Accessed June 22, 2017
Edge S, Byrd D, Compton C, Fritz A, Greene F, Trotti A (2010) AJCC cancer staging manual, 7th edn. Springer, France
Facility Oncology Registry Data Standards Manual (2010)
Mathiesen O, Carl J, Bonderup O, Panduro J (1990) Axillary sampling and the risk of erroneous staging of breast cancer. An analysis of 960 consecutive patients. Acta Oncol 29(6):721–725
Axelsson CK, Mouridsen HT, Zedeler K (1992) Axillary dissection of level I and II lymph nodes is important in breast cancer classification. The Danish Breast Cancer Cooperative Group (DBCG). Eur J Cancer 28A(8–9):1415–1418
https://www.nccn.org/professionals/physician_gls/pdf/breast.pdf (2016)
Charlson ME, Pompei P, Ales KL, MacKenzie CR (1987) A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis 40(5):373–383
Society of Surgical Oncology - Don’t routinely use sentinel node biopsy in clinically node negative women ≥ 70 years of age with hormone receptor positive invasive breast cancer. Choosing Wisely web site. http://www.choosingwisely.org/clinician-lists/sso-sentinel-node-biopsy-in-node-negative-women-70-and-over/. Accessed June 22, 2017
Swenson KK, Nissen MJ, Ceronsky C, Swenson L, Lee MW, Tuttle TM (2002) Comparison of side effects between sentinel lymph node and axillary lymph node dissection for breast cancer. Ann Surg Oncol 9(8):745–753
Bafford A, Gadd M, Gu X, Lipsitz S, Golshan M (2010) Diminishing morbidity with the increased use of sentinel node biopsy in breast carcinoma. Am J Surg 200(3):374–377. doi:10.1016/j.amjsurg.2009.10.012
Marrazzo A, Taormina P, David M, Riili I, Lo Gerfo D, Casà L, Noto A, Mercadante S (2007) Surgical treatment of early breast cancer in day surgery. Chir Ital 59(5):687–691
Meguerditchian AN, Stewart A, Roistacher J, Watroba N, Cropp M, Edge SB (2010) Claims data linked to hospital registry data enhance evaluation of the quality of care of breast cancer. J Surg Oncol 101(7):593–599. doi:10.1002/jso.21528
Mallin K, Palis BE, Watroba N, Stewart AK, Walczak D, Singer J, Barron J, Blumenthal W, Haydu G, Edge SB (2013) Completeness of American Cancer Registry Treatment Data: implications for quality of care research. J Am Coll Surg 216(3):428–437. doi:10.1016/j.jamcollsurg.2012.12.016
Freedman RA, Vaz-Luis I, Barry WT, Lii H, Lin NU, Winer EP, Keating NL (2014) Patterns of chemotherapy, toxicity, and short-term outcomes for older women receiving adjuvant trastuzumab-based therapy. Breast Cancer Res Treat 145(2):491–501. doi:10.1007/s10549-014-2968-9
Partridge AH, Hughes ME, Warner ET, Ottesen RA, Wong YN, Edge SB, Theriault RL, Blayney DW, Niland JC, Winer EP, Weeks JC, Tamimi RM (2016) Subtype-dependent relationship between young age at diagnosis and breast cancer survival. J Clin Oncol 34(27):3308–3314. doi:10.1200/JCO.2015.65.8013
Sun J, Chia S (2017) Adjuvant chemotherapy and HER-2-directed therapy for early-stage breast cancer in the elderly. Br J Cancer 116(1):4–9. doi:10.1038/bjc.2016.360
Acknowledgements
The data used in the study are derived from a limited dataset of the National Cancer Data Base (NCDB). The authors acknowledge the efforts of the American College of Surgeons, the Commission on Cancer, and the American Cancer Society in the creation of the National Cancer Data Base. The American College of Surgeons and the Commission on Cancer have not verified and are not responsible for the analytic or statistical methodology employed, or the conclusions drawn from these data by the authors.
Funding
This study was supported by the following: Dr. Freedman is supported by the American Cancer Society (125912-MRSG-14-240-01-CPPB) and Susan G. Komen (CCR CCR14298143).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Dr. Freedman receives institutional funding only from Genentech, Eisai, and Puma Biotechnology. All other authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. For this type of study formal consent is not required.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Dominici, L.S., Sineshaw, H.M., Jemal, A. et al. Patterns of axillary evaluation in older patients with breast cancer and associations with adjuvant therapy receipt. Breast Cancer Res Treat 167, 555–566 (2018). https://doi.org/10.1007/s10549-017-4528-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-017-4528-6