Abstract
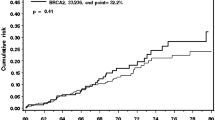
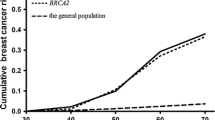
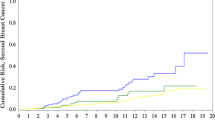
The purpose of this study is to estimate the age-specific annual risks of breast cancer in a woman with a germline BRCA mutation and an affected first-degree relative according to the age of breast cancer diagnosis in the relative. Women with BRCA mutations with no previous diagnosis of breast cancer and with one first-degree relative with breast cancer were followed for breast cancers for a mean of 5.9 years (minimum 2 years). Age-specific annual breast cancer risks were calculated, according to the age of breast cancer diagnosis in the proband and the first-degree relative. 1114 cancer-free women with a BRCA mutation with a single first-degree relative with breast cancer were eligible for the study. 122 women (11.0 %) were diagnosed with incident breast cancer. The annual risk of breast cancer was 2.0 % for women with BRCA1 mutations and was 1.6 % for women with BRCA2 mutations. The age of breast cancer diagnosis in the first-degree relative did not affect the annual breast cancer risks for BRCA1 mutation carriers. For BRCA2 mutation carriers, the annual breast cancer risk was 4.5 % for women with a first-degree relative diagnosed with breast cancer under the age of 30 years and was 0.7 % for women with a relative diagnosed over the age of 60. Among women with BRCA2 mutations, a family history of early-onset breast cancer is a risk factor for developing breast cancer. Risk assessment for healthy BRCA2 mutation carriers should consider the ages of breast cancers diagnosed in first-degree relatives.
Similar content being viewed by others
References
Antoniou A, Pharoah PD, Narod S, Risch HA, Eyfjord JE, Hopper JL, Loman N, Olsson H, Johannsson O, Borg A et al (2003) Average risks of breast and ovarian cancer associated with BRCA1 or BRCA2 mutations detected in case Series unselected for family history: a combined analysis of 22 studies. Am J Hum Genet 72(5):1117–1130
King MC, Marks JH, Mandell JB (2003) Breast and ovarian cancer risks due to inherited mutations in BRCA1 and BRCA2. Science 302(5645):643–646
Cullinane CA, Lubinski J, Neuhausen SL, Ghadirian P, Lynch HT, Isaacs C, Weber B, Moller P, Offit K, Kim-Sing C et al (2005) Effect of pregnancy as a risk factor for breast cancer in BRCA1/BRCA2 mutation carriers. Int J Cancer J Int Cancer 117(6):988–991
Gronwald J, Byrski T, Huzarski T, Cybulski C, Sun P, Tulman A, Narod SA, Lubinski J (2006) Influence of selected lifestyle factors on breast and ovarian cancer risk in BRCA1 mutation carriers from Poland. Breast Cancer Res Treat 95(2):105–109
Antoniou AC, Shenton A, Maher ER, Watson E, Woodward E, Lalloo F, Easton DF, Evans DG (2006) Parity and breast cancer risk among BRCA1 and BRCA2 mutation carriers. Breast Cancer Res 8(6):R72
Kotsopoulos J, Lubinski J, Lynch HT, Klijn J, Ghadirian P, Neuhausen SL, Kim-Sing C, Foulkes WD, Moller P, Isaacs C et al (2007) Age at first birth and the risk of breast cancer in BRCA1 and BRCA2 mutation carriers. Breast Cancer Res Treat 105(2):221–228
Kotsopoulos J, Lubinski J, Lynch HT, Neuhausen SL, Ghadirian P, Isaacs C, Weber B, Kim-Sing C, Foulkes WD, Gershoni-Baruch R et al (2005) Age at menarche and the risk of breast cancer in BRCA1 and BRCA2 mutation carriers. Cancer Causes Control 16(6):667–674
Kotsopoulos J, Lubinski J, Salmena L, Lynch HT, Kim-Sing C, Foulkes WD, Ghadirian P, Neuhausen SL, Demsky R, Tung N et al (2012) Breastfeeding and the risk of breast cancer in BRCA1 and BRCA2 mutation carriers. Breast Cancer Research 14(2):R42
Metcalfe K, Lubinski J, Lynch HT, Ghadirian P, Foulkes WD, Kim-Sing C, Neuhausen S, Tung N, Rosen B, Gronwald J et al (2010) Family History of Cancer and Cancer Risks in Women with BRCA1 or BRCA2 Mutations. J Natl Cancer Inst 102(24):1874–1878
Metcalfe KA, Foulkes WD, Kim-Sing C, Ainsworth P, Rosen B, Armel S, Poll A, Eisen A, Gilchrist D, Chudley A et al (2008) Family history as a predictor of uptake of cancer preventive procedures by women with a BRCA1 or BRCA2 mutation. Clin Genet 73(5):474–479
Metcalfe KA, Finch A, Poll A, Horsman D, Kim-Sing C, Scott J, Royer R, Sun P, Narod SA (2009) Breast cancer risks in women with a family history of breast or ovarian cancer who have tested negative for a BRCA1 or BRCA2 mutation. Br J Cancer 100(2):421–425
Howlader N, Noone AM, Krapcho M, Garshell J, D M, SF A, CL K, M Y, J R, Z T et al (2014) SEER Cancer Statistics Review, 1975–2012. National Cancer Institute, Bethesda
Acknowledgments
Dr. John Semple is supported through the Canadian Breast Cancer Foundation (Ontario Chapter). This work is supported through the Canadian Cancer Society Research Initiative. C.E. is the Sondra J. and Stephen R. Hardis Endowed Chair in Cancer Genomic Medicine at the Cleveland Clinic and an ACS Clinical Research Professor. This study complies with the current laws of the country in which it was performed. This study was carried out in Canada.
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in this study involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
The members of the Hereditary Breast Cancer Clinical Study Group were available in the Appendix.
Appendix: Hereditary breast cancer clinical study group:
Appendix: Hereditary breast cancer clinical study group:
P. Ainsworth, L. Bordeleau, T. Byrski, A. Chudley, F. Couch, C. Cullinane, C. Cybulski, M. Daly, R. Demsky, T. Donenberg, A. Eisen, G. Evans, T. Fallen, E. Friedman, J. Garber, D. Gilchrist, O. Ginsburg, B. Gorski, C. Isaacs, N. Kauff, J. Klijn, E. Lemire, L. Maehle, S. Manoukian, J. McCuaig, W. McKinnon, J. McLennan, W. Meschino, P. Moller, K. Offit, O. Olopade, B. Pasini, D. Rayson, B. Rosen, H. Saal, L. Senter, C. Singer, D. Stoppa–Lyonnet, K. Sweet, E. Warner, J. Weitzel, M. Wood, and D. Zakalik.
Rights and permissions
About this article
Cite this article
Semple, J., Metcalfe, K.A., Lubinski, J. et al. Does the age of breast cancer diagnosis in first-degree relatives impact on the risk of breast cancer in BRCA1 and BRCA2 mutation carriers?. Breast Cancer Res Treat 154, 163–169 (2015). https://doi.org/10.1007/s10549-015-3596-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-015-3596-8