Abstract
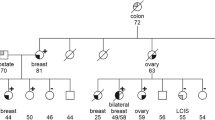
Variants of uncertain clinical significance (VUS) in the high-penetrance breast cancer susceptibility genes BRCA1 and BRCA2 represent a major obstacle in genetic counseling of high-risk breast cancer families. We analyzed a missense VUS located in BRCA2 (p.Asn3124Ile; HGVS: BRCA2 c.9371A > T) present in seven independent high-risk breast cancer families that were counseled and genetically tested in South-West Germany. The VUS was identified by DNA sequencing. We analyzed co-occurrence with deleterious BRCA1/2 mutations, segregation, evolutionary conservation, in silico impact prediction, and prevalence in the general population. All carriers of the VUS suffered from breast or ovarian cancer. In two families, an additional high burden of other cancers such as pancreatic, prostate, and gastric cancers was reported, one further family included two cases of male breast cancer. The VUS did not co-occur with deleterious BRCA1/2 mutations and segregated in two affected individuals of one family. In contrast to the 7/1,347 (0,5 %) tested high-risk BC families without clearly pathogenic mutations in BRCA1/2, none of 3,126 healthy population controls sharing the same ethnic and geographical background were found to carry this VUS (p = 0.0002). In-silico prediction revealed strong evolutionary conservation of the asparagine residue, residing in the C-terminal oligonucleotide-binding-fold-3 region, and a most likely damaging impact of this exchange on the protein structure. The BRCA2 p.Asn3124Ile (BRCA2 c.9371A > T) variant is a rare mutation with a damaging effect on the BRCA2 protein that is strongly associated with familial breast and ovarian cancer risk, indicating its most likely pathogenic nature and clinical relevance.

Similar content being viewed by others
Abbreviations
- VUS:
-
Variant of uncertain clinical significance
- BC:
-
Breast cancer
- OC:
-
Ovarian cancer
- PCC:
-
Pancreatic cancer
- PC:
-
Prostate cancer
- GC:
-
Gastric cancer
- CRC:
-
Colorectal cancer
- LC:
-
Lung cancer
- ER:
-
Estrogen receptor
- PR:
-
Progesterone receptor
- HER2:
-
Human epidermal growth factor receptor 2
References
Jemal A, Bray F, Center MM, Ferlay J, Ward E, Forman D (2011) Global cancer statistics. CA Cancer J Clin 61(2):69–90. doi:10.3322/caac.20107
Balmain A, Gray J, Ponder B (2003) The genetics and genomics of cancer. Nat Genet 33(Suppl):238–244. doi:10.1038/ng1107
Miki Y, Swensen J, Shattuck-Eidens D, Futreal PA, Harshman K, Tavtigian S, Liu Q, Cochran C, Bennett LM, Ding W et al (1994) A strong candidate for the breast and ovarian cancer susceptibility gene BRCA1. Science 266(5182):66–71
Wooster R, Bignell G, Lancaster J, Swift S, Seal S, Mangion J, Collins N, Gregory S, Gumbs C, Micklem G (1995) Identification of the breast cancer susceptibility gene BRCA2. Nature 378(6559):789–792. doi:10.1038/378789a0
Meindl A, German Consortium for Hereditary B, Ovarian C (2002) Comprehensive analysis of 989 patients with breast or ovarian cancer provides BRCA1 and BRCA2 mutation profiles and frequencies for the German population. Int J Cancer 97(4):472–480. doi:10.1002/ijc.1626
Meindl A, Hellebrand H, Wiek C, Erven V, Wappenschmidt B, Niederacher D, Freund M, Lichtner P, Hartmann L, Schaal H, Ramser J, Honisch E, Kubisch C, Wichmann HE, Kast K, Deissler H, Engel C, Muller-Myhsok B, Neveling K, Kiechle M, Mathew CG, Schindler D, Schmutzler RK, Hanenberg H (2010) Germline mutations in breast and ovarian cancer pedigrees establish RAD51C as a human cancer susceptibility gene. Nat Genet 42(5):410–414. doi:10.1038/ng.569
Seal S, Thompson D, Renwick A, Elliott A, Kelly P, Barfoot R, Chagtai T, Jayatilake H, Ahmed M, Spanova K, North B, McGuffog L, Evans DG, Eccles D, Breast Cancer Susceptibility C, Easton DF, Stratton MR, Rahman N (2006) Truncating mutations in the Fanconi anemia J gene BRIP1 are low-penetrance breast cancer susceptibility alleles. Nat Genet 38(11):1239–1241. doi:10.1038/ng1902
Rahman N, Seal S, Thompson D, Kelly P, Renwick A, Elliott A, Reid S, Spanova K, Barfoot R, Chagtai T, Jayatilake H, McGuffog L, Hanks S, Evans DG, Eccles D, Breast Cancer Susceptibility C, Easton DF, Stratton MR (2007) PALB2, which encodes a BRCA2-interacting protein, is a breast cancer susceptibility gene. Nat Genet 39(2):165–167. doi:10.1038/ng1959
Loveday C, Turnbull C, Ramsay E, Hughes D, Ruark E, Frankum JR, Bowden G, Kalmyrzaev B, Warren-Perry M, Snape K, Adlard JW, Barwell J, Berg J, Brady AF, Brewer C, Brice G, Chapman C, Cook J, Davidson R, Donaldson A, Douglas F, Greenhalgh L, Henderson A, Izatt L, Kumar A, Lalloo F, Miedzybrodzka Z, Morrison PJ, Paterson J, Porteous M, Rogers MT, Shanley S, Walker L, Breast Cancer Susceptibility C, Eccles D, Evans DG, Renwick A, Seal S, Lord CJ, Ashworth A, Reis-Filho JS, Antoniou AC, Rahman N (2011) Germline mutations in RAD51D confer susceptibility to ovarian cancer. Nat Genet 43(9):879–882. doi:10.1038/ng.893
Gonzalez KD, Noltner KA, Buzin CH, Gu D, Wen-Fong CY, Nguyen VQ, Han JH, Lowstuter K, Longmate J, Sommer SS, Weitzel JN (2009) Beyond Li Fraumeni Syndrome: clinical characteristics of families with p53 germline mutations. J Clin Oncol 27(8):1250–1256. doi:10.1200/JCO.2008.16.6959
Meijers-Heijboer H, van den Ouweland A, Klijn J, Wasielewski M, de Snoo A, Oldenburg R, Hollestelle A, Houben M, Crepin E, van Veghel-Plandsoen M, Elstrodt F, van Duijn C, Bartels C, Meijers C, Schutte M, McGuffog L, Thompson D, Easton D, Sodha N, Seal S, Barfoot R, Mangion J, Chang-Claude J, Eccles D, Eeles R, Evans DG, Houlston R, Murday V, Narod S, Peretz T, Peto J, Phelan C, Zhang HX, Szabo C, Devilee P, Goldgar D, Futreal PA, Nathanson KL, Weber B, Rahman N, Stratton MR, Consortium CH-BC (2002) Low-penetrance susceptibility to breast cancer due to CHEK2(*)1100delC in noncarriers of BRCA1 or BRCA2 mutations. Nat Genet 31(1):55–59. doi:10.1038/ng879
FitzGerald MG, Marsh DJ, Wahrer D, Bell D, Caron S, Shannon KE, Ishioka C, Isselbacher KJ, Garber JE, Eng C, Haber DA (1998) Germline mutations in PTEN are an infrequent cause of genetic predisposition to breast cancer. Oncogene 17(6):727–731. doi:10.1038/sj.onc.1201984
Hearle N, Schumacher V, Menko FH, Olschwang S, Boardman LA, Gille JJ, Keller JJ, Westerman AM, Scott RJ, Lim W, Trimbath JD, Giardiello FM, Gruber SB, Offerhaus GJ, de Rooij FW, Wilson JH, Hansmann A, Moslein G, Royer-Pokora B, Vogel T, Phillips RK, Spigelman AD, Houlston RS (2006) Frequency and spectrum of cancers in the Peutz-Jeghers syndrome. Clin Cancer Res 12(10):3209–3215. doi:10.1158/1078-0432.CCR-06-0083
Renwick A, Thompson D, Seal S, Kelly P, Chagtai T, Ahmed M, North B, Jayatilake H, Barfoot R, Spanova K, McGuffog L, Evans DG, Eccles D, Breast Cancer Susceptibility C, Easton DF, Stratton MR, Rahman N (2006) ATM mutations that cause ataxia-telangiectasia are breast cancer susceptibility alleles. Nat Genet 38(8):873–875. doi:10.1038/ng1837
Antoniou A, Pharoah PD, Narod S, Risch HA, Eyfjord JE, Hopper JL, Loman N, Olsson H, Johannsson O, Borg A, Pasini B, Radice P, Manoukian S, Eccles DM, Tang N, Olah E, Anton-Culver H, Warner E, Lubinski J, Gronwald J, Gorski B, Tulinius H, Thorlacius S, Eerola H, Nevanlinna H, Syrjakoski K, Kallioniemi OP, Thompson D, Evans C, Peto J, Lalloo F, Evans DG, Easton DF (2003) Average risks of breast and ovarian cancer associated with BRCA1 or BRCA2 mutations detected in case Series unselected for family history: a combined analysis of 22 studies. Am J Hum Genet 72(5):1117–1130. doi:10.1086/375033
King MC, Marks JH, Mandell JB, New York Breast Cancer Study G (2003) Breast and ovarian cancer risks due to inherited mutations in BRCA1 and BRCA2. Science 302(5645):643–646. doi:10.1126/science.1088759
Callahan MJ, Crum CP, Medeiros F, Kindelberger DW, Elvin JA, Garber JE, Feltmate CM, Berkowitz RS, Muto MG (2007) Primary fallopian tube malignancies in BRCA-positive women undergoing surgery for ovarian cancer risk reduction. J Clin Oncol 25(25):3985–3990. doi:10.1200/JCO.2007.12.2622
Thompson D, Easton DF, Breast Cancer Linkage C (2002) Cancer Incidence in BRCA1 mutation carriers. J Natl Cancer Inst 94(18):1358–1365
Iqbal J, Ragone A, Lubinski J, Lynch HT, Moller P, Ghadirian P, Foulkes WD, Armel S, Eisen A, Neuhausen SL, Senter L, Singer CF, Ainsworth P, Kim-Sing C, Tung N, Friedman E, Llacuachaqui M, Ping S, Narod SA, Hereditary Breast Cancer Study G (2012) The incidence of pancreatic cancer in BRCA1 and BRCA2 mutation carriers. Br J Cancer 107(12):2005–2009. doi:10.1038/bjc.2012.483
Hahn SA, Greenhalf B, Ellis I, Sina-Frey M, Rieder H, Korte B, Gerdes B, Kress R, Ziegler A, Raeburn JA, Campra D, Grutzmann R, Rehder H, Rothmund M, Schmiegel W, Neoptolemos JP, Bartsch DK (2003) BRCA2 germline mutations in familial pancreatic carcinoma. J Natl Cancer Inst 95(3):214–221
Gallagher DJ, Gaudet MM, Pal P, Kirchhoff T, Balistreri L, Vora K, Bhatia J, Stadler Z, Fine SW, Reuter V, Zelefsky M, Morris MJ, Scher HI, Klein RJ, Norton L, Eastham JA, Scardino PT, Robson ME, Offit K (2010) Germline BRCA mutations denote a clinicopathologic subset of prostate cancer. Clin Cancer Res 16(7):2115–2121. doi:10.1158/1078-0432.CCR-09-2871
Rath MG, Fathali-Zadeh F, Langheinz A, Tchatchou S, Voigtlander T, Heil J, Golatta M, Schott S, Drasseck T, Behnecke A, Burgemeister AL, Evers C, Bugert P, Junkermann H, Schneeweiss A, Bartram CR, Sohn C, Sutter C, Burwinkel B (2012) Molecular and clinical characterization of an in frame deletion of uncertain clinical significance in the BRCA2 gene. Breast Cancer Res Treat 133(2):725–734. doi:10.1007/s10549-011-1917-0
Moran A, O’Hara C, Khan S, Shack L, Woodward E, Maher ER, Lalloo F, Evans DG (2012) Risk of cancer other than breast or ovarian in individuals with BRCA1 and BRCA2 mutations. Fam Cancer 11(2):235–242. doi:10.1007/s10689-011-9506-2
Antoniou AC, Beesley J, McGuffog L, Sinilnikova OM, Healey S, Neuhausen SL, Ding YC, Rebbeck TR, Weitzel JN, Lynch HT, Isaacs C, Ganz PA, Tomlinson G, Olopade OI, Couch FJ, Wang X, Lindor NM, Pankratz VS, Radice P, Manoukian S, Peissel B, Zaffaroni D, Barile M, Viel A, Allavena A, Dall’Olio V, Peterlongo P, Szabo CI, Zikan M, Claes K, Poppe B, Foretova L, Mai PL, Greene MH, Rennert G, Lejbkowicz F, Glendon G, Ozcelik H, Andrulis IL, Ontario Cancer Genetics N, Thomassen M, Gerdes AM, Sunde L, Cruger D, Birk Jensen U, Caligo M, Friedman E, Kaufman B, Laitman Y, Milgrom R, Dubrovsky M, Cohen S, Borg A, Jernstrom H, Lindblom A, Rantala J, Stenmark-Askmalm M, Melin B, Swe B, Nathanson K, Domchek S, Jakubowska A, Lubinski J, Huzarski T, Osorio A, Lasa A, Duran M, Tejada MI, Godino J, Benitez J, Hamann U, Kriege M, Hoogerbrugge N, van der Luijt RB, van Asperen CJ, Devilee P, Meijers-Heijboer EJ, Blok MJ, Aalfs CM, Hogervorst F, Rookus M, Hebon, Cook M, Oliver C, Frost D, Conroy D, Evans DG, Lalloo F, Pichert G, Davidson R, Cole T, Cook J, Paterson J, Hodgson S, Morrison PJ, Porteous ME, Walker L, Kennedy MJ, Dorkins H, Peock S, Embrace, Godwin AK, Stoppa-Lyonnet D, de Pauw A, Mazoyer S, Bonadona V, Lasset C, Dreyfus H, Leroux D, Hardouin A, Berthet P, Faivre L, Gemo, Loustalot C, Noguchi T, Sobol H, Rouleau E, Nogues C, Frenay M, Venat-Bouvet L, Gemo, Hopper JL, Daly MB, Terry MB, John EM, Buys SS, Yassin Y, Miron A, Goldgar D, Breast Cancer Family R, Singer CF, Dressler AC, Gschwantler-Kaulich D, Pfeiler G, Hansen TV, Jonson L, Agnarsson BA, Kirchhoff T, Offit K, Devlin V, Dutra-Clarke A, Piedmonte M, Rodriguez GC, Wakeley K, Boggess JF, Basil J, Schwartz PE, Blank SV, Toland AE, Montagna M, Casella C, Imyanitov E, Tihomirova L, Blanco I, Lazaro C, Ramus SJ, Sucheston L, Karlan BY, Gross J, Schmutzler R, Wappenschmidt B, Engel C, Meindl A, Lochmann M, Arnold N, Heidemann S, Varon-Mateeva R, Niederacher D, Sutter C, Deissler H, Gadzicki D, Preisler-Adams S, Kast K, Schonbuchner I, Caldes T, de la Hoya M, Aittomaki K, Nevanlinna H, Simard J, Spurdle AB, Holland H, Chen X, kConFab, Platte R, Chenevix-Trench G, Easton DF, Cimba (2010) Common breast cancer susceptibility alleles and the risk of breast cancer for BRCA1 and BRCA2 mutation carriers: implications for risk prediction. Cancer Res 70(23):9742–9754. doi:10.1158/0008-5472.CAN-10-1907
Roy R, Chun J, Powell SN (2012) BRCA1 and BRCA2: different roles in a common pathway of genome protection. Nat Rev Cancer 12(1):68–78. doi:10.1038/nrc3181
Howlett NG, Taniguchi T, Olson S, Cox B, Waisfisz Q, De Die-Smulders C, Persky N, Grompe M, Joenje H, Pals G, Ikeda H, Fox EA, D’Andrea AD (2002) Biallelic inactivation of BRCA2 in Fanconi anemia. Science 297(5581):606–609. doi:10.1126/science.1073834
Gowen LC, Johnson BL, Latour AM, Sulik KK, Koller BH (1996) Brca1 deficiency results in early embryonic lethality characterized by neuroepithelial abnormalities. Nat Genet 12(2):191–194. doi:10.1038/ng0296-191
Balmana J, Diez O, Rubio IT, Cardoso F, Group EGW (2011) BRCA in breast cancer: ESMO clinical practice guidelines. Ann Oncol Off J Eur Soc Med Oncol/ESMO 22(Suppl 6):vi31–34. doi:10.1093/annonc/mdr373
Domchek SM, Friebel TM, Singer CF, Evans DG, Lynch HT, Isaacs C, Garber JE, Neuhausen SL, Matloff E, Eeles R, Pichert G, Van t’veer L, Tung N, Weitzel JN, Couch FJ, Rubinstein WS, Ganz PA, Daly MB, Olopade OI, Tomlinson G, Schildkraut J, Blum JL, Rebbeck TR (2010) Association of risk-reducing surgery in BRCA1 or BRCA2 mutation carriers with cancer risk and mortality. JAMA J Am Med Assoc 304(9):967–975. doi:10.1001/jama.2010.1237
King MC, Wieand S, Hale K, Lee M, Walsh T, Owens K, Tait J, Ford L, Dunn BK, Costantino J, Wickerham L, Wolmark N, Fisher B, National Surgical Adjuvant B, Bowel P (2001) Tamoxifen and breast cancer incidence among women with inherited mutations in BRCA1 and BRCA2: national surgical adjuvant breast and bowel project (NSABP-P1) breast cancer prevention trial. JAMA J Am Med Assoc 286(18):2251–2256
Bryant HE, Schultz N, Thomas HD, Parker KM, Flower D, Lopez E, Kyle S, Meuth M, Curtin NJ, Helleday T (2005) Specific killing of BRCA2-deficient tumours with inhibitors of poly(ADP-ribose) polymerase. Nature 434(7035):913–917. doi:10.1038/nature03443
Rottenberg S, Jaspers JE, Kersbergen A, van der Burg E, Nygren AO, Zander SA, Derksen PW, de Bruin M, Zevenhoven J, Lau A, Boulter R, Cranston A, O’Connor MJ, Martin NM, Borst P, Jonkers J (2008) High sensitivity of BRCA1-deficient mammary tumors to the PARP inhibitor AZD2281 alone and in combination with platinum drugs. Proc Natl Acad Sci USA 105(44):17079–17084. doi:10.1073/pnas.0806092105
Radice P, De Summa S, Caleca L, Tommasi S (2011) Unclassified variants in BRCA genes: guidelines for interpretation. Ann Oncol Off J Eur Soc Med Oncol/ESMO 22(Suppl 1):i18–i23. doi:10.1093/annonc/mdq661
Frank TS, Deffenbaugh AM, Reid JE, Hulick M, Ward BE, Lingenfelter B, Gumpper KL, Scholl T, Tavtigian SV, Pruss DR, Critchfield GC (2002) Clinical characteristics of individuals with germline mutations in BRCA1 and BRCA2: analysis of 10,000 individuals. J Clin Oncol 20(6):1480–1490
Miller SA, Dykes DD, Polesky HF (1988) A simple salting out procedure for extracting DNA from human nucleated cells. Nucleic Acids Res 16(3):1215
Benson DA, Cavanaugh M, Clark K, Karsch-Mizrachi I, Lipman DJ, Ostell J, Sayers EW (2013) GenBank. Nucleic Acids Res 41(Database issue):D36–42. doi:10.1093/nar/gks1195
Henikoff S, Henikoff JG (1992) Amino acid substitution matrices from protein blocks. Proc Natl Acad Sci USA 89(22):10915–10919
Luu TD, Rusu AM, Walter V, Ripp R, Moulinier L, Muller J, Toursel T, Thompson JD, Poch O, Nguyen H (2012) MSV3d: database of human misSense variants mapped to 3D protein structure. Database J Biol Databases Curation 2012 bas018. doi:10.1093/database/bas018
Schwarz JM, Rodelsperger C, Schuelke M, Seelow D (2010) MutationTaster evaluates disease-causing potential of sequence alterations. Nat Methods 7(8):575–576. doi:10.1038/nmeth0810-575
Reva B, Antipin Y, Sander C (2011) Predicting the functional impact of protein mutations: application to cancer genomics. Nucleic Acids Res 39(17):e118. doi:10.1093/nar/gkr407
Kanehisa M, Goto S (2000) KEGG: kyoto encyclopedia of genes and genomes. Nucleic Acids Res 28(1):27–30
Tavtigian SV, Deffenbaugh AM, Yin L, Judkins T, Scholl T, Samollow PB, de Silva D, Zharkikh A, Thomas A (2006) Comprehensive statistical study of 452 BRCA1 missense substitutions with classification of eight recurrent substitutions as neutral. J Med Genet 43(4):295–305. doi:10.1136/jmg.2005.033878
Szabo C, Masiello A, Ryan JF, Brody LC (2000) The breast cancer information core: database design, structure, and scope. Hum Mutat 16(2):123–131. doi:10.1002/1098-1004(200008)16:2<123:AID-HUMU4>3.0.CO;2-Y
Fokkema IF, Taschner PE, Schaafsma GC, Celli J, Laros JF, den Dunnen JT (2011) LOVD v. 2.0: the next generation in gene variant databases. Hum Mutat 32(5):557–563. doi:10.1002/humu.21438
Beroud C, Collod-Beroud G, Boileau C, Soussi T, Junien C (2000) UMD (universal mutation database): a generic software to build and analyze locus-specific databases. Hum Mutat 15(1):86–94. doi:10.1002/(SICI)1098-1004(200001)15:1<86:AID-HUMU16>3.0.CO;2-4
Fischer C, Engel C, Sutter C, Zachariae S, Schmutzler R, Meindl A, Heidemann S, Grimm T, Goecke TO, Debatin I, Horn D, Wieacker P, Gadzicki D, Becker K, Schafer D, Stock F, Voigtlander T, German Consortium for Hereditary B, Ovarian C (2012) BRCA1/2 testing: uptake, phenocopies, and strategies to improve detection rates in initially negative families. Clin Genet 82(5):478–483. doi:10.1111/j.1399-0004.2011.01788.x
Waterhouse AM, Procter JB, Martin DM, Clamp M, Barton GJ (2009) Jalview version 2–a multiple sequence alignment editor and analysis workbench. Bioinformatics 25(9):1189–1191. doi:10.1093/bioinformatics/btp033
Yang H, Jeffrey PD, Miller J, Kinnucan E, Sun Y, Thoma NH, Zheng N, Chen PL, Lee WH, Pavletich NP (2002) BRCA2 function in DNA binding and recombination from a BRCA2-DSS1-ssDNA structure. Science 297(5588):1837–1848. doi:10.1126/science.297.5588.1837
Becker AA, Graeser MK, Landwehr C, Hilger T, Baus W, Wappenschmidt B, Meindl A, Weber RG, Schmutzler RK (2012) A 24-color metaphase-based radiation assay discriminates heterozygous BRCA2 mutation carriers from controls by chromosomal radiosensitivity. Breast Cancer Res Treat 135(1):167–175. doi:10.1007/s10549-012-2119-0
Karchin R, Agarwal M, Sali A, Couch F, Beattie MS (2008) Classifying Variants of Undetermined Significance in BRCA2 with protein likelihood ratios. Cancer Inform 6:203–216
Guidugli L, Pankratz VS, Singh N, Thompson J, Erding CA, Engel C, Schmutzler R, Domchek S, Nathanson K, Radice P, Singer C, Tonin PN, Lindor NM, Goldgar DE, Couch FJ (2013) A classification model for BRCA2 DNA binding domain missense variants based on homology-directed repair activity. Cancer Res 73(1):265–275. doi:10.1158/0008-5472.CAN-12-2081
Kwiatkowska E, Teresiak M, Lamperska KM, Karczewska A, Breborowicz D, Stawicka M, Godlewski D, Krzyzosiak WJ, Mackiewicz A (2001) BRCA2 germline mutations in male breast cancer patients in the Polish population. Hum Mutat 17(1):73. doi:10.1002/1098-1004(2001)17:1<73:AID-HUMU12>3.0.CO;2-O
Couch FJ, Johnson MR, Rabe KG, Brune K, de Andrade M, Goggins M, Rothenmund H, Gallinger S, Klein A, Petersen GM, Hruban RH (2007) The prevalence of BRCA2 mutations in familial pancreatic cancer. Cancer Epidemiol Biomarkers Prev 16(2):342–346. doi:10.1158/1055-9965.EPI-06-0783
Breast Cancer Linkage C (1999) Cancer risks in BRCA2 mutation carriers. J Natl Cancer Inst 91(15):1310–1316
van Asperen CJ, Brohet RM, Meijers-Heijboer EJ, Hoogerbrugge N, Verhoef S, Vasen HF, Ausems MG, Menko FH, Gomez Garcia EB, Klijn JG, Hogervorst FB, van Houwelingen JC, van’t Veer LJ, Rookus MA, van Leeuwen FE, Netherlands Collaborative Group on Hereditary Breast C (2005) Cancer risks in BRCA2 families: estimates for sites other than breast and ovary. J Med Genet 42(9):711–719. doi:10.1136/jmg.2004.028829
Levy-Lahad E, Friedman E (2007) Cancer risks among BRCA1 and BRCA2 mutation carriers. Br J Cancer 96(1):11–15. doi:10.1038/sj.bjc.6603535
Jakubowska A, Nej K, Huzarski T, Scott RJ, Lubinski J (2002) BRCA2 gene mutations in families with aggregations of breast and stomach cancers. Br J Cancer 87(8):888–891. doi:10.1038/sj.bjc.6600562
Liede A, Karlan BY, Narod SA (2004) Cancer risks for male carriers of germline mutations in BRCA1 or BRCA2: a review of the literature. J Clin Oncol 22(4):735–742. doi:10.1200/JCO.2004.05.055
Easton DF, Steele L, Fields P, Ormiston W, Averill D, Daly PA, McManus R, Neuhausen SL, Ford D, Wooster R, Cannon-Albright LA, Stratton MR, Goldgar DE (1997) Cancer risks in two large breast cancer families linked to BRCA2 on chromosome 13q12-13. Am J Hum Genet 61(1):120–128
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Harald Martin Surowy, Christian Sutter and Barbara Burwinkel have contributed equally to this study.
Rights and permissions
About this article
Cite this article
Surowy, H.M., Sutter, C., Mittnacht, M. et al. Clinical and molecular characterization of the BRCA2 p.Asn3124Ile variant reveals substantial evidence for pathogenic significance. Breast Cancer Res Treat 145, 451–460 (2014). https://doi.org/10.1007/s10549-014-2943-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-014-2943-5